Advanced Hypervalent Iodine Reagents for Efficient Gem-Difluorovinylation in Commercial Synthesis
Advanced Hypervalent Iodine Reagents for Efficient Gem-Difluorovinylation in Commercial Synthesis
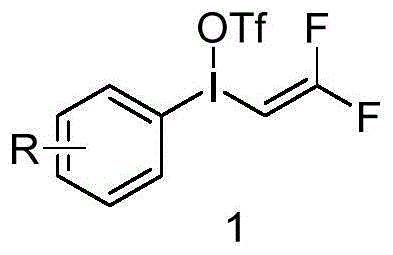
The landscape of organofluorine chemistry is undergoing a significant transformation with the introduction of novel electrophilic transfer reagents, as detailed in patent CN113402361A. This groundbreaking intellectual property discloses a new class of hypervalent iodine compounds, specifically (2,2-difluorovinyl)(phenyl)-λ3-iodane trifluoromethanesulfonate, which serves as a powerful tool for the direct introduction of gem-difluorovinyl groups into organic molecules. The gem-difluorovinyl moiety is a critical structural motif found in numerous biologically active compounds, acting as a metabolic stable isostere for carbonyl groups and enhancing membrane permeability in drug candidates. For R&D directors and process chemists, the ability to install this functionality directly, rather than through tedious multi-step sequences, represents a paradigm shift in synthetic efficiency. The patent highlights that this reagent is not only synthetically accessible but also exhibits remarkable stability and reactivity, making it an ideal candidate for the scalable production of high-value pharmaceutical and agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the incorporation of gem-difluorovinyl groups into complex molecular scaffolds has been a formidable challenge for synthetic chemists, often necessitating indirect and laborious pathways. Traditional strategies frequently rely on the synthesis of specific precursor compounds, such as carbonyl derivatives, which must then undergo subsequent conversion reactions using reagents like difluoromethylphosphine ylides. Alternatively, methods involving difluorocarbene precursors generated from diazo compounds pose significant safety hazards due to the explosive nature of diazo species and the requirement for stringent handling protocols. Furthermore, approaches based on C-F bond elimination from alpha-trifluoromethyl-substituted olefins often suffer from poor atom economy and require harsh reaction conditions that can degrade sensitive functional groups. These conventional routes typically involve multiple isolation and purification steps, leading to accumulated yield losses and substantial increases in manufacturing costs. For procurement managers, the reliance on specialized, often hazardous reagents translates to higher supply chain risks and increased expenditure on safety compliance and waste disposal.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN113402361A offers a streamlined, direct approach utilizing a pre-formed hypervalent iodine transfer reagent. This novel strategy bypasses the need for in situ generation of reactive intermediates or the preparation of unstable precursors. The core innovation lies in the unique reactivity of the trivalent iodine center, which facilitates a clean ligand exchange mechanism to transfer the gem-difluorovinyl group to a wide array of nucleophiles. As illustrated in the reaction scheme below, the synthesis of the reagent itself is straightforward, involving the reaction of a cyano-iodonium salt with a gem-difluorovinyl stannane. This direct transfer capability allows for the functionalization of carboxylic acids and amides under remarkably mild conditions, typically at room temperature, without the need for external heating or cryogenic cooling beyond the initial reagent preparation. This simplification of the synthetic route drastically reduces the operational complexity, making it highly attractive for commercial scale-up where robustness and reproducibility are paramount.

Mechanistic Insights into Hypervalent Iodine-Mediated Transfer
The efficacy of this gem-difluorovinyl transfer reagent is rooted in the distinct electronic properties of the λ3-iodane center, which acts as a potent electrophile. In the context of the synthesis described in the patent, the mechanism proceeds through a ligand exchange process where the cyanide ligand on the iodine(III) species is displaced by the gem-difluorovinyl group from the tributyltin precursor. This exchange is driven by the formation of a strong tin-cyanide bond and the stabilization of the resulting hypervalent iodine species by the triflate counterion. Once formed, the reagent functions as an electrophilic source of the difluorovinyl cation equivalent. When reacted with nucleophiles such as carboxylates or amides, often in the presence of a mild base or silver salt promoter, the nucleophile attacks the iodine center, followed by reductive elimination or ligand coupling that delivers the gem-difluorovinyl group to the substrate. This mechanistic pathway avoids the generation of free radical species or highly reactive carbenes that could lead to non-selective side reactions, thereby ensuring high chemoselectivity. For process development teams, understanding this mechanism is crucial for optimizing reaction parameters, such as solvent choice and stoichiometry, to maximize yield and minimize the formation of iodobenzene byproducts.
Furthermore, the structural integrity of the reagent contributes significantly to impurity control and product purity, a key concern for regulatory compliance in pharmaceutical manufacturing. The patent specifies that the reagent can be isolated as a stable solid, which allows for rigorous quality control testing prior to use, unlike transient intermediates generated in situ. The use of dichloromethane as the preferred solvent ensures good solubility for both the hypervalent iodine precursor and the organotin reagent, facilitating homogeneous reaction kinetics. The mild reaction temperatures, ranging from -40°C during preparation to room temperature during the transfer step, prevent thermal decomposition of the sensitive fluorinated moiety. This thermal stability is essential for maintaining the fidelity of the gem-difluorovinyl group, preventing isomerization or defluorination which are common pitfalls in fluorine chemistry. By controlling the reaction environment through inert atmosphere protection (nitrogen) and precise temperature management, manufacturers can achieve consistent batch-to-batch reproducibility, a critical metric for supply chain reliability.
How to Synthesize (2,2-Difluorovinyl)(phenyl)-λ3-iodane Triflate Efficiently
The preparation of this high-value transfer reagent is designed to be operationally simple, leveraging standard laboratory equipment and readily available starting materials. The process begins with the dissolution of the cyano-iodonium precursor in anhydrous dichloromethane, followed by cooling to low temperatures to manage the exothermicity of the subsequent ligand exchange. The addition of the gem-difluorovinyl tributyltin reagent is performed dropwise to maintain control over the reaction rate, after which the mixture is allowed to warm to ambient temperature to drive the conversion to completion. Following the reaction, the workup procedure involves concentration under reduced pressure and precipitation using diethyl ether, a technique that effectively isolates the ionic hypervalent iodine product from neutral organic byproducts. The detailed standardized synthesis steps, including specific molar ratios and purification techniques, are outlined in the guide below to ensure successful replication of the patent results.
- Dissolve cyano(phenyl)-λ3-iodonium trifluoromethanesulfonate in dichloromethane and cool to -40°C under nitrogen protection.
- Add a solution of gem-difluorovinyl tributyltin in dichloromethane dropwise, then slowly warm to room temperature and stir for 1-2 hours.
- Concentrate the reaction mixture, add diethyl ether to precipitate the solid product, wash with ether, and filter to obtain the pure reagent.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this hypervalent iodine technology offers substantial strategic benefits for procurement and supply chain operations, primarily driven by the simplification of the manufacturing process. The elimination of transition metal catalysts, which are often required in traditional cross-coupling approaches, removes the need for expensive and time-consuming metal scavenging steps. This reduction in downstream processing directly correlates to lower production costs and shorter cycle times, allowing for faster time-to-market for new drug candidates. Additionally, the reagent's stability and ease of storage mitigate the risks associated with the supply of hazardous or unstable intermediates, ensuring a more resilient supply chain. The ability to source raw materials that are commercially available or easily synthesized further enhances the economic viability of this route, making it a sustainable choice for long-term production campaigns.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant reduction in unit operations. By replacing multi-step precursor syntheses with a direct transfer reaction, manufacturers save on solvent usage, energy consumption for heating and cooling, and labor hours associated with multiple isolations. The absence of heavy metal catalysts also eliminates the cost of specialized filtration media and the regulatory burden of proving residual metal compliance in the final API. Furthermore, the high yields reported in the patent examples suggest efficient atom utilization, minimizing the waste of expensive fluorinated starting materials. These factors combine to create a leaner, more cost-effective manufacturing process that improves the overall margin profile of the final fluorinated intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the robustness of the reagent and the simplicity of its synthesis. Unlike methods relying on diazo compounds or cryogenic conditions that require specialized infrastructure, this process can be executed in standard glass-lined or stainless steel reactors found in most fine chemical facilities. The raw materials, such as the cyano-iodonium salts and organotin species, are part of established supply chains for hypervalent iodine chemistry, reducing the risk of raw material shortages. Moreover, the stability of the isolated reagent allows for inventory buffering, enabling manufacturers to produce the transfer reagent in large batches and store it for just-in-time use in downstream functionalization reactions, thereby decoupling the supply of the reagent from the immediate production schedule of the final product.
- Scalability and Environmental Compliance: Scaling this technology from gram to kilogram or tonne scale is facilitated by the mild reaction conditions and the absence of gas evolution or highly exothermic events that are difficult to manage in large vessels. The use of dichloromethane, while requiring proper containment, is a well-understood solvent in the industry with established recovery and recycling protocols. The environmental footprint is further reduced by the avoidance of toxic heavy metals and the generation of less hazardous waste streams compared to traditional fluorination methods. This alignment with green chemistry principles not only simplifies waste disposal but also supports corporate sustainability goals, making the process more attractive to environmentally conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gem-difluorovinyl transfer technology. These insights are derived directly from the experimental data and claims presented in patent CN113402361A, providing a factual basis for evaluating its suitability for your specific project needs. Understanding the scope and limitations of the reagent is essential for effective process design and risk assessment.
Q: What are the primary advantages of this hypervalent iodine reagent over traditional methods?
A: Unlike traditional methods requiring multi-step precursor synthesis or dangerous diazo compounds, this reagent allows for direct, one-step gem-difluorovinylation under mild conditions with high yields.
Q: Is this reagent compatible with sensitive functional groups?
A: Yes, the reagent operates under mild conditions (room temperature) and is compatible with various nucleophiles like carboxylic acids and amides, preserving sensitive functionalities.
Q: How does this technology impact production costs?
A: By eliminating the need for transition metal catalysts and complex multi-step sequences, the process significantly reduces purification costs and raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gem Difluoro Vinyl Transfer Reagent Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced hypervalent iodine chemistry in accelerating the development of next-generation fluorinated pharmaceuticals. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of the transfer reagent itself or the downstream gem-difluorovinylated products, our facility is optimized to handle complex fluorination chemistry with the highest standards of safety and quality.
We invite you to collaborate with us to leverage this innovative technology for your pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to discuss your project specifics,索取 specific COA data for our available fluorinated building blocks, and obtain comprehensive route feasibility assessments that demonstrate how we can optimize your supply chain for maximum efficiency and cost-effectiveness.
