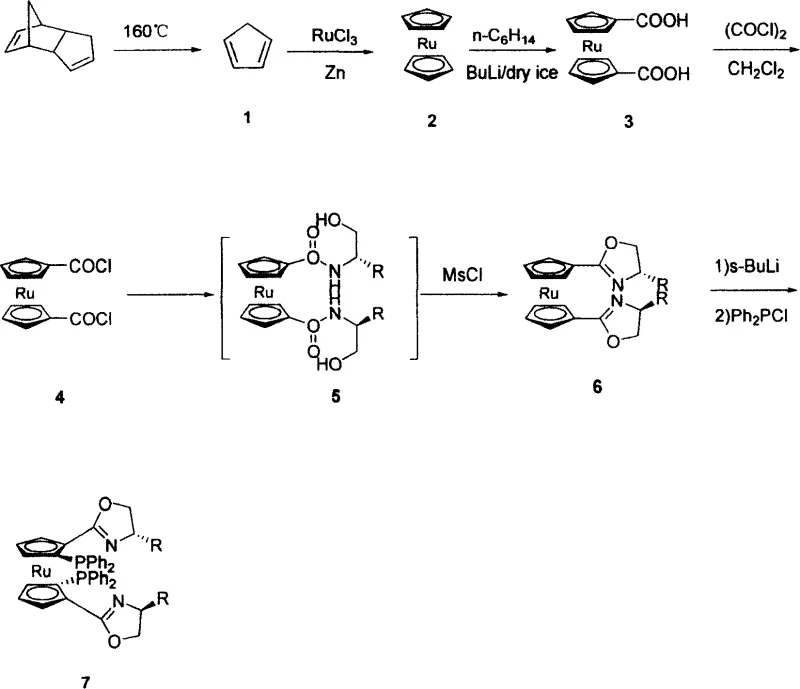
Advanced C2-Symmetric Ruthenocene Ligand Manufacturing for High-Performance Asymmetric Catalysis Applications
The landscape of asymmetric catalysis has been profoundly transformed by the development of specialized chiral ligands, as evidenced by the innovations detailed in patent CN100381451C. This specific intellectual property introduces a robust methodology for synthesizing C2-symmetric chiral ruthenocene ligands, which represent a significant leap forward in organometallic chemistry. Unlike traditional ferrocene-based systems, these ruthenium-containing structures offer enhanced thermal stability and distinct electronic environments that are critical for achieving high enantioselectivity in complex synthetic transformations. The patent outlines a comprehensive six-step synthetic route that begins with readily available dicyclopentadiene and culminates in a sophisticated bisoxazoline diphenylphosphino ruthenocene derivative. For R&D directors and technical decision-makers, understanding the nuances of this synthesis is vital, as it provides a reliable pathway to access high-value chiral catalysts that are essential for the production of advanced pharmaceutical intermediates and fine chemicals. The structural integrity and stereochemical purity of these ligands directly correlate with the efficiency of downstream catalytic processes, making this technology a cornerstone for modern asymmetric synthesis strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral metallocene ligands has been plagued by inefficiencies that hinder commercial viability and scalability. Conventional methods often rely on multi-step sequences that require the isolation and purification of unstable intermediates, leading to significant material loss and increased operational costs. Many traditional routes utilize iron-based ferrocene scaffolds which, while useful, sometimes lack the necessary electronic tunability required for challenging asymmetric transformations such as intramolecular cyclizations or specific carbon-carbon bond formations. Furthermore, older synthetic protocols frequently employ harsh reaction conditions or expensive transition metal catalysts that are difficult to remove from the final product, posing risks for downstream applications in sensitive industries like pharmaceuticals. The reliance on stoichiometric amounts of chiral auxiliaries in some legacy methods also drives up the cost of goods sold, making the final catalyst prohibitively expensive for large-scale manufacturing. These limitations create bottlenecks in the supply chain, where the inability to produce high-purity ligands consistently can delay critical drug development timelines and compromise the quality of the final active pharmaceutical ingredients.
The Novel Approach
The methodology presented in the patent data offers a transformative solution by streamlining the synthesis into a more efficient and controllable process. A key innovation is the implementation of a one-pot reaction strategy in the later stages of the synthesis, where the amide intermediate is directly converted to the bisoxazoline structure without the need for intermediate isolation. This approach not only simplifies the operational workflow but also drastically reduces the consumption of solvents and reagents, aligning with green chemistry principles. The use of ruthenocene as the core scaffold provides a more robust platform compared to ferrocene, offering better stability under the rigorous conditions often required for catalytic cycles. The synthetic route leverages readily available starting materials like dicyclopentadiene and ruthenium trichloride, ensuring that the raw material supply chain is secure and cost-effective. By optimizing reaction conditions, such as the precise temperature control during lithiation and the use of specific solvents like dichloromethane and n-hexane, the process achieves high yields and purity levels that are essential for commercial adoption. This novel approach effectively addresses the pain points of traditional synthesis, offering a scalable and economically viable pathway for producing high-performance chiral ligands.
Mechanistic Insights into C2-Symmetric Ruthenocene Ligand Formation
The core value of this technology lies in the precise construction of the C2-symmetric architecture, which is fundamental to its ability to induce high stereoselectivity in catalytic reactions. The synthesis begins with the depolymerization of dicyclopentadiene to generate cyclopentadiene, which is then metallated with ruthenium trichloride to form the ruthenocene core. This metallocene structure is subsequently functionalized through a lithiation-carboxylation sequence to introduce carboxylic acid groups at the 1,1' positions, setting the stage for the introduction of chirality. The conversion of these carboxylic acids to acid chlorides using oxalyl chloride is a critical activation step that enables the subsequent coupling with chiral beta-amino alcohols. The formation of the oxazoline rings is particularly noteworthy, as it locks the chiral information into the ligand framework, creating a rigid environment that effectively differentiates between enantiotopic faces of a substrate during catalysis. The final phosphination step introduces the diphenylphosphino groups, which serve as the coordinating sites for the active metal center in the catalytic cycle, completing the P,N-ligand system that is renowned for its versatility.
Impurity control is a paramount concern in the production of such high-value ligands, and the patent details specific measures to ensure product integrity. The use of dry ice and n-hexane mixtures during the carboxylation step helps to manage the exothermic nature of the reaction and prevents the formation of side products that could arise from over-lithiation. The one-pot cyclization step minimizes the exposure of reactive intermediates to air and moisture, which are common sources of degradation and impurity generation in organometallic chemistry. Purification strategies, such as column chromatography using specific eluent systems like ethyl acetate and petroleum ether, are employed to remove unreacted starting materials and by-products, ensuring that the final ligand meets the stringent purity specifications required for asymmetric catalysis. The structural characterization data, including NMR spectra, confirms the successful formation of the C2-symmetric structure and the absence of diastereomeric impurities that could compromise catalytic performance. This rigorous attention to mechanistic detail and purity control underscores the reliability of the process for producing consistent, high-quality ligands.
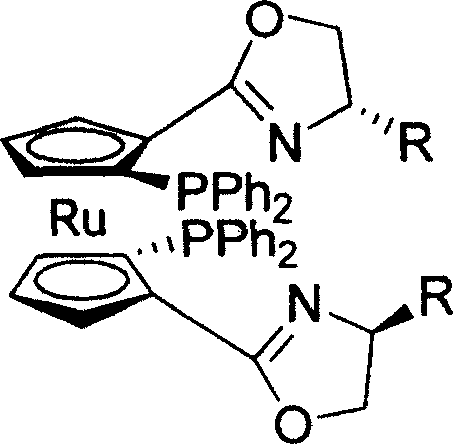
How to Synthesize C2-Symmetric Ruthenocene Ligand Efficiently
Implementing this synthesis route requires a deep understanding of organometallic handling and precise process control to ensure safety and reproducibility. The detailed standardized synthesis steps involve careful management of air-sensitive reagents like sec-butyllithium and strict temperature regulation during the lithiation phases to prevent decomposition. Operators must be trained in the handling of volatile solvents and the execution of distillation processes for the initial preparation of cyclopentadiene. The one-pot cyclization step demands precise stoichiometric addition of reagents to drive the reaction to completion without generating excessive waste. While the patent provides a robust framework, scaling this process requires optimization of mixing parameters and heat transfer capabilities to maintain the reaction profile observed in the laboratory examples. For a comprehensive guide on the specific operational parameters and safety protocols, please refer to the standardized synthesis steps provided in the technical documentation below.
- Preparation of 1,1'-dicarboxyruthenocene precursor via lithiation and carboxylation of ruthenocene.
- One-pot cyclization with beta-amino alcohol and methanesulfonyl chloride to form 1,1'-bisoxazoline ruthenocene.
- Final lithiation at -78°C followed by reaction with diphenylphosphine chloride to yield the C2-symmetric ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthesis route offers compelling advantages that translate directly into cost savings and operational efficiency. The reliance on commodity chemicals such as dicyclopentadiene and ruthenium trichloride ensures that the raw material base is broad and less susceptible to supply shocks compared to routes requiring exotic or proprietary starting materials. The elimination of intermediate isolation steps in the one-pot cyclization process significantly reduces the volume of solvents required and the energy consumed for drying and purification, leading to a lower environmental footprint and reduced waste disposal costs. This streamlined process flow also shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations. The high yields reported in the patent examples suggest that the process is materially efficient, minimizing the loss of expensive ruthenium metal and chiral auxiliaries. These factors combine to create a supply chain that is not only cost-effective but also resilient and capable of supporting the rigorous quality standards demanded by the pharmaceutical and fine chemical industries.
- Cost Reduction in Manufacturing: The integration of a one-pot synthesis step for the formation of the bisoxazoline ring eliminates the need for intermediate workup and purification, which significantly reduces labor costs and solvent consumption. By avoiding the isolation of the amide intermediate, the process minimizes material handling losses and reduces the time required for production, leading to substantial cost savings in the overall manufacturing budget. The use of standard solvents and reagents further ensures that procurement costs remain stable and predictable, avoiding the premiums associated with specialized or hazardous chemicals. This efficiency allows for a more competitive pricing structure for the final ligand, making it an attractive option for cost-sensitive applications in drug discovery and process development.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including dicyclopentadiene and ruthenium salts, are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the synthetic route, with its tolerance for standard laboratory and plant equipment, ensures that production can be easily transferred between facilities without significant requalification efforts. This flexibility enhances supply continuity, ensuring that customers can rely on consistent delivery schedules even in the face of regional disruptions. The scalability of the process from gram to kilogram scales demonstrates its readiness for commercial production, providing assurance that supply volumes can be ramped up to meet increasing demand without compromising quality or lead times.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing waste generation through the one-pot strategy and the use of recyclable solvents. The reduction in solvent usage and the elimination of intermediate isolation steps lower the volume of hazardous waste that requires treatment and disposal, simplifying compliance with environmental regulations. The high atom economy of the reaction sequence ensures that a greater proportion of the raw materials are incorporated into the final product, reducing the overall environmental impact of the manufacturing process. This alignment with sustainability goals makes the ligand an attractive choice for companies looking to reduce their carbon footprint and meet corporate social responsibility targets while maintaining high production standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this C2-symmetric ruthenocene ligand. These answers are derived from the technical specifications and beneficial effects outlined in the patent data, providing clarity on the ligand's performance and manufacturing feasibility. Understanding these aspects is crucial for stakeholders evaluating the integration of this technology into their existing synthetic workflows. The responses highlight the ligand's versatility, the robustness of the synthesis, and the strategic advantages it offers for asymmetric catalysis applications.
Q: What are the advantages of ruthenocene ligands over traditional ferrocene ligands?
A: Ruthenocene ligands offer superior thermal stability and unique electronic properties due to the ruthenium center, often resulting in higher enantioselectivity and reactivity in asymmetric catalytic cycles compared to iron-based analogues.
Q: Is the one-pot cyclization step suitable for large-scale manufacturing?
A: Yes, the one-pot conversion of amide intermediates to bisoxazoline structures eliminates isolation steps, significantly reducing solvent waste and processing time, which enhances scalability and cost-efficiency for industrial production.
Q: What types of asymmetric reactions can this ligand facilitate?
A: This C2-symmetric ligand is highly effective for carbon-carbon bond formation, asymmetric cyclopropanation, intramolecular Wacker-Type cyclization, and olefin asymmetric oxidation, providing excellent stereocontrol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ruthenocene Ligand Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at translating complex laboratory protocols, such as the C2-symmetric ruthenocene ligand synthesis, into robust industrial processes that maintain stringent purity specifications. We operate rigorous QC labs equipped with advanced analytical instrumentation to ensure that every batch of ligand meets the exacting standards required for high-performance asymmetric catalysis. Our commitment to quality and consistency makes us a trusted partner for pharmaceutical and fine chemical companies seeking reliable access to advanced chiral ligands. We understand the critical nature of supply chain continuity and are dedicated to providing solutions that support your long-term production goals.
We invite you to engage with our technical procurement team to discuss your specific requirements and explore how we can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our manufacturing capabilities can reduce your overall cost of goods. We encourage you to contact us to obtain specific COA data for our available ligand inventory and to request route feasibility assessments for your custom projects. Our team is ready to provide the technical support and commercial flexibility needed to accelerate your development timelines and ensure the success of your catalytic processes.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
