Advanced Chiral Pyridoxal Catalysts for Scalable Asymmetric Synthesis of Beta-Amino Alcohols
Introduction to Next-Generation Organocatalysis
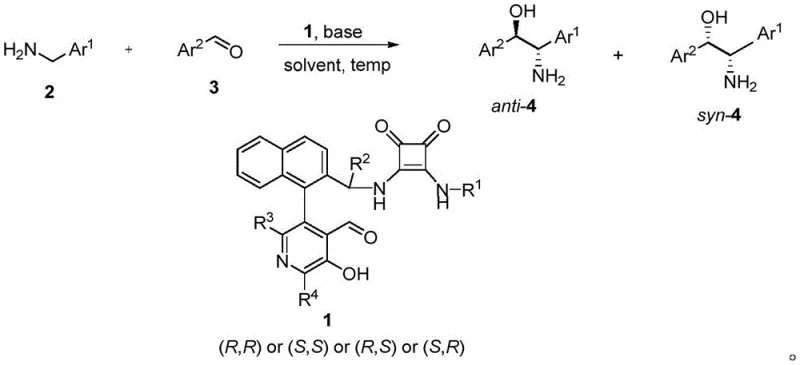
The landscape of asymmetric synthesis is undergoing a significant transformation, driven by the urgent need for sustainable and efficient manufacturing processes in the pharmaceutical and fine chemical sectors. Patent CN115043775A introduces a groundbreaking class of chiral pyridoxal catalysts that mimic biological enzymatic processes to achieve high-precision stereocontrol. Unlike traditional methods that rely heavily on transition metals, this technology leverages the inherent chirality of Vitamin B6 derivatives to catalyze the biomimetic aldol reaction between aryl methylamines and aryl aldehydes. This innovation provides a robust pathway for synthesizing chiral beta-amino alcohols, which are critical building blocks for numerous active pharmaceutical ingredients (APIs). By integrating this technology, manufacturers can access a reliable pharmaceutical intermediates supplier network that prioritizes both purity and environmental compliance.
The core breakthrough lies in the structural design of the catalyst, which features a unique biaryl architecture attached to the pyridoxal scaffold. This specific configuration creates a highly defined chiral environment that effectively discriminates between prochiral faces during the bond-forming event. The result is a reaction system that operates under remarkably mild conditions, often at temperatures as low as -5°C, yet delivers exceptional diastereoselectivity and enantioselectivity. For R&D teams focused on process development, this represents a paradigm shift away from harsh reagents towards biomimetic precision, enabling the creation of complex molecular architectures with reduced impurity profiles and simplified downstream processing requirements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino alcohols has been dominated by stoichiometric chiral auxiliaries or transition metal-catalyzed reactions. These conventional approaches suffer from several inherent drawbacks that impact both cost and operational efficiency. Metal-catalyzed systems often require stringent exclusion of moisture and oxygen, necessitating specialized equipment and increasing capital expenditure. Furthermore, the presence of heavy metals in the final product poses significant regulatory hurdles, requiring extensive purification steps such as activated carbon treatment or scavenger resin filtration to meet ppm-level specifications. These additional unit operations not only extend the production cycle but also lead to substantial yield losses, thereby inflating the overall cost of goods sold.
Additionally, many traditional organocatalytic systems struggle with substrate scope limitations, particularly when dealing with inert primary amines like benzylamine. Previous generations of pyridoxal catalysts were often restricted to strongly activated amines containing adjacent electron-withdrawing groups, limiting their utility in diverse synthetic campaigns. The reliance on cryogenic conditions below -40°C in some legacy protocols further exacerbates energy consumption and operational complexity. For supply chain managers, these factors translate into longer lead times and higher vulnerability to raw material fluctuations, making the search for more robust and versatile catalytic platforms a strategic priority for modern chemical enterprises seeking cost reduction in fine chemical manufacturing.
The Novel Approach
The novel approach detailed in the patent data utilizes a specifically engineered chiral pyridoxal catalyst that overcomes these historical barriers through rational molecular design. By incorporating a bulky biaryl group at the 4-position of the pyridoxal ring, the catalyst achieves superior steric hindrance that directs the incoming nucleophile with high fidelity. This structural modification allows the reaction to proceed efficiently with simple aryl methylamines, expanding the accessible chemical space significantly. The reaction conditions are notably温和 (mild), typically operating between -5°C and -15°C in common solvent systems like Toluene/DCM mixtures, which are easily handled in standard glass-lined reactors without the need for extreme cryogenic infrastructure.
Moreover, this organocatalytic strategy eliminates the risk of metal contamination entirely, simplifying the purification workflow and ensuring a cleaner impurity profile from the outset. The catalytic loading is low, often around 5 mol%, which enhances the economic viability of the process by minimizing the amount of expensive chiral material required per batch. The versatility of this system is evidenced by its ability to tolerate various functional groups on both the amine and aldehyde components, including halogens, ethers, and heterocycles. This broad substrate tolerance makes it an ideal candidate for library synthesis and rapid analog generation in drug discovery, providing a high-purity chiral beta-amino alcohols source that aligns with the rigorous quality standards of the global pharmaceutical industry.
Mechanistic Insights into Biomimetic Aldol Catalysis
The mechanistic elegance of this pyridoxal-catalyzed transformation lies in its faithful imitation of enzymatic transamination and aldol processes found in nature. The catalytic cycle begins with the condensation of the chiral pyridoxal catalyst with the aryl methylamine substrate to form a chiral aldimine intermediate. This step is crucial as it transfers the chirality from the catalyst to the reactive species. Subsequently, the acidic alpha-proton of the aldimine is deprotonated by the added base, such as DBU or DBN, generating a resonance-stabilized carbanion or enamine-like nucleophile. The unique biaryl scaffold of the catalyst shields one face of this planar intermediate, forcing the subsequent nucleophilic attack on the aldehyde electrophile to occur from the less hindered trajectory.
This facial selectivity is the key determinant of the observed high enantiomeric excess (ee) and diastereomeric ratio (dr). Following the carbon-carbon bond formation, the resulting adduct undergoes hydrolysis to release the chiral beta-amino alcohol product and regenerate the free pyridoxal catalyst, completing the cycle. The stability of the carbanion intermediate is enhanced by the electron-withdrawing nature of the protonated pyridinium nitrogen, allowing the reaction to proceed under mild thermal conditions without decomposition. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as base strength and solvent polarity, to optimize kinetics and selectivity for specific substrate pairs, ensuring consistent quality in commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Chiral Pyridoxal Catalyst Efficiently
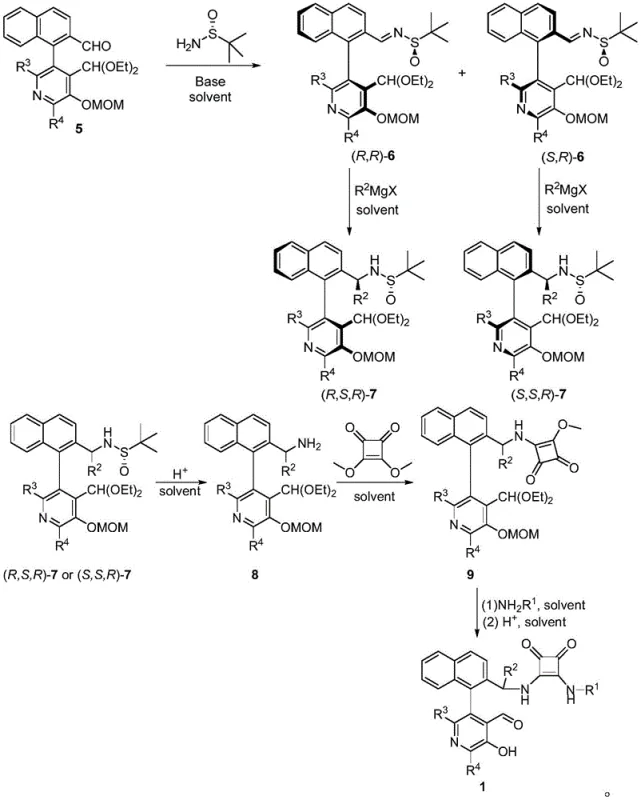
The preparation of the catalyst itself is a multi-step sequence that begins with commercially available starting materials, ensuring a secure and scalable supply chain. The initial step involves the condensation of a protected pyridoxal derivative with (R)-tert-butyl sulfinamide to introduce the chiral auxiliary. This is followed by a stereoselective Grignard addition to establish the benzylic stereocenter, which dictates the absolute configuration of the final catalyst. Subsequent acid-mediated deprotection removes the sulfinyl group, yielding the chiral amine intermediate. Finally, condensation with dimethyl squarate and subsequent amidation with a primary amine installs the squaramide moiety, which is essential for hydrogen-bonding activation in the catalytic cycle. The detailed standardized synthesis steps are outlined below to guide process implementation.
- Prepare the chiral pyridoxal catalyst by condensing the starting aldehyde with tert-butyl sulfinamide, followed by Grignard addition and hydrolysis to establish chirality.
- Mix the aryl methylamine and aryl aldehyde substrates with the chiral catalyst (5 mol%) and a base such as DBU or DBN in a solvent mixture like Toluene/DCM.
- Maintain the reaction at low temperatures (-5°C to -15°C) for 24-48 hours, then quench with hydroxylamine solution and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain directors, the adoption of this pyridoxal catalytic technology offers compelling strategic advantages beyond mere technical performance. The elimination of transition metals from the synthesis route fundamentally alters the cost structure of production. Without the need for expensive metal salts or ligands, and more importantly, without the requisite downstream metal scavenging steps, the overall processing time is significantly shortened. This streamlining of the manufacturing workflow leads to substantial cost savings by reducing solvent usage, waste disposal volumes, and labor hours associated with purification. The qualitative improvement in process efficiency directly contributes to a more competitive pricing model for the final intermediates.
Furthermore, the robustness of the reaction conditions enhances supply chain reliability. The ability to run reactions at moderately low temperatures (-5°C to -15°C) rather than deep cryogenic levels reduces energy consumption and minimizes the risk of batch failures due to temperature excursions. The starting materials, including the aryl amines and aldehydes, are commodity chemicals with stable global availability, mitigating the risk of raw material shortages. This stability ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands. The scalability of the process is further supported by the use of common organic solvents like toluene and dichloromethane, which are well-understood in large-scale industrial settings, facilitating a smooth transition from pilot plant to full commercial production.
- Cost Reduction in Manufacturing: The metal-free nature of this organocatalytic process removes the financial burden associated with precious metal catalysts and their removal. By avoiding heavy metal scavengers and the associated filtration and testing protocols, manufacturers can achieve a leaner production flow. This reduction in unit operations translates to lower operational expenditures and a smaller environmental footprint, aligning with green chemistry principles while improving the bottom line through efficient resource utilization.
- Enhanced Supply Chain Reliability: The reliance on abundant organic starting materials and standard solvents ensures a resilient supply chain不受 (unaffected by) the volatility often seen in the rare earth or precious metal markets. The mild reaction conditions reduce equipment stress and maintenance requirements, leading to higher asset availability and consistent output. This reliability is critical for maintaining continuous production schedules and meeting strict delivery commitments to downstream API manufacturers without interruption.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing reaction parameters that are easily replicated in large vessels. The absence of toxic metal waste simplifies effluent treatment and regulatory compliance, reducing the administrative and financial load associated with environmental permits. This ease of scale-up allows for rapid capacity expansion to meet growing demand, ensuring that the supply of critical chiral intermediates remains uninterrupted even during periods of market surge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral pyridoxal catalyst technology. These insights are derived directly from the patent specifications and experimental data, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the advantages of using pyridoxal catalysts over traditional metal catalysts?
A: Pyridoxal catalysts offer a metal-free alternative, eliminating the need for expensive heavy metal removal processes and reducing environmental toxicity while maintaining high enantioselectivity.
Q: What is the typical enantioselectivity achieved with this method?
A: The patented method demonstrates extremely high enantioselectivity, with ee values frequently exceeding 90% and often reaching up to 99% depending on the substrate.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild (low temperature, ambient pressure) and utilize readily available starting materials, making the process highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Pyridoxal Catalyst Supplier
As the pharmaceutical industry continues to demand higher purity and more sustainable synthesis routes, NINGBO INNO PHARMCHEM stands ready to support your development goals with our expertise in advanced organocatalysis. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing floor is seamless and efficient. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards, guaranteeing the consistency required for GMP environments.
We invite you to engage with our technical team to explore how this innovative pyridoxal catalyst technology can optimize your specific synthetic challenges. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your production volume. We encourage potential partners to contact our technical procurement team to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with confidence and precision.
