Advanced Iridium-Catalyzed Synthesis of Phenyl-(3-aryl-2-propenyl) Selenides for Commercial Scale-up
Advanced Iridium-Catalyzed Synthesis of Phenyl-(3-aryl-2-propenyl) Selenides for Commercial Scale-up
The landscape of organoselenium chemistry has evolved significantly with the introduction of transition metal catalysis, offering new pathways for constructing complex molecular architectures essential for modern drug discovery. Patent CN102249969A introduces a groundbreaking methodology for the synthesis of phenyl-(3-aryl-2-propenyl) selenide compounds, utilizing a sophisticated iridium-based catalytic system. This technology addresses long-standing challenges in the field by enabling the direct coupling of sodium phenylselenolate with allyl carbonate derivatives under remarkably mild conditions. The resulting compounds serve as critical precursors for selenium-containing heterocycles and potential anticancer agents, making this synthetic route highly valuable for the production of high-purity pharmaceutical intermediates. By leveraging the unique electronic properties of iridium complexes combined with specialized chiral ligands, this process achieves superior regiocontrol compared to traditional methods.
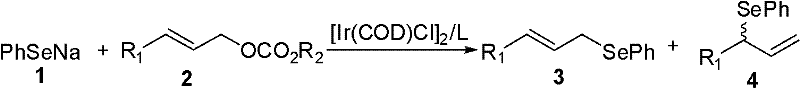
For procurement specialists and supply chain managers seeking a reliable pharmaceutical intermediate supplier, understanding the robustness of this catalytic cycle is paramount. The ability to operate within a temperature window of -10°C to 50°C eliminates the need for extreme cryogenic conditions or high-energy heating, which translates directly into operational expenditure savings. Furthermore, the compatibility of this method with a diverse range of substrates, including those with electron-donating and electron-withdrawing groups, ensures that a wide library of analogues can be accessed from a single platform. This versatility is crucial for R&D teams aiming to optimize structure-activity relationships without being bottlenecked by synthetic feasibility. The patent explicitly highlights the formation of linear products with high fidelity, minimizing the formation of branched isomers that often complicate purification workflows in large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of allyl selenides has relied heavily on the nucleophilic substitution of allyl halides with aryl selenide anions, a pathway fraught with significant inefficiencies. These traditional protocols often suffer from prolonged reaction times and mediocre yields, primarily due to the competing elimination reactions and the relatively poor leaving group ability of halides in certain steric environments. Alternative approaches involving allyl acetates activated by zinc iodide or palladium catalysts have also been explored, yet these methods frequently struggle with poor atom economy and limited substrate scope. The use of palladium, in particular, introduces concerns regarding heavy metal contamination, necessitating rigorous and costly purification steps to meet stringent pharmaceutical quality standards. Additionally, many of these legacy processes lack the necessary regioselectivity, producing mixtures of linear and branched isomers that require resource-intensive chromatographic separation, thereby inflating the overall cost of goods.
The Novel Approach
In stark contrast, the novel iridium-catalyzed allylic substitution described in the patent represents a paradigm shift in efficiency and selectivity. By employing allyl carbonates as electrophiles, the reaction benefits from the generation of a highly reactive pi-allyl iridium intermediate that facilitates smooth nucleophilic attack by the selenium species. This mechanistic advantage allows the reaction to proceed with exceptional regioselectivity, often exceeding a ratio of 99:1 in favor of the desired linear product. The operational simplicity is another key differentiator; the reaction can be conducted in common organic solvents such as dichloromethane or toluene without the need for exotic additives. For manufacturers focused on cost reduction in pharmaceutical intermediate manufacturing, this translates to a streamlined process that reduces waste generation and solvent consumption. The mild reaction conditions further enhance the safety profile of the operation, making it an attractive candidate for scale-up in standard glass-lined or stainless steel reactors.
Mechanistic Insights into Iridium-Catalyzed Allylic Substitution
The core of this technological advancement lies in the precise orchestration of the catalytic cycle involving the [Ir(COD)Cl]2 precursor and a specialized chiral phosphoramidite ligand. Upon activation, typically involving an amine base to neutralize generated acid, the iridium center coordinates with the allyl carbonate to form a cationic pi-allyl complex. This intermediate is electronically tuned by the chiral ligand to favor nucleophilic attack at the terminal carbon position, thereby dictating the high linear regioselectivity observed in the experimental data. The ligand structure, featuring a binaphthyl backbone, provides a rigid chiral environment that not only controls stereochemistry but also stabilizes the transition state against decomposition. Understanding this interaction is vital for process chemists aiming to troubleshoot potential deviations during scale-up, as the integrity of the ligand-metal complex is directly correlated with catalytic turnover numbers.
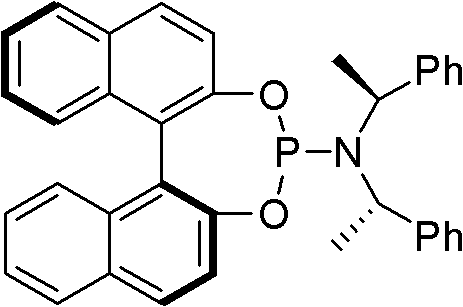
Impurity control is inherently built into this mechanism due to the kinetic preference for the linear pathway over the branched pathway. In conventional thermal reactions, thermodynamic control might lead to equilibration and isomer mixtures, but the iridium-catalyzed process operates under kinetic control, effectively locking in the desired product geometry. This minimizes the presence of regioisomeric impurities that could otherwise act as genotoxic alerts or complicate crystallization processes downstream. Furthermore, the use of sodium phenylselenolate as a nucleophile is advantageous because it is a potent soft nucleophile that matches well with the soft electrophilic character of the pi-allyl iridium species. This matching of hardness and softness principles ensures rapid reaction kinetics even at lower temperatures, preserving sensitive functional groups on the aromatic rings of the substrate. For quality assurance teams, this mechanistic robustness means a more consistent impurity profile across different batches, facilitating easier regulatory filing and validation.
How to Synthesize Phenyl-(3-aryl-2-propenyl) Selenides Efficiently
Executing this synthesis requires careful attention to the preparation of the active catalyst species prior to the introduction of the substrates. The protocol involves pre-forming the iridium-ligand complex in a dry, inert atmosphere to prevent oxidation of the sensitive metal center, followed by the sequential addition of the selenium salt and the carbonate electrophile. The choice of solvent plays a critical role, with 1,2-dichloroethane often providing the optimal balance of solubility and reaction rate, although dichloromethane and toluene are also viable alternatives depending on the specific substrate solubility profile. Reaction monitoring is typically achieved via thin-layer chromatography or HPLC, with completion usually reached within 6 to 48 hours depending on the steric demand of the R1 group. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures.
- Prepare the active iridium catalyst by reacting [Ir(COD)Cl]2 with chiral ligand L and n-propylamine in THF at 50°C, followed by cooling and solvent removal.
- Combine sodium phenylselenolate and allyl carbonate substrates in an organic solvent such as DCE or DCM under an inert argon atmosphere.
- Stir the reaction mixture at temperatures between -10°C and 50°C for 6 to 48 hours, then purify the crude product via column chromatography.
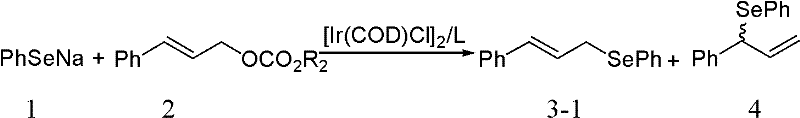
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this iridium-catalyzed technology offers substantial benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the purification train; because the reaction produces minimal regioisomeric byproducts, the need for extensive preparative chromatography is significantly reduced or potentially eliminated in favor of crystallization. This reduction in downstream processing time directly correlates to increased throughput and lower labor costs per kilogram of finished product. Moreover, the mild operating temperatures reduce the energy load on manufacturing facilities, aligning with sustainability goals and lowering utility expenses. For supply chain heads concerned with continuity, the use of stable allyl carbonate starting materials ensures a reliable feedstock supply, as these compounds are generally more stable and easier to store than reactive allyl halides.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the reduction in purification complexity lead to significant cost optimization opportunities. By avoiding the use of stoichiometric activators like zinc iodide and minimizing the formation of hard-to-remove impurities, the overall material cost per unit is lowered. The high atom economy of the carbonate leaving group compared to halides also contributes to less chemical waste disposal, further enhancing the economic viability of the process. Additionally, the catalyst loading is kept relatively low, ensuring that the cost of the precious metal does not disproportionately impact the final product price.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions allows for flexible scheduling and reduced risk of batch failure. Since the reaction tolerates a variety of functional groups and solvents, procurement teams have multiple options for sourcing raw materials without compromising the reaction outcome. This flexibility mitigates the risk of supply disruptions caused by the unavailability of a specific grade of solvent or reagent. The stability of the intermediates also allows for potential telescoping of steps, where the crude product from one stage can be fed directly into the next, shortening the overall manufacturing lead time.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of exothermic hazards associated with highly reactive halides. The mild temperature profile means that standard cooling systems are sufficient, removing the need for specialized cryogenic equipment. From an environmental standpoint, the reduction in solvent usage and waste generation supports compliance with increasingly strict environmental regulations. The process generates fewer hazardous byproducts, simplifying waste treatment protocols and reducing the environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and specifications outlined in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is essential for successfully transferring this technology from the bench to the pilot plant.
Q: What is the regioselectivity of this iridium-catalyzed reaction?
A: The process demonstrates exceptionally high regioselectivity, typically achieving ratios greater than 99:1 for the linear product over the branched isomer, significantly simplifying downstream purification.
Q: Which solvents are compatible with this synthesis method?
A: The patent specifies several effective organic solvents including 1,2-dichloroethane (DCE), dichloromethane (DCM), toluene, and tetrahydrofuran (THF), allowing flexibility for process optimization.
Q: What temperature range is required for optimal yield?
A: The reaction proceeds efficiently under mild conditions ranging from -10°C to 50°C, with room temperature often providing a balance between reaction rate and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyl-(3-aryl-2-propenyl) Selenides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality organoselenium intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch meets the highest industry standards. Our commitment to technical excellence means we can adapt the iridium-catalyzed protocol to suit your specific volume requirements while maintaining the high regioselectivity and yield demonstrated in the patent literature.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this catalytic method for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency, quality, and mutual success.
