Advanced Synthetic Route for Xiawu Base B: Enhancing Purity and Commercial Scalability
Advanced Synthetic Route for Xiawu Base B: Enhancing Purity and Commercial Scalability
The pharmaceutical industry continuously seeks robust and efficient synthetic pathways for bioactive natural products, particularly alkaloids derived from traditional medicinal plants. Patent CN108191878B discloses a novel and highly effective method for the preparation of Xiawu Base B, a significant alkaloid component found in Corydalis decumbens. This compound is renowned for its therapeutic potential in treating hypertension, apoplexy, and rheumatism, acting as a potent agent for lowering blood pressure and relieving pain. The disclosed technology represents a substantial leap forward in medicinal chemistry, offering a streamlined approach that overcomes the limitations of previous extraction or synthesis methods which were often plagued by low yields and complex purification requirements. By leveraging modern catalytic strategies, this invention provides a reliable foundation for the consistent supply of high-purity Xiawu Base B.
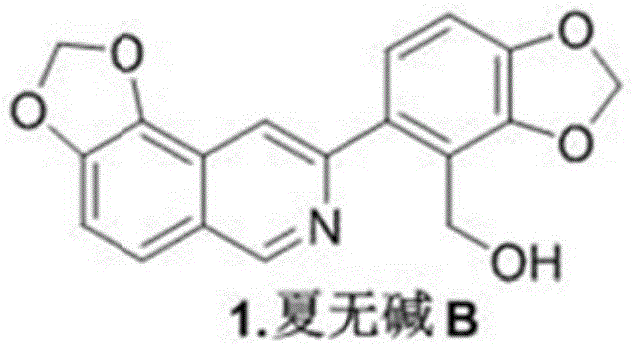
The strategic design of this synthesis begins with the selection of inexpensive and commercially abundant starting materials, specifically isovanillin and 4-bromopiperonyl derivatives. This choice is critical for procurement managers aiming to stabilize supply chains against volatile raw material markets. The route is meticulously engineered to maximize atom economy and minimize waste generation, aligning with modern green chemistry principles. Furthermore, the process avoids the use of hazardous or prohibitively expensive reagents in the key bond-forming steps, instead utilizing standard transition metal catalysts that are well-understood in industrial settings. This ensures that the transition from laboratory bench to kilogram-scale production is seamless, addressing the critical need for a reliable pharmaceutical intermediate supplier capable of meeting global demand without compromising on quality or delivery timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Xiawu Base B has been challenging, with few reported methods that are suitable for large-scale application. Traditional approaches often relied on direct extraction from plant sources, which is inherently limited by seasonal availability, geographical constraints, and the low concentration of the target alkaloid within the biomass. Such biological sourcing leads to significant batch-to-batch variability in purity and impurity profiles, posing severe risks for regulatory compliance in drug manufacturing. Moreover, synthetic routes described in prior art frequently suffered from harsh reaction conditions, requiring extreme temperatures or pressures that degrade sensitive functional groups. These legacy methods often involved multiple protection and deprotection steps that drastically reduced overall yield and increased the consumption of solvents and reagents, thereby inflating the cost of goods sold (COGS) and creating substantial environmental burdens through waste disposal.
The Novel Approach
In stark contrast, the methodology outlined in patent CN108191878B introduces a convergent synthetic strategy that dramatically simplifies the construction of the complex isoquinoline skeleton. The novel approach utilizes a modular assembly where two key fragments are synthesized independently and then coupled via a palladium-catalyzed carbon-carbon bond formation. This convergence allows for better control over the quality of intermediates before the final assembly, ensuring a cleaner reaction profile. A standout feature of this innovation is the final functionalization step, which employs a ruthenium-catalyzed C-H activation to install the crucial hydroxymethyl group directly. This eliminates the need for pre-functionalized starting materials for this specific position, reducing the step count and avoiding the generation of stoichiometric metallic waste associated with traditional organometallic additions. The result is a process that is not only chemically elegant but also economically superior.
Mechanistic Insights into Ru-Catalyzed C-H Activation and Cyclization
The core of this synthetic breakthrough lies in the sophisticated application of transition metal catalysis to construct the heterocyclic core and install peripheral functionalities with high precision. The formation of the isoquinoline ring (Compound 2) from the alkyne-aldehyde precursor (Compound 3) is achieved through a silver-mediated cyclization with ammonium acetate. This transformation likely proceeds via an imine intermediate followed by electrocyclic ring closure, a mechanism that is highly sensitive to reaction conditions. The patent specifies the use of silver nitrate and ammonium acetate in t-butanol at room temperature, conditions that are remarkably mild compared to traditional acid-catalyzed cyclizations which often require refluxing strong acids. This mildness preserves the integrity of the methylenedioxy groups present on the aromatic rings, preventing acid-catalyzed hydrolysis which is a common side reaction that leads to difficult-to-remove impurities.
Furthermore, the final step involving the conversion of Compound 2 to Xiawu Base B (Compound 1) showcases the power of modern C-H functionalization. The reaction utilizes ruthenium trichloride as a catalyst in conjunction with dimethylzinc and paraformaldehyde. Mechanistically, the ruthenium center activates the specific C-H bond on the aromatic ring adjacent to the nitrogen, directing the insertion of the formaldehyde equivalent. The use of paraformaldehyde as a C1 source is particularly advantageous from a safety and handling perspective compared to gaseous formaldehyde. The presence of zinc bromide acts as a Lewis acid promoter, facilitating the activation of the formaldehyde species. This catalytic cycle avoids the use of stoichiometric amounts of toxic heavy metals or cryogenic conditions typically required for lithiation-based functionalization, thereby enhancing the operational safety and environmental profile of the manufacturing process.
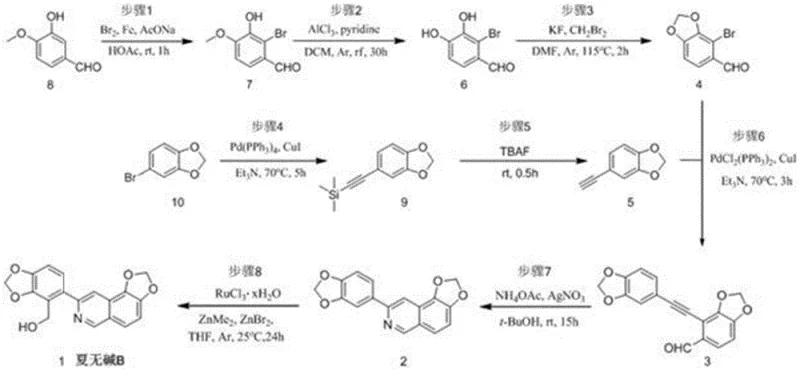
How to Synthesize Xiawu Base B Efficiently
The synthesis of Xiawu Base B is executed through a logical eight-step sequence that balances chemical efficiency with operational simplicity. The process initiates with the derivatization of isovanillin, proceeding through bromination, demethylation, and methylenation to generate the key aryl bromide intermediate. Parallel to this, the alkyne fragment is prepared from 4-bromopiperonyl ring via a Sonogashira coupling with trimethylsilylacetylene followed by deprotection. These two fragments are then united through a second Sonogashira coupling to form the extended conjugated system. The subsequent cyclization and final C-H activation steps complete the molecular architecture. For detailed operational parameters, including specific solvent volumes, stirring rates, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process parameters defined in the patent examples.
- Prepare Compound 4 via bromination of isovanillin, demethylation, and methylenation.
- Synthesize terminal alkyne Compound 5 from 4-bromopiperonyl ring via silylation and deprotection.
- Couple Compounds 4 and 5, cyclize to isoquinoline, and perform Ru-catalyzed hydroxymethylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the strategic selection of starting materials; isovanillin and 4-bromopiperonyl derivatives are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This diversification of the supply base ensures continuity of supply even during market fluctuations or geopolitical disruptions. Additionally, the process operates under relatively mild thermal conditions, with most reactions occurring between room temperature and 115°C. This reduces the energy load on manufacturing facilities, leading to lower utility costs and a smaller carbon footprint, which is increasingly important for meeting corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive and specialized reagents in favor of cost-effective alternatives. For instance, the use of paraformaldehyde as a C1 building block is significantly cheaper than using protected hydroxymethyl equivalents or organolithium reagents. Furthermore, the catalytic nature of the key coupling and activation steps means that expensive metals like palladium and ruthenium are used in minimal quantities and can potentially be recovered and recycled. The high selectivity of the reactions minimizes the formation of byproducts, which reduces the burden on downstream purification processes such as chromatography or recrystallization, thereby lowering solvent consumption and labor costs associated with isolation.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route contributes directly to supply chain resilience. The reactions described are tolerant to minor variations in reagent quality and do not require stringent exclusion of moisture or oxygen beyond standard inert atmosphere techniques (e.g., argon blanket). This tolerance simplifies the engineering controls required in the production plant, reducing the likelihood of batch failures due to equipment malfunction or operator error. The ability to produce the key intermediates (Compound 4 and Compound 5) in a modular fashion allows for inventory buffering; these stable intermediates can be stockpiled and then rapidly converted to the final API intermediate upon demand, drastically reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the use of common organic solvents such as tetrahydrofuran, dichloromethane, and ethanol, which are easily handled in standard stainless steel reactors. The avoidance of cryogenic conditions or high-pressure hydrogenation simplifies the reactor design requirements. From an environmental perspective, the atom economy of the coupling reactions is high, and the waste streams are primarily composed of benign salts and recoverable solvents. This simplifies wastewater treatment and ensures compliance with increasingly strict environmental regulations regarding heavy metal discharge and volatile organic compound (VOC) emissions, making it an ideal candidate for green manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Xiawu Base B using this patented methodology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for R&D and procurement teams evaluating this technology for integration into their supply chains.
Q: What are the key advantages of this new synthesis method for Xiawu Base B?
A: The method utilizes readily available starting materials like isovanillin and employs mild reaction conditions, significantly reducing production costs and improving suitability for industrial scale-up compared to prior art.
Q: How is the hydroxymethyl group introduced in the final step?
A: The final step involves a ruthenium-catalyzed C-H activation reaction using paraformaldehyde as a safe and low-cost hydrocarbon activation attacking reagent to introduce the hydroxymethyl moiety.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process avoids extreme conditions and uses common solvents and catalysts, making it highly scalable for commercial production of high-purity pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Xiawu Base B Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for complex pharmaceutical intermediates. Our team of expert chemists has thoroughly analyzed the synthetic route disclosed in CN108191878B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art facilities capable of handling the specific catalytic requirements of this process, including dedicated lines for palladium and ruthenium chemistry with rigorous QC labs to ensure stringent purity specifications. Our commitment to quality assurance means that every batch of Xiawu Base B produced meets the highest international standards, providing you with the confidence needed to advance your drug development programs.
We invite you to collaborate with us to leverage this advanced technology for your specific applications. Whether you require custom synthesis services or bulk supply of this key alkaloid intermediate, our technical procurement team is ready to assist. Please contact us today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
