Optimizing Ibrutinib Precursor Production: A Technical Analysis of Patent CN111004239B
Optimizing Ibrutinib Precursor Production: A Technical Analysis of Patent CN111004239B
The pharmaceutical industry continuously seeks robust manufacturing routes for kinase inhibitors, particularly for blockbuster drugs like Ibrutinib. Patent CN111004239B, published in September 2022, introduces a significant technological advancement in the preparation of the key Ibrutinib precursor, (R)-3-(4-phenoxyphenyl)-1-(piperidine-3-yl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine (Compound I). This document details a novel synthetic strategy that overcomes the inherent limitations of conventional Mitsunobu couplings by employing a transient phosphine protection mechanism. For R&D directors and process chemists, this represents a shift from multi-step isolation protocols to a streamlined, one-pot methodology that promises enhanced purity profiles and operational simplicity. The core innovation lies in the pre-activation of the heterocyclic core to prevent side reactions, a critical bottleneck in previous industrial attempts.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Compound I has relied on direct Mitsunobu condensation between the pyrazolo-pyrimidine core (Compound III) and the chiral piperidine alcohol (Compound IV). However, industrial experience and prior art, such as US 7514444, have highlighted a persistent challenge: the poor regioselectivity of Compound III. The heterocyclic ring possesses multiple nucleophilic nitrogen atoms, leading to competitive side reactions where the alcohol couples at unintended positions or forms polymeric byproducts. As illustrated in the side reaction pathway below, the high nucleophilicity of the amino groups on Compound III often results in the formation of unwanted intermediates like Compound VI, drastically reducing the yield of the desired product.
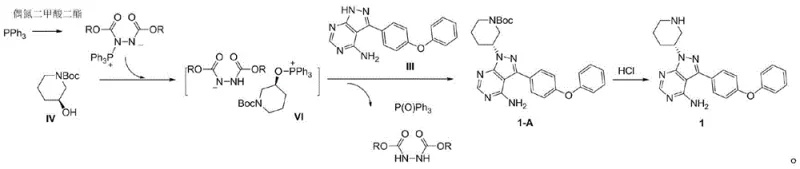
Furthermore, traditional routes often require rigorous purification steps between the coupling and the final acrylation or deprotection stages. The necessity to isolate unstable intermediates increases the risk of decomposition and exposes the process to potential contamination. For supply chain managers, these inefficiencies translate into longer cycle times and higher production costs, as significant amounts of raw materials are lost to side products that are difficult to separate. The complexity of managing multiple reaction vessels and work-up procedures also limits the scalability of these older methods, making them less attractive for large-scale commercial manufacturing.
The Novel Approach
The methodology described in CN111004239B fundamentally reengineers the reaction sequence to address these selectivity issues. Instead of mixing all reagents simultaneously, the process initiates by reacting Compound III with triphenylphosphine and an azodicarboxylic diester to form a protected intermediate (III-B). This step effectively masks the reactive amine sites on the pyrazolo-pyrimidine ring through the formation of a phosphine-ylide complex. Once this protective shell is established, Compound IV is introduced into the system. This sequential addition ensures that the Mitsunobu coupling occurs exclusively at the intended nitrogen atom, bypassing the formation of the problematic side products seen in conventional routes.
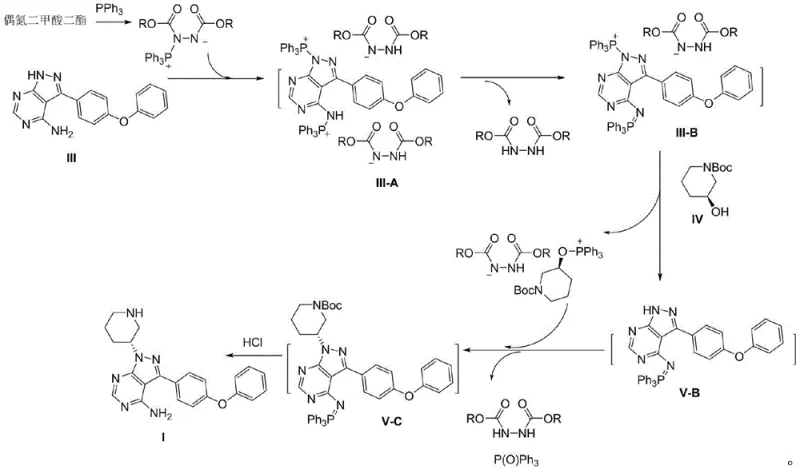
Following the coupling, the process proceeds directly to deprotection using hydrochloric acid within the same reactor. This "one-pot" design eliminates the need for intermediate isolation, filtration, or drying, which are typically resource-intensive operations. The result is a continuous flow of chemistry that maintains the integrity of the chiral center while maximizing the conversion of starting materials into the final precursor. By integrating protection, coupling, and deprotection into a single vessel, the method not only simplifies the operational workflow but also significantly enhances the overall mass balance of the synthesis, offering a compelling alternative for industrial scale-up.
Mechanistic Insights into Phosphine-Mediated Protection Strategy
The success of this novel route hinges on the unique behavior of the phosphine-azodicarboxylate adduct. When Compound III reacts with triphenylphosphine and the azo reagent, it forms an intermediate species, identified in the patent as III-B or potentially III-C depending on the specific protonation state. Mechanistically, the phosphine attacks the azo compound to generate a betaine intermediate, which then interacts with the nucleophilic nitrogens of the heterocycle. This interaction temporarily reduces the electron density on the ring nitrogens, rendering them less susceptible to unwanted alkylation by the activated alcohol species. This transient protection is crucial because it creates a kinetic window where the desired coupling can proceed with high fidelity.
Moreover, the stability of this protected intermediate allows for the subsequent addition of the coupling partner without immediate degradation. The patent data indicates that HPLC analysis shows the complete disappearance of starting Compound III (less than 0.5% remaining) and the quantitative formation of the protected species (over 99.5%). This high conversion rate at the protection stage is a strong indicator of process robustness. Upon completion of the coupling with Compound IV, the addition of concentrated hydrochloric acid serves a dual purpose: it hydrolyzes the phosphine-ylide bond to regenerate the free amine and simultaneously removes the Boc protecting group from the piperidine ring. This tandem deprotection is efficient and avoids the need for separate acidic treatment steps, further streamlining the synthesis.
How to Synthesize (R)-3-(4-phenoxyphenyl)-1-(piperidine-3-yl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine Efficiently
The execution of this synthesis requires precise control over reagent addition and temperature to maintain the integrity of the phosphine intermediates. The process begins by dissolving Compound III and triphenylphosphine in tetrahydrofuran, followed by the dropwise addition of the azodicarboxylate at low temperatures (0°C) to manage exothermicity. After the protection phase is complete, typically indicated by HPLC monitoring, the catalyst and Compound IV are added directly to the mixture. The reaction is then heated to facilitate the coupling, followed by acid treatment for final deprotection.
- React Compound III with triphenylphosphine and azodicarboxylic diester to form a protected phosphine intermediate (III-B).
- Add Compound IV and a phase-transfer catalyst to the reaction mixture to facilitate coupling without isolating the intermediate.
- Treat the resulting intermediate (V-C) with hydrochloric acid to remove protecting groups and isolate the final precursor (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the manufacturing process. By eliminating intermediate isolation steps, the method reduces the consumption of solvents, filtration media, and drying energy. This reduction in unit operations directly correlates to lower variable costs per kilogram of product. Additionally, the "one-pot" nature of the reaction minimizes the physical footprint required for production, allowing existing facilities to increase throughput without significant capital investment in new reactor trains.
- Cost Reduction in Manufacturing: The elimination of purification steps between the protection and coupling phases removes the need for expensive chromatographic columns or extensive recrystallization processes often required to remove regio-isomers in traditional methods. Since the phosphine protection strategy inherently suppresses side reactions, the crude product profile is much cleaner, reducing the burden on downstream purification. This leads to substantial cost savings in terms of both material usage and waste disposal, as fewer byproducts mean less hazardous waste to treat.
- Enhanced Supply Chain Reliability: The robustness of the one-pot process translates to more predictable batch cycles. Traditional multi-step syntheses are prone to delays caused by intermediate quality failures or extended drying times. By consolidating the reaction into a single vessel, the risk of batch rejection is minimized, and the turnaround time between batches is significantly shortened. This reliability ensures a more consistent supply of the critical Ibrutinib precursor, mitigating the risk of stockouts for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges. This method mitigates those risks by reducing the number of transfer operations where spills or exposure could occur. Furthermore, the high atom economy and reduced solvent usage align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The ability to run the reaction at moderate temperatures (25-55°C) also lowers energy consumption compared to processes requiring extreme heating or cooling.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: How does this new method improve upon traditional Mitsunobu reactions for Ibrutinib synthesis?
A: Traditional methods suffer from low yields due to multiple nucleophilic sites on the pyrazolo-pyrimidine core reacting indiscriminately. This patent introduces a transient phosphine protection strategy that blocks these active amine sites, ensuring the reaction occurs selectively at the desired position, thereby significantly improving yield and purity.
Q: Is purification required between steps in this new process?
A: No, the process is designed as a 'one-pot' synthesis. The intermediate formed after the initial protection step is not isolated or purified. The subsequent coupling and deprotection steps occur in the same reactor, which drastically reduces solvent usage, processing time, and material loss.
Q: What catalysts are compatible with this synthesis route?
A: The patent specifies that both nitrogen-containing catalysts (such as tetrabutylammonium bromide) and phosphine-containing catalysts (such as tetraphenylphosphonium bromide) are effective. This flexibility allows manufacturers to select catalysts based on availability and cost without compromising reaction efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ibrutinib Precursor Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving oncology therapies. Our technical team has extensively analyzed the pathways described in CN111004239B and possesses the expertise to implement this advanced one-pot synthesis at commercial scales. We offer extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to manufacturing plant is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Ibrutinib precursor meets the exacting standards required for GMP API synthesis.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and efficient synthetic route. By optimizing the reaction parameters and utilizing our supply chain network, we can deliver significant value to your production timeline. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term supply goals for Ibrutinib and related kinase inhibitors.
