Advanced Chiral Spiroindane Ligands: Enabling High-Selectivity Asymmetric Catalysis for Pharma
Advanced Chiral Spiroindane Ligands: Enabling High-Selectivity Asymmetric Catalysis for Pharma
The landscape of asymmetric synthesis is constantly evolving, driven by the relentless demand for enantiomerically pure pharmaceutical intermediates. Patent CN116041207A introduces a significant breakthrough in this domain with the disclosure of a novel class of chiral spiroindane ligands. These compounds, characterized by a rigid 1,1'-spirobis[indene] backbone functionalized with N-acyl amino acid moieties, represent a strategic advancement over traditional phosphine and diol-based systems. The core innovation lies in the specific structural integration of the spiro-indane framework with amino acid derivatives, creating a ligand environment that exerts precise stereochemical control during catalytic cycles. This technology addresses critical bottlenecks in modern drug discovery, particularly in the realm of C-H bond activation, where achieving high enantioselectivity has historically been a formidable challenge.
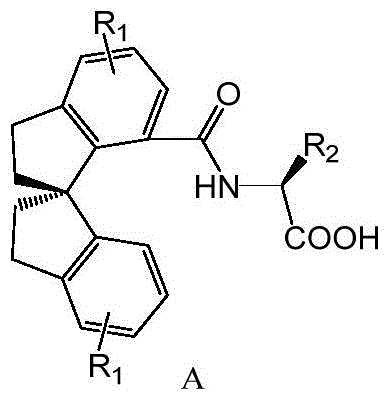
For R&D directors evaluating new catalytic technologies, the structural integrity and modularity of these ligands are paramount. The general formula A allows for diversification at the R1 and R2 positions, enabling fine-tuning of steric and electronic properties to match specific substrate requirements. This adaptability is crucial for developing robust processes that can handle diverse molecular architectures without requiring a complete redesign of the catalytic system. The patent explicitly highlights the ligand's capability to form stable cationic complexes with transition metals like palladium, which serves as the foundation for its exceptional performance in asymmetric carbon-carbon and carbon-heteroatom bond formation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of chiral compounds has relied heavily on racemate resolution or stoichiometric chiral induction, both of which suffer from inherent inefficiencies. Racemate resolution is fundamentally limited by a maximum theoretical yield of 50%, resulting in significant material waste and increased processing costs for the discarded enantiomer. Furthermore, stoichiometric chiral auxiliaries often require harsh conditions for attachment and removal, generating substantial chemical waste and complicating the purification process. Even among catalytic methods, traditional ligands such as BINOL derivatives can exhibit flexibility that compromises the rigidity of the chiral pocket, leading to variable enantioselectivity across different substrate classes. In the specific context of C-H activation, the inertness of the C-H bond requires highly active and selective catalysts, which many conventional ligand systems fail to provide consistently under mild conditions.
The Novel Approach
The methodology described in CN116041207A circumvents these limitations through a rationally designed ligand architecture that combines the rigidity of the spiroindane skeleton with the coordinating ability of amide and carboxyl groups. By introducing the N-acyl amino acid structure directly into the spiro ring system, the inventors have created a ligand that enforces a strict C2 symmetric environment around the metal center. This rigidity minimizes conformational freedom, ensuring that the substrate approaches the catalytic site in a highly predictable manner. The result is a catalytic system that operates with high turnover and exceptional enantioselectivity, often exceeding 90% ee, even for challenging transformations. This approach not only improves the efficiency of the reaction but also simplifies downstream processing by reducing the formation of diastereomeric impurities that are difficult to separate.
Mechanistic Insights into Pd-Catalyzed Asymmetric C-H Activation
The efficacy of this ligand class is rooted in its ability to stabilize high-valent palladium intermediates while maintaining a chiral environment conducive to enantio-determining steps. When complexed with palladium, the ligand forms a cationic species that is highly electrophilic, facilitating the difficult task of cleaving strong C-H bonds. The spiroindane backbone acts as a bulky, rigid scaffold that shields one face of the metal center, forcing the substrate to coordinate from the less hindered side. This steric differentiation is amplified by the specific orientation of the amino acid side chain (R2 group), which can engage in secondary interactions with the substrate to further enhance selectivity. The patent data indicates that this mechanism is particularly effective for constructing C-C and C-X bonds, enabling the direct functionalization of unactivated positions on complex molecules.
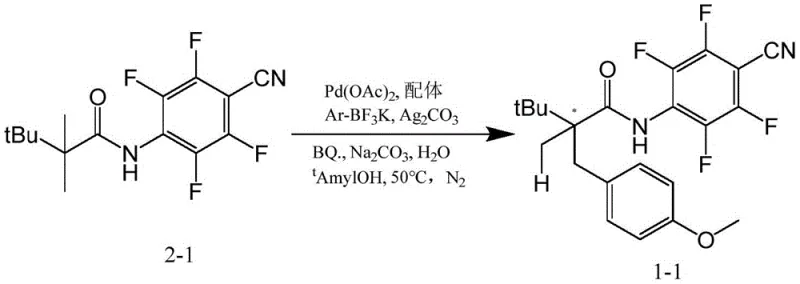
Furthermore, the ligand design effectively suppresses common side reactions such as beta-hydride elimination or non-selective background reactions. The coordination geometry imposed by the tridentate-like binding mode (involving the amide oxygen, nitrogen, and potentially the carboxylate) locks the metal in a specific configuration that favors the desired reductive elimination pathway. This level of control is essential for the synthesis of complex pharmaceutical intermediates where regio- and stereoselectivity are critical quality attributes. The successful application in the synthesis of compound 1-1, achieving a 95% yield and 99% ee value as shown in the patent examples, serves as a powerful validation of this mechanistic hypothesis. It demonstrates that the ligand can tolerate various functional groups, including nitriles and fluorines, which are prevalent in modern medicinal chemistry.
How to Synthesize Chiral Spiroindane Ligand Efficiently
The preparation of these high-value ligands follows a logical and scalable synthetic route that begins with commercially available or easily accessible spiroindane diols. The process involves a sequence of protection, functionalization, and coupling steps that are well-suited for industrial scale-up. Key to the synthesis is the conversion of the spiroindane carboxylic acid precursor (Formula B) into its corresponding acid chloride, which is then coupled with the desired amino acid. This amide bond formation is the critical step that installs the chiral information from the amino acid onto the rigid spiro scaffold. The conditions employed, such as the use of oxalyl chloride and triethylamine in dichloromethane, are standard in fine chemical manufacturing, minimizing the need for specialized equipment or hazardous reagents.
- Activation of 7-carboxy-1,1'-spiroindane carboxylic acid (Formula B) using oxalyl chloride and DMF catalyst in DCM at 0-10°C to form the acyl chloride intermediate.
- Coupling reaction where the activated acyl chloride is added to an amino acid derivative (Formula C) in the presence of triethylamine base at room temperature.
- Purification of the final ligand (Formula A) via quenching and column chromatography, achieving yields up to 95% with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this ligand technology offers tangible benefits related to cost efficiency and supply security. The synthetic route relies on commodity chemicals such as oxalyl chloride, triethylamine, and common amino acids, which ensures a stable and competitive supply chain for raw materials. Unlike exotic ligands that require multi-step syntheses from rare natural products, the spiroindane core can be produced in large quantities through established petrochemical pathways. This abundance of starting materials translates directly into lower production costs and reduced vulnerability to supply disruptions. Additionally, the high catalytic efficiency means that lower loadings of the expensive metal catalyst may be required, further driving down the cost of goods sold for the final API intermediate.
- Cost Reduction in Manufacturing: The implementation of this ligand system eliminates the need for stoichiometric chiral reagents and the associated waste disposal costs. By shifting from resolution-based strategies to catalytic asymmetric synthesis, manufacturers can theoretically double their yield from the same amount of starting material. The robust nature of the catalyst also allows for milder reaction conditions, reducing energy consumption associated with heating or cooling. Furthermore, the high selectivity minimizes the formation of byproducts, simplifying purification workflows and reducing solvent usage, which constitutes a significant portion of manufacturing expenses in the pharmaceutical industry.
- Enhanced Supply Chain Reliability: The modular nature of the ligand synthesis allows for flexible production scheduling. Since the core spiroindane acid (Formula B) can be stockpiled and then coupled with various amino acids on demand, suppliers can respond quickly to changing customer requirements without maintaining large inventories of finished ligands. This "semi-finished" strategy mitigates the risk of obsolescence and ensures that specific ligand variants can be delivered with shorter lead times. The use of stable intermediates also enhances the shelf-life of the supply, reducing losses due to degradation during storage and transport.
- Scalability and Environmental Compliance: The synthetic procedures described in the patent utilize solvents like DCM and THF, which, while requiring careful handling, are well-understood in terms of recovery and recycling in modern chemical plants. The high atom economy of the catalytic process aligns with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. The ability to run reactions at or near room temperature reduces the carbon footprint of the manufacturing process. Moreover, the high purity of the product obtained directly from the reaction reduces the need for extensive recrystallization or chromatography, streamlining the path to commercial scale production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral spiroindane ligand technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these details is essential for project managers assessing the feasibility of integrating this new catalytic system into existing production lines.
Q: What are the key structural advantages of this new spiroindane ligand?
A: The ligand features a rigid C2-symmetric spiroindane backbone combined with an N-acyl amino acid motif. This unique architecture provides a highly defined chiral environment around the metal center, crucial for achieving enantioselectivities exceeding 90% in challenging C-H activation reactions.
Q: How does this ligand improve upon traditional BINOL-derived systems?
A: Unlike flexible BINOL derivatives, the spiroindane skeleton offers superior rigidity and stability. The patent data demonstrates that this rigidity translates to broader substrate applicability and higher turnover frequencies in palladium-catalyzed asymmetric transformations, particularly for sterically hindered substrates.
Q: Is the synthesis of this ligand scalable for industrial production?
A: Yes, the preparation method utilizes standard organic solvents like DCM and THF with common reagents such as oxalyl chloride and triethylamine. The multi-step synthesis of the precursor acid (Formula B) involves robust reactions like methylation, triflation, and carbonylation, all of which are amenable to kilogram-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Spiroindane Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced chiral ligands like those described in CN116041207A for the next generation of pharmaceutical synthesis. As a dedicated CDMO partner, we possess the technical expertise to translate these laboratory-scale innovations into robust, commercial-scale processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ligand or intermediate meets the exacting standards required for GMP production.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific drug development programs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your current synthetic route, highlighting exactly where this new ligand can drive efficiency. Please contact our technical procurement team today to request specific COA data for our chiral building blocks and to discuss route feasibility assessments for your target molecules. Let us help you secure a competitive advantage through superior catalytic performance and supply chain reliability.
