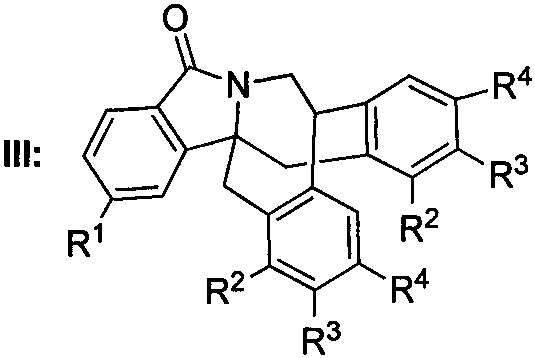
Scalable Synthesis of Bridged Isoindolinone Derivatives for Advanced Fungicide Development
The pharmaceutical and agrochemical industries are constantly seeking novel heterocyclic scaffolds that offer enhanced biological activity and improved synthetic accessibility. In this context, patent CN112824411B introduces a groundbreaking class of bridged isoindolinone derivatives, characterized by a unique polycyclic architecture that demonstrates potent fungicidal properties. Unlike traditional isoindolinone syntheses that often struggle with structural complexity and low yields, this invention provides a robust general formula (Structure III) that allows for extensive diversification at the R1 through R4 positions. This structural flexibility is critical for optimizing biological efficacy against resistant fungal strains. By leveraging a strategic combination of organolithium chemistry and Lewis acid catalysis, the disclosed method transforms simple o-bromobenzaldehyde precursors into high-value endocyclic isoindolinone cores. For R&D directors and procurement specialists alike, this represents a significant opportunity to access a new chemical space with a reliable agrochemical intermediate supplier capable of navigating complex heterocyclic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polycyclic isoindolinone derivatives has been plagued by significant synthetic hurdles that hinder commercial viability. Traditional routes frequently rely on multi-step sequences involving expensive transition metal catalysts, such as palladium or rhodium, which not only inflate raw material costs but also introduce challenging impurity profiles related to heavy metal residues. Furthermore, many existing methodologies require harsh reaction conditions, including high temperatures and strong acidic or basic environments, which can lead to substrate decomposition and poor selectivity. The background art highlights that substrate complexity is a major bottleneck, often necessitating the preparation of specialized, hard-to-source starting materials that delay project timelines. Additionally, the operational complexity of purifying these dense polycyclic structures often results in substantial yield losses during chromatography, making the cost reduction in fungicide manufacturing a difficult target to achieve using legacy technologies.
The Novel Approach
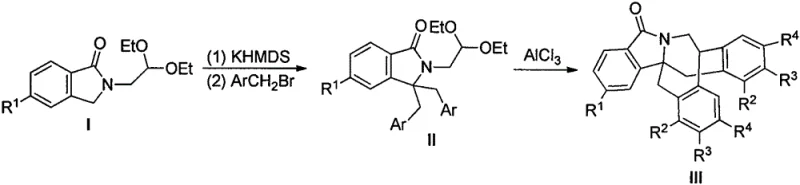
The methodology disclosed in the patent offers a transformative solution by streamlining the synthesis into a concise, three-step sequence that prioritizes operational simplicity and atom economy. The core innovation lies in the use of a lithium-halogen exchange followed by carbon monoxide insertion to rapidly build the isoindolinone skeleton from readily available o-bromobenzaldehyde imines. This is followed by a mild alkylation step using potassium bis(trimethylsilyl)amide (KHMDS) and a final, elegant cyclization mediated by aluminum trichloride. This route eliminates the need for precious metal catalysts entirely, replacing them with abundant and cost-effective Lewis acids. The ability to perform key transformations at room temperature significantly reduces energy consumption compared to thermal cyclization methods. By simplifying the purification workflow to standard silica gel chromatography with common eluents like ethyl acetate and petroleum ether, this approach drastically lowers the barrier to entry for commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into AlCl3-Catalyzed Cyclization and Lithiation
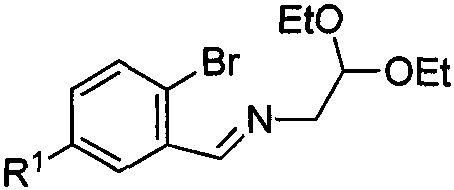
The success of this synthetic strategy hinges on a sophisticated understanding of organometallic reactivity and electrophilic aromatic substitution. The initial formation of the isoindolinone core (Intermediate I) involves a low-temperature lithiation at -78°C, where n-butyllithium facilitates a halogen-lithium exchange on the aromatic ring. The subsequent exposure to carbon monoxide allows for the insertion of a carbonyl group, effectively closing the five-membered lactam ring upon hydrolysis. This step is crucial for establishing the nitrogen-containing heterocycle that serves as the foundation for the bridged system. The structural integrity of the starting imine, as depicted in the patent data, ensures that the nitrogen atom is protected and positioned correctly for the subsequent cyclization events, preventing unwanted side reactions during the harsh lithiation conditions.
The final cyclization step to form the bridged structure III is a masterpiece of Lewis acid catalysis. When Intermediate II is treated with powdered aluminum trichloride (AlCl3) in dichloromethane, the Lewis acid coordinates with the carbonyl oxygen or potentially activates the benzylic position, facilitating an intramolecular Friedel-Crafts alkylation. This reaction forge the critical carbon-carbon bond that bridges the isoindolinone core with the pendant aromatic ring, creating the rigid, three-dimensional architecture essential for high-affinity binding to fungal targets. The mechanism avoids the formation of unstable carbocation intermediates that typically lead to polymerization, thanks to the controlled environment provided by the solvent and the stoichiometry of the AlCl3. This precise control over the reaction pathway ensures high purity specifications and minimizes the formation of regioisomers, which is a common pain point in the synthesis of fused polycyclic systems.
How to Synthesize Bridged Isoindolinone Derivatives Efficiently
The synthesis of these high-value fungicidal intermediates requires strict adherence to inert atmosphere techniques and precise temperature control to maximize yield and purity. The process begins with the careful generation of the lithiated species at cryogenic temperatures, followed by a controlled warming and alkylation sequence that builds molecular complexity without compromising stability. The final cyclization is remarkably robust, proceeding efficiently at room temperature, which simplifies the equipment requirements for production. While the general procedure is outlined below, achieving consistent results on a multi-kilogram scale demands expertise in handling reactive organolithium reagents and moisture-sensitive Lewis acids. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup protocols optimized for reproducibility.
- Preparation of Starting Material I via lithiation of o-bromobenzaldehyde imine followed by carbon monoxide insertion at -78°C.
- Alkylation of the isoindolinone precursor using KHMDS and substituted benzyl bromides at room temperature to form Intermediate II.
- Intramolecular cyclization of Intermediate II using powdered aluminum trichloride (AlCl3) in dichloromethane to yield the final bridged derivative III.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits that directly impact the bottom line. By shifting away from precious metal catalysis to base-metal Lewis acids like aluminum chloride, the raw material costs are significantly reduced, and the supply chain becomes less vulnerable to the volatility of the platinum group metals market. Furthermore, the elimination of heavy metal catalysts removes the need for expensive and time-consuming scavenging steps to meet regulatory limits on residual metals in agrochemical products. This simplification of the downstream processing workflow translates to shorter batch cycle times and higher throughput in manufacturing facilities. The use of common solvents such as tetrahydrofuran and dichloromethane ensures that the process can be implemented in existing infrastructure without requiring specialized reactor modifications, thereby enhancing supply chain reliability and reducing lead time for high-purity agrochemical intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive palladium or rhodium catalysts with inexpensive aluminum trichloride and potassium bases results in substantial cost savings per kilogram of product. Additionally, the ability to conduct the critical cyclization step at room temperature eliminates the energy costs associated with heating reactors to high temperatures for extended periods. The simplified purification process, which relies on standard silica gel chromatography rather than preparative HPLC or complex crystallization sequences, further reduces solvent consumption and waste disposal costs. These factors combine to create a highly economical manufacturing process that improves the overall margin profile for fungicide production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, primarily o-bromobenzaldehyde derivatives and aminoacetals, are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, particularly the tolerance of the final cyclization step to ambient temperatures, reduces the risk of batch failures due to equipment malfunction or temperature excursions. This reliability ensures a consistent supply of critical intermediates, allowing downstream formulation teams to plan their production schedules with greater confidence and stability. The modular nature of the synthesis also allows for rapid adjustment of R-groups to meet changing market demands for specific fungicidal profiles.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, as demonstrated by the successful preparation of various derivatives in the patent examples with good yields. The absence of toxic heavy metals simplifies the environmental compliance landscape, reducing the burden on wastewater treatment facilities and lowering the cost of hazardous waste disposal. The use of standard organic solvents allows for efficient recovery and recycling systems to be integrated into the production line, aligning with modern green chemistry principles. This environmental friendliness not only reduces operational costs but also enhances the corporate sustainability profile of the manufacturing entity, which is increasingly important for global agrochemical customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these bridged isoindolinone derivatives. The answers are derived directly from the experimental data and mechanistic insights provided in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing product pipeline. We encourage further discussion with our technical team to explore specific customization options for your project needs.
Q: What are the key advantages of this synthesis method over conventional polycyclic isoindolinone preparations?
A: Conventional methods often suffer from complicated substrates, lengthy synthesis procedures, and complex operations. This novel approach utilizes readily available o-bromobenzaldehyde derivatives and employs a streamlined three-step sequence with mild reaction conditions (mostly room temperature), significantly simplifying the operational complexity and improving overall process efficiency.
Q: What is the biological activity profile of these bridged isoindolinone derivatives?
A: These compounds exhibit significant fungicidal activity against various plant pathogens. Specifically, derivatives such as III-3, III-5, and III-15 show over 70% inhibition against Sclerotinia sclerotiorum, while others demonstrate potent activity against Botrytis cinerea and Rhizoctonia cerealis, making them highly valuable candidates for agricultural fungicide development.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly amenable to scale-up. It avoids the use of expensive transition metal catalysts that require rigorous removal, relies on standard reagents like AlCl3 and KHMDS, and utilizes simple workup procedures involving hydrolysis and solvent extraction, which are easily adaptable to multi-kilogram production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bridged Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel bridged isoindolinone scaffold in the next generation of agricultural fungicides. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your innovative molecules can transition smoothly from the laboratory to the field. Our state-of-the-art facilities are equipped to handle the sensitive organolithium and Lewis acid chemistry required for this synthesis, maintaining stringent purity specifications through our rigorous QC labs. We understand that consistency and quality are paramount in agrochemical manufacturing, and our dedicated process chemistry team is ready to optimize every step of this route to meet your specific cost and timeline objectives.
We invite you to collaborate with us to unlock the full commercial potential of these derivatives. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that identifies specific opportunities to optimize your supply chain and reduce manufacturing expenses. We encourage you to contact our technical procurement team today to request specific COA data for our reference standards and comprehensive route feasibility assessments tailored to your project requirements. Let us help you bring these powerful fungicidal solutions to market faster and more efficiently.
