Advanced Synthesis of 4-Substituted 2,2'-Bipyridyl Derivatives for High-Performance OLED and Catalytic Applications
Advanced Synthesis of 4-Substituted 2,2'-Bipyridyl Derivatives for High-Performance OLED and Catalytic Applications
The development of functionalized heterocyclic compounds remains a cornerstone of modern materials science, particularly in the realms of organic optoelectronics and homogeneous catalysis. Patent CN101440059A introduces a robust and versatile synthetic methodology for producing 4-substituted 2,2'-bipyridyl derivatives, a class of compounds renowned for their unique sequestering abilities and electronic properties. These molecules act as pivotal ligands capable of coordinating with a wide array of metal ions, including europium, terbium, platinum, osmium, and ruthenium, thereby facilitating applications ranging from trace metal ion detection to high-efficiency electroluminescent devices. The strategic innovation lies in the utilization of an N-oxide intermediate to overcome the inherent electronic deactivation of the pyridine ring, enabling precise regioselective functionalization at the 4-position. This approach not only simplifies the synthetic pathway compared to traditional methods but also enhances the purity profile essential for high-performance electronic materials. For R&D directors and procurement specialists seeking reliable sources of specialized ligands, understanding this N-oxide activation strategy provides critical insight into the feasibility of scaling these complex architectures for commercial deployment in the OLED and photovoltaic sectors.
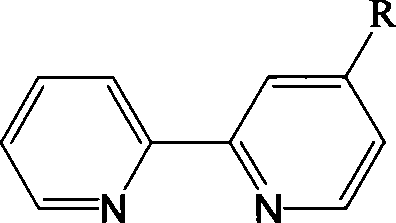
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of asymmetrically substituted 2,2'-bipyridines has been plagued by significant chemical challenges that hinder efficient commercial production. Traditional routes often rely on direct cross-coupling reactions or lithiation strategies that require extremely severe reaction conditions, such as cryogenic temperatures and highly reactive organometallic reagents, which pose substantial safety risks and operational costs in a manufacturing environment. Furthermore, achieving regioselectivity at the 4-position of the bipyridine scaffold is notoriously difficult due to the electron-withdrawing nature of the nitrogen atoms, which deactivates the ring towards electrophilic substitution. Consequently, conventional methods frequently suffer from low yields, the formation of complex isomeric mixtures that are difficult to separate and purify, and the necessity for expensive protecting group strategies. These inefficiencies translate directly into supply chain bottlenecks and elevated costs for downstream users in the fine chemical and electronic materials industries, making the search for a more streamlined process a high priority for procurement managers.
The Novel Approach
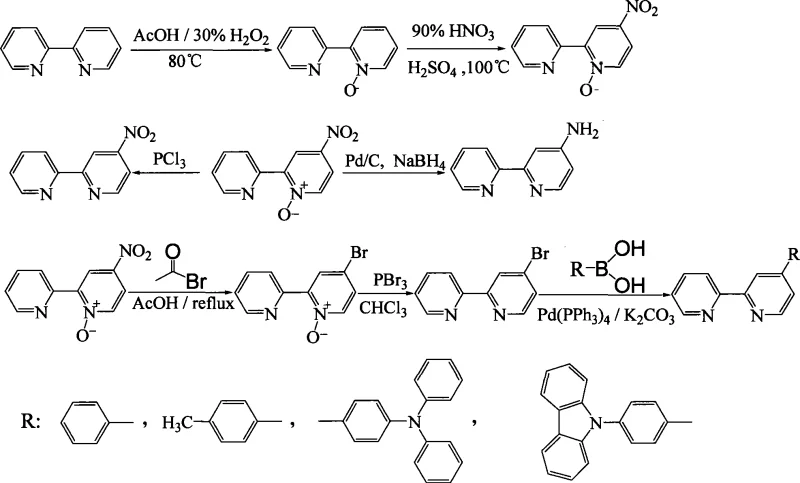
The methodology disclosed in the patent offers a transformative solution by leveraging the electronic activation provided by N-oxidation to direct substitution specifically to the desired 4-position. By first converting the industrial-grade 2,2'-bipyridine into its N-oxide form, the electron density of the pyridine ring is sufficiently modified to allow for smooth nitration and subsequent bromination under relatively mild conditions. This sequence effectively installs a versatile bromine handle at the 4-position, which can then be utilized in palladium-catalyzed cross-coupling reactions to introduce a diverse range of aryl substituents. The process eliminates the need for harsh lithiation steps and minimizes the formation of unwanted isomers, thereby drastically simplifying the purification workflow. This strategic shift from direct functionalization to an activation-substitution-deactivation pathway represents a significant advancement in process chemistry, offering a scalable route to high-purity intermediates that are critical for the fabrication of advanced optoelectronic devices and catalytic systems.
Mechanistic Insights into N-Oxide Activated Regioselective Substitution
The core mechanistic advantage of this synthesis lies in the profound electronic influence of the N-oxide moiety on the pyridine ring's reactivity profile. In the initial oxidation step, hydrogen peroxide in acetic acid converts one of the pyridine nitrogens into an N-oxide, which acts as a strong electron-donating group through resonance, effectively activating the ring towards electrophilic aromatic substitution. This activation is crucial because unmodified bipyridine is too electron-deficient to undergo nitration efficiently without decomposition or polymerization. Once activated, the introduction of the nitro group occurs selectively at the 4-position, para to the nitrogen, driven by the electronic distribution of the N-oxide resonance structures. Following nitration, the nitro group serves as a leaving group precursor or is converted to a bromide via reaction with acetyl bromide, maintaining the regiochemical integrity established in the previous step. The final deoxygenation using phosphorus trichloride restores the aromatic pyridine system while retaining the newly installed substituent, completing the transformation of a simple industrial feedstock into a highly functionalized building block ready for further diversification.
From an impurity control perspective, this stepwise approach offers superior manageability compared to one-pot direct substitution methods. Each intermediate, including the N-oxide, the nitro-N-oxide, and the bromo-N-oxide, possesses distinct physical and chemical properties that facilitate isolation and purification via standard techniques such as recrystallization or column chromatography. For instance, the patent details specific recrystallization solvents like ethanol and hexane that effectively remove side products and unreacted starting materials at each stage. This modular purification strategy ensures that the final 4-substituted products meet the stringent purity specifications required for electronic applications, where trace metal impurities or structural isomers can severely degrade device performance. By breaking the synthesis into discrete, controllable transformations, the process minimizes the accumulation of complex byproducts, thereby enhancing the overall robustness and reproducibility of the manufacturing campaign for these high-value chemical intermediates.
How to Synthesize 4-Bromo-2,2'-dipyridyl Efficiently
The synthesis of the key intermediate, 4-bromo-2,2'-dipyridyl, serves as the foundational step for generating the broader library of 4-substituted derivatives described in the patent. This specific transformation involves a four-stage sequence starting from commercially available 2,2'-bipyridine, progressing through oxidation, nitration, bromination, and finally deoxygenation. The detailed standardized synthetic steps provided in the patent outline precise stoichiometric ratios, temperature controls, and workup procedures necessary to achieve optimal yields, such as the 59.6% isolated yield reported for the final bromo-intermediate. Understanding this specific pathway is essential for process chemists aiming to replicate the results or adapt the conditions for larger scale production, as it demonstrates the practical viability of the N-oxide strategy. The following guide summarizes the critical operational parameters derived from the experimental examples to assist in the efficient preparation of this vital building block.
- Oxidize industrial 2,2'-bipyridine with hydrogen peroxide in acetic acid to form the N-oxide intermediate, activating the pyridine ring for subsequent substitution.
- Perform regioselective nitration at the 4-position using nitric acid and sulfuric acid, followed by bromination with acetyl bromide to install the halogen handle.
- Deoxygenate the N-oxide using phosphorus trichloride to restore the pyridine nitrogen, then utilize Suzuki coupling with aryl boronic acids to introduce diverse substituents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this N-oxide mediated synthetic route presents compelling economic and logistical advantages over legacy manufacturing processes. The primary benefit stems from the utilization of 2,2'-bipyridine as a starting material, which is identified as a common industrial product, ensuring a stable and abundant supply base that mitigates the risk of raw material shortages. Furthermore, the avoidance of extreme cryogenic conditions and highly sensitive organolithium reagents reduces the requirement for specialized low-temperature reactor infrastructure, allowing for production in standard stainless steel vessels commonly found in multi-purpose fine chemical plants. This compatibility with existing manufacturing assets significantly lowers the barrier to entry for scale-up and reduces capital expenditure requirements for new production lines. Additionally, the high regioselectivity of the process minimizes the generation of difficult-to-separate isomers, leading to reduced solvent consumption and waste disposal costs associated with extensive purification efforts, thereby contributing to a more sustainable and cost-effective supply chain operation.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents typically required for direct pyridine functionalization, such as n-butyllithium, results in substantial cost savings regarding both raw material procurement and safety management protocols. By utilizing cheaper oxidants like hydrogen peroxide and common acids like acetic and sulfuric acid for the activation steps, the overall cost of goods sold (COGS) is significantly optimized. Moreover, the high yields reported for intermediate steps, such as the 89% yield for the bromination of the N-oxide, indicate an efficient atom economy that reduces material waste and maximizes output per batch. This efficiency translates directly into competitive pricing for the final 4-substituted derivatives, making them more accessible for high-volume applications in the display and lighting industries without compromising on quality.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals for the initial steps of the synthesis creates a resilient supply chain that is less susceptible to the volatility often seen with specialized fine chemical precursors. The robustness of the reaction conditions, which tolerate standard heating and reflux rather than requiring precise cryogenic control, ensures consistent batch-to-batch reproducibility even when scaling from pilot to commercial tonnage. This reliability is critical for downstream customers in the OLED and photovoltaic sectors who require uninterrupted delivery of high-purity ligands to maintain their own production schedules. The ability to source the core scaffold from established industrial suppliers further de-risks the procurement process, providing a secure foundation for long-term supply agreements and strategic partnerships.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable due to its reliance on well-understood unit operations such as oxidation, nitration, and filtration, which are easily adapted for large-scale continuous or batch processing. The use of phosphorus trichloride for deoxygenation, while requiring careful handling, is a standard industrial procedure with established waste treatment protocols, ensuring compliance with environmental regulations. By minimizing the use of exotic catalysts in the early stages and focusing on stoichiometric transformations that are easy to monitor and control, the process facilitates a smoother technology transfer from laboratory to plant. This scalability ensures that the supply of these critical intermediates can grow in tandem with the expanding demand for organic electronic materials, supporting the industry's transition to next-generation display and energy technologies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these 4-substituted 2,2'-bipyridyl derivatives, based on the detailed disclosures within the patent documentation. These insights are intended to clarify the process capabilities and potential integration points for partners in the fine chemical and electronic materials sectors. Understanding these specifics helps stakeholders evaluate the suitability of this technology for their specific R&D pipelines and manufacturing requirements.
Q: Why is the N-oxide strategy preferred for 4-position substitution on bipyridine?
A: Direct electrophilic substitution on 2,2'-bipyridine is difficult due to the electron-deficient nature of the pyridine rings and protonation under acidic conditions. Converting one ring to an N-oxide significantly activates the ring towards electrophilic attack, specifically directing nitration and bromination to the 4-position with high regioselectivity, which is otherwise hard to achieve.
Q: What are the primary applications of these 4-substituted derivatives?
A: These derivatives serve as critical ligands for coordinating with metal ions such as ruthenium, europium, and platinum. They are extensively used in organic light-emitting diodes (OLEDs) as emissive or transport materials, in dye-sensitized solar cells as photosensitizers, and as indicators for detecting trace metal ions in analytical chemistry.
Q: Is the starting material readily available for large-scale production?
A: Yes, the synthesis begins with 2,2'-bipyridine, which is described in the patent as a common industrial product. This ensures a stable and cost-effective supply chain foundation, avoiding the need for complex de novo synthesis of the bipyridine core itself.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Substituted 2,2'-Bipyridyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity ligands play in the advancement of organic optoelectronic materials and catalytic systems. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated N-oxide activation chemistry described in patent CN101440059A can be successfully translated from the laboratory bench to industrial reality. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 4-substituted 2,2'-bipyridyl derivatives meets the exacting standards required for OLED and solar cell applications. Our commitment to quality assurance means that clients can rely on us for consistent material performance, minimizing the risk of device failure due to chemical impurities.
We invite R&D directors and procurement leaders to collaborate with us to leverage this advanced synthetic technology for their next-generation projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can support your supply chain goals and accelerate your time to market for innovative electronic materials.
