Revolutionizing Phenanthridine Synthesis: Electrochemical Routes for High-Purity Pharmaceutical Intermediates
The landscape of heterocyclic chemistry is undergoing a significant transformation driven by the need for greener, more efficient synthetic methodologies. Patent CN110684989B introduces a groundbreaking electrochemical approach for the synthesis of 6-azidomethylphenanthridine compounds, a class of molecules with profound implications in medicinal chemistry and functional material science. Unlike traditional thermal or photochemical pathways that often demand harsh conditions and expensive reagents, this innovation leverages electricity as a clean reagent to drive oxidative cyclization at room temperature. For R&D directors and procurement strategists, this represents a pivotal shift towards sustainable manufacturing, offering a route that drastically simplifies the operational complexity while maintaining high yields exceeding 70%. The ability to construct the phenanthridine core directly from distyryl azide precursors without intermediate isolation marks a substantial advancement in step economy.
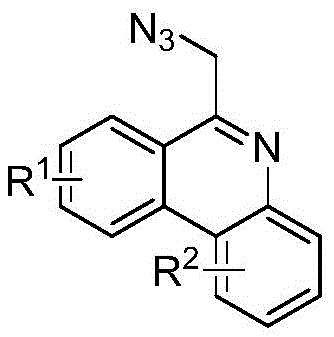
As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented technologies is crucial for securing supply chains against regulatory and cost pressures. The electrochemical method described not only reduces the environmental footprint by utilizing a benign acetonitrile-water solvent system but also enhances process safety by avoiding the high-temperature conditions typically required for azide transformations. This technical insight report delves deep into the mechanistic advantages and commercial viability of this process, providing a comprehensive analysis for stakeholders looking to optimize their production of complex nitrogen-containing heterocycles. By adopting this methodology, manufacturers can achieve cost reduction in pharmaceutical intermediates manufacturing through reduced energy consumption and simplified downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 6-azidomethylphenanthridine derivatives has been plagued by inefficient multi-step sequences that rely on hazardous chemical oxidants and extreme thermal conditions. The conventional pathway, as illustrated in prior art, typically involves the initial synthesis of a 6-methylphenanthridine scaffold followed by a radical bromination using N-bromosuccinimide (NBS) under reflux or photochemical irradiation. This bromination step is notoriously difficult to control, often leading to poly-brominated impurities and requiring rigorous purification to ensure product quality. Furthermore, the subsequent nucleophilic substitution with sodium azide must be conducted with extreme caution due to the potential instability of organic azides at elevated temperatures.
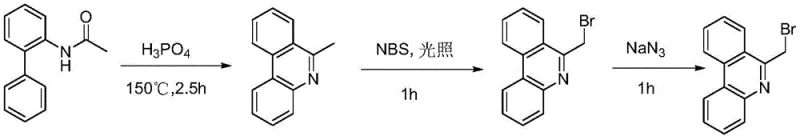
The reliance on stoichiometric amounts of brominating agents generates significant amounts of succinimide waste, complicating waste management and increasing the overall cost of goods sold. Additionally, the requirement for high temperatures, often reaching 150°C in cyclization steps, imposes severe demands on reactor materials and energy infrastructure. For supply chain heads, these factors translate into longer lead times, higher safety compliance costs, and increased vulnerability to raw material price fluctuations for specialized reagents like NBS. The cumulative effect of these inefficiencies makes the conventional route less attractive for large-scale commercial production where margin compression is a constant threat.
The Novel Approach
In stark contrast, the electrochemical synthesis method disclosed in the patent offers a streamlined, one-pot solution that bypasses the need for pre-functionalized intermediates and harsh chemical oxidants. By utilizing an undivided electrolytic cell equipped with a graphite anode and a platinum cathode, the reaction proceeds via anodic oxidation to generate reactive radical cation species that spontaneously undergo intramolecular cyclization. This elegant mechanism allows for the direct conversion of olefin azides into the target phenanthridine structure under mild conditions, specifically at room temperature and ambient pressure. The elimination of external chemical oxidants not only reduces reagent costs but also simplifies the reaction mixture, thereby facilitating easier product isolation.
The operational simplicity of this novel approach is a game-changer for commercial scale-up of complex pharmaceutical intermediates. The use of a mixed solvent system comprising acetonitrile and water in a 10:1 volume ratio provides an optimal balance between substrate solubility and electrolyte conductivity, ensuring consistent reaction performance. Moreover, the application of a constant low voltage (2.2V-2.5V) ensures precise control over the reaction kinetics, minimizing side reactions and maximizing selectivity. This level of control is essential for producing high-purity OLED material or API precursors where trace impurities can compromise downstream biological activity or material performance. The robustness of this method across various substituted substrates demonstrates its versatility as a platform technology for diverse chemical libraries.
Mechanistic Insights into Electrochemical Oxidative Cyclization
The core of this technological breakthrough lies in the unique electrochemical mechanism that facilitates the formation of the carbon-nitrogen bond without the need for transition metal catalysts. Upon applying a constant potential, the olefin azide substrate undergoes single-electron oxidation at the anode surface to form a radical cation intermediate. This highly reactive species triggers a cascade of intramolecular cyclization events, where the aromatic ring attacks the activated vinyl group, closing the phenanthridine ring system. Simultaneously, the azide moiety remains intact or participates in stabilizing the transition state, ultimately leading to the formation of the 6-azidomethyl group. The presence of sodium azide in the electrolyte serves a dual purpose: it acts as a supporting electrolyte component and potentially stabilizes cationic intermediates through weak coordination.
From an impurity control perspective, this electrochemical pathway offers distinct advantages over thermal radical processes. In traditional thermal methods, homolytic cleavage of bonds can lead to uncontrolled radical propagation and the formation of dimerization or polymerization byproducts. However, the electrochemical generation of radicals is confined to the electrode surface and is strictly governed by the applied potential, effectively suppressing uncontrolled chain reactions. This spatial and energetic confinement results in a cleaner reaction profile with fewer side products, reducing the burden on purification units. For R&D teams, this means that the crude product often requires minimal workup, such as simple extraction and drying, before achieving specification-grade purity, significantly accelerating the development timeline for new drug candidates.
How to Synthesize 6-Azidomethylphenanthridine Efficiently
Implementing this electrochemical protocol requires careful attention to electrode configuration and electrolyte composition to maximize yield and reproducibility. The patent details a robust procedure where the substrate concentration is maintained at approximately 0.05 mol/L to ensure efficient mass transfer to the electrode surface. The choice of lithium perchlorate as the supporting electrolyte is critical for maintaining conductivity in the acetonitrile-water mixture without interfering with the redox potentials of the organic substrates. Operators should note that the reaction time is relatively short, typically completing within 3 to 5 hours, which enhances throughput in a batch processing environment. Detailed standardized synthesis steps see the guide below.
- Prepare the electrolyte solution by dissolving the olefin azide substrate and sodium azide in a mixed solvent of acetonitrile and water (10: 1 ratio) with lithium perchlorate.
- Insert a graphite anode and a platinum cathode into the undivided cell and apply a constant DC voltage of 2.3V for 3 hours at room temperature.
- Quench the reaction with water, extract the organic phase with ethyl acetate, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical synthesis route presents a compelling value proposition centered on cost efficiency and operational resilience. The primary driver of cost reduction is the elimination of expensive stoichiometric oxidants and transition metal catalysts, which are often subject to volatile market pricing and supply constraints. By replacing these chemical reagents with electricity, the variable cost per kilogram of product is significantly lowered, improving overall profit margins. Furthermore, the simplified workflow reduces the labor hours required for reaction monitoring and workup, contributing to additional operational savings that accumulate over large production volumes.
- Cost Reduction in Manufacturing: The economic benefits of this process extend beyond mere reagent savings; the absence of heavy metal catalysts eliminates the need for costly metal scavenging resins and extensive purification protocols. In traditional catalytic cycles, removing trace metals to meet ppm-level specifications for pharmaceutical ingredients can account for a substantial portion of the total processing cost. By utilizing an electrochemical method that relies on electron transfer, the final product is inherently free from metal contamination, streamlining the quality control process. This reduction in downstream processing complexity translates directly into lower utility consumption and reduced waste disposal fees, creating a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Dependence on specialized reagents like N-bromosuccinimide or exotic ligands introduces single points of failure in the supply chain, where disruptions can halt production entirely. The electrochemical method relies on commodity chemicals such as sodium azide, acetonitrile, and graphite electrodes, all of which are widely available from multiple global suppliers. This diversification of the raw material base enhances supply security and reduces the risk of production stoppages due to vendor-specific issues. Additionally, the mild reaction conditions reduce the wear and tear on reactor vessels and ancillary equipment, extending asset life and minimizing unplanned maintenance downtime that could impact delivery schedules.
- Scalability and Environmental Compliance: Scaling electrochemical processes is increasingly feasible with modern flow chemistry technologies, allowing for seamless transition from pilot plant to full commercial production. The undivided cell design described in the patent is particularly amenable to scale-up, as it avoids the complexities associated with membrane separation in divided cells. From an environmental standpoint, the use of water as a co-solvent and the generation of minimal chemical waste align with green chemistry principles, facilitating easier regulatory approval and permitting. This sustainability profile is becoming a critical factor for multinational corporations aiming to meet their carbon neutrality goals, making suppliers who adopt such technologies preferred partners in the global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on safety, scalability, and product quality. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines. The responses highlight the practical advantages that differentiate this method from legacy synthetic routes.
Q: How does the electrochemical method improve safety compared to traditional azide synthesis?
A: Traditional methods often require high temperatures (up to 150°C) and hazardous brominating agents like NBS, which pose explosion risks with azides. This electrochemical protocol operates at room temperature, significantly mitigating thermal runaway risks associated with energetic azido groups.
Q: Is this electrochemical process scalable for industrial production?
A: Yes, the process utilizes an undivided cell with inexpensive electrode materials (graphite and platinum) and avoids complex photochemical equipment. The simplicity of the setup and the use of common solvents like acetonitrile facilitate straightforward scale-up from laboratory to commercial tonnage.
Q: What are the purity advantages of avoiding transition metal catalysts?
A: By relying on electrons as the primary reagent rather than transition metal catalysts, the final product is free from heavy metal residues. This eliminates the need for expensive and time-consuming metal scavenging steps, ensuring high-purity specifications suitable for sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Azidomethylphenanthridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-value heterocyclic intermediates. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering products with stringent purity specifications, leveraging our rigorous QC labs to verify that every batch meets the exacting standards required by the global pharmaceutical industry. Our capability to handle complex electrochemical setups allows us to offer this advanced synthesis route as a premium service to our clients.
We invite you to collaborate with us to explore how this technology can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals. Let us be your partner in driving innovation and efficiency in the synthesis of next-generation pharmaceutical intermediates.
