Scalable Synthesis of Omarigliptin Chiral Intermediates for Global Pharmaceutical Manufacturing
Introduction to Advanced Omarigliptin Intermediate Manufacturing
The pharmaceutical landscape for Type 2 diabetes treatment has been significantly advanced by the development of super-long-acting dipeptidyl peptidase-4 (DPP-4) inhibitors, specifically Omarigliptin (development code MK-3102). This potent oral hypoglycemic agent offers a unique once-weekly dosing regimen, providing continuous enzyme inhibition without the risks of weight gain or hypoglycemia associated with older therapies. The chemical structure, characterized by a complex chiral tetrahydropyran core linked to a difluorophenyl group and a pyrrolopyrazole moiety, presents significant synthetic challenges for large-scale production. As detailed in patent CN107459501B, overcoming these challenges requires a departure from traditional, linear syntheses towards more convergent and stereo-controlled strategies. The ability to manufacture the key chiral intermediate efficiently is the linchpin for reliable supply chains globally.
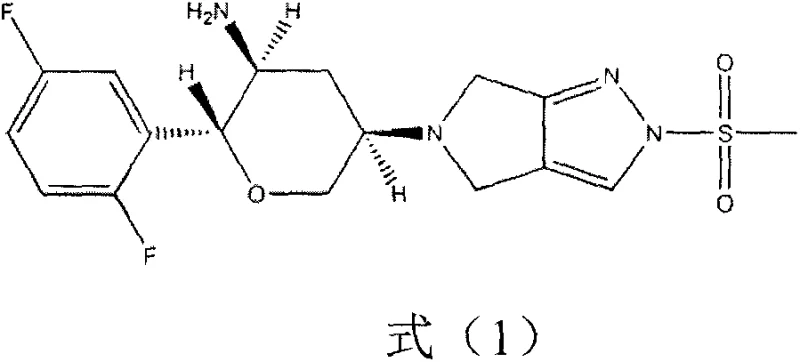
This technical insight report analyzes a novel preparation method disclosed in the aforementioned patent, which addresses critical bottlenecks in the existing supply chain. By leveraging robust organic transformations such as asymmetric aldol condensation and ring-closing metathesis, this route offers a viable pathway for cost reduction in pharmaceutical intermediate manufacturing. For R&D directors and procurement specialists, understanding the mechanistic nuances and commercial implications of this route is essential for securing a competitive edge in the production of next-generation antidiabetic medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the chiral tetrahydropyran core found in Omarigliptin has relied on pathways that are chemically elegant but commercially inefficient. Two prominent prior art routes, described in WO2016127916 and US20090187028, illustrate these systemic inefficiencies. A primary bottleneck in these conventional methods is the dependence on Boc-L-propargyl glycine as a starting material. This specific amino acid derivative is not only difficult to procure in bulk quantities but also commands a high market price due to its own complex synthesis requiring three to four preparatory steps. Furthermore, these legacy routes frequently employ noble metal catalysts in early-stage cyclization reactions, such as the DRK reaction, which can lead to the formation of non-corresponding isomers, complicating downstream purification and reducing overall yield.
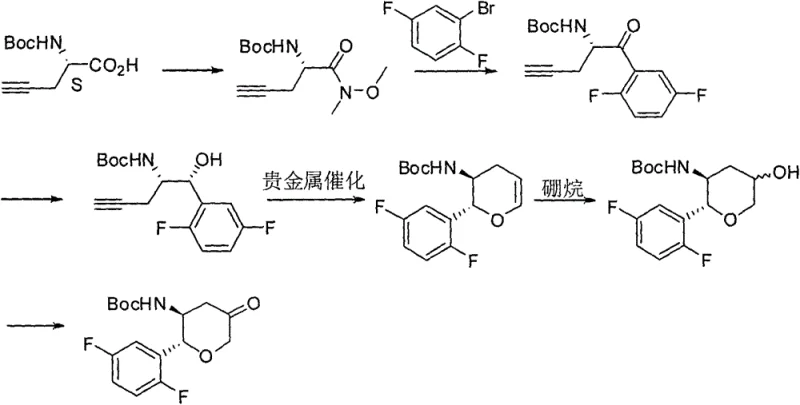
The reliance on such specialized and expensive starting materials creates a fragile supply chain vulnerable to raw material shortages and price volatility. Additionally, the use of sensitive organometallic reagents often necessitates stringent anhydrous conditions and low-temperature operations that are difficult to maintain consistently in multi-ton reactor environments. These factors collectively contribute to a high cost of goods sold (COGS) and extended lead times, making the conventional approach less attractive for generic manufacturers aiming to enter the market with competitive pricing structures.
The Novel Approach
In stark contrast, the novel methodology presented in CN107459501B introduces a paradigm shift by utilizing inexpensive, commodity-grade chemicals as the foundation for the synthesis. The new route initiates with the amidation of (S)-4-benzyl-2-oxazolidinone and crotonyl chloride, both of which are readily available in the global chemical market. This strategic choice eliminates the dependency on the problematic propargyl glycine derivative entirely. The core carbon skeleton is constructed through a highly stereoselective aldol condensation with 2,5-difluorobenzaldehyde, facilitated by Lewis acids such as dibutylboron trifluoromethanesulfonate or titanium tetrachloride. This step effectively installs the critical chiral centers with high fidelity using the Evans auxiliary strategy.
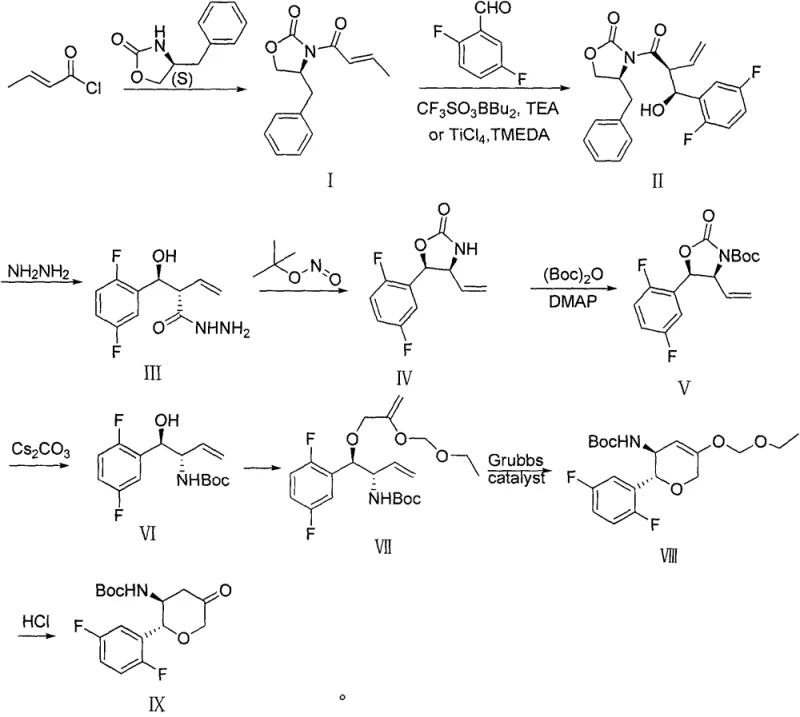
Subsequent transformations, including hydrazine condensation, rearrangement, and a pivotal Grubbs-catalyzed ring-closing metathesis (RCM), allow for the efficient construction of the six-membered pyran ring. By deferring the use of the precious metal catalyst (Grubbs) to a later stage where the molecular complexity is higher, the process maximizes the value added by the catalyst while minimizing waste. This holistic redesign of the synthetic tree results in a process that is not only chemically robust but also inherently more scalable, offering a clear pathway for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Stereoselective Aldol Condensation and RCM
The success of this novel synthesis hinges on two critical mechanistic events: the stereocontrolled aldol addition and the ring-closing metathesis. In the second step of the sequence, the enolate derived from the crotonyl-oxazolidinone intermediate reacts with 2,5-difluorobenzaldehyde. The presence of the chiral oxazolidinone auxiliary dictates the facial selectivity of the aldehyde approach, ensuring the formation of the desired syn-aldol product with high diastereomeric excess. Whether catalyzed by boron species at cryogenic temperatures (-70°C) or titanium complexes, this step establishes the absolute configuration at the C2 and C3 positions of the future pyran ring, which is vital for the biological activity of the final DPP-4 inhibitor. The rigorous control of temperature and stoichiometry in this phase is paramount to preventing epimerization and ensuring the purity profile meets stringent regulatory standards.
Following the establishment of the acyclic precursor, the synthesis proceeds through a series of functional group manipulations to install the necessary olefinic handles for cyclization. The eighth step utilizes a Grubbs catalyst to effect a ring-closing metathesis (RCM). This transformation is thermodynamically driven by the release of ethylene gas and kinetically controlled by the catalyst's ability to mediate the exchange of alkylidene units. The formation of the six-membered oxygen-containing heterocycle (the tetrahydropyran ring) via RCM is particularly advantageous because it tolerates a wide range of functional groups present in the intermediate, such as the protected amine and the fluorinated aromatic ring. This chemoselectivity minimizes side reactions and simplifies the isolation of the cyclic intermediate, directly contributing to the high overall yield reported in the patent examples.
Furthermore, the impurity profile of this route is managed through the stability of the intermediates. The Boc-protected amine and the oxazolidinone-derived intermediates exhibit sufficient thermal and chemical stability to withstand standard workup procedures, including aqueous washes and silica gel chromatography. This stability reduces the risk of degradation products that are often difficult to separate from the target molecule. By designing a route where each intermediate is isolable and stable, the process allows for rigorous quality control checkpoints, ensuring that any deviations in stereochemistry or purity can be corrected before proceeding to the next high-value step, thereby safeguarding the integrity of the final high-purity pharmaceutical intermediate.
How to Synthesize Omarigliptin Chiral Intermediate Efficiently
The execution of this synthesis requires precise adherence to the reaction conditions outlined in the patent to maximize yield and stereochemical integrity. The process begins with the activation of the oxazolidinone followed by the critical low-temperature aldol reaction, which sets the stereochemical tone for the entire sequence. Subsequent steps involve careful pH control during the hydrazine treatment and rearrangement phases to prevent racemization. The final cyclization and deprotection steps must be monitored closely to ensure complete conversion without over-reaction. For a detailed breakdown of the specific molar ratios, solvent choices, and temperature profiles required for each transformation, please refer to the standardized protocol below.
- Perform amidation of (S)-4-benzyl-2-oxazolidinone with crotonyl chloride to form the Evans auxiliary intermediate.
- Execute stereoselective aldol condensation with 2,5-difluorobenzaldehyde using boron or titanium Lewis acids.
- Conduct hydrazine condensation followed by rearrangement and Boc protection to establish the amine functionality.
- Complete the pyran ring formation via Grubbs-catalyzed ring-closing metathesis and final hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers transformative benefits that extend beyond simple chemistry. The primary driver for cost optimization lies in the substitution of expensive, custom-synthesized starting materials with commodity chemicals. By eliminating the need for Boc-L-propargyl glycine, manufacturers can bypass a significant portion of the upstream supply chain that is prone to bottlenecks and price surges. This shift fundamentally alters the cost structure of the intermediate, allowing for substantial cost savings in raw material acquisition. Furthermore, the use of standard reagents like triethylamine, dichloromethane, and cesium carbonate ensures that sourcing is not limited to niche suppliers, thereby enhancing supply security and negotiating leverage.
Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes directly to supply continuity. Unlike processes that require exotic catalysts or ultra-low temperatures throughout the entire sequence, this route confines sensitive operations to specific, manageable steps. The intermediates generated are stable solids or oils that can be stored and transported if necessary, providing flexibility in production scheduling. This stability reduces the risk of batch failures due to minor deviations in processing parameters, a common issue in complex chiral syntheses. Consequently, manufacturers can offer more reliable delivery schedules to their downstream API partners, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of stockouts in the global diabetes medication market.
Scalability and Environmental Compliance: The scalability of this process is evidenced by the use of unit operations that are standard in the fine chemical industry, such as liquid-liquid extraction, crystallization, and column chromatography. The avoidance of heavy metal contaminants in the early stages simplifies the purification burden, as there is less need for aggressive scavenging resins or complex filtration systems to meet residual metal specifications. Additionally, the atom economy of the ring-closing metathesis step is favorable, generating ethylene as the primary byproduct, which is easily vented. This cleaner reaction profile aligns with modern environmental, health, and safety (EHS) standards, reducing the burden of waste disposal and facilitating regulatory approval for commercial manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Omarigliptin intermediates using this patented methodology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical implementation of the technology.
Q: Why is the conventional synthesis of Omarigliptin intermediates considered costly?
A: Conventional routes often rely on Boc-L-propargyl glycine, a starting material that is difficult to source commercially and requires complex multi-step preparation, significantly driving up raw material costs.
Q: What are the primary advantages of the novel synthesis route described in CN107459501B?
A: The novel route utilizes readily available starting materials like crotonyl chloride and avoids early-stage noble metal catalysis, resulting in higher yields, simplified purification, and better suitability for industrial scale-up.
Q: How does this method ensure high stereochemical purity?
A: The process employs an Evans oxazolidinone auxiliary during the critical aldol condensation step, which provides excellent stereocontrol, ensuring the correct configuration required for the biological activity of the final DPP-4 inhibitor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Omarigliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensively evaluated the synthetic route described in CN107459501B and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel chemistry are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with rigorous QC labs capable of monitoring chiral purity and impurity profiles at every stage, guaranteeing that our stringent purity specifications are consistently met for every batch delivered.
We invite global pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains for DPP-4 inhibitors. By leveraging our process engineering capabilities, we can help you achieve significant efficiency gains and cost reductions. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can support your long-term strategic goals in the diabetes care sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
