Revolutionizing Huntington’s Disease API Intermediate Production via Aqueous Phase Transfer Catalysis
Revolutionizing Huntington’s Disease API Intermediate Production via Aqueous Phase Transfer Catalysis
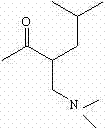
The pharmaceutical landscape for treating neurodegenerative disorders such as Huntington's Chorea (HD) relies heavily on the efficient supply of high-purity intermediates like 9,10-dimethoxy-1,3,4,6,7,11b-hexahydro-3-isobutyl-2H-benzo[a]quinolizidine-2-one. Patent CN102260255B introduces a transformative synthetic methodology that addresses the longstanding inefficiencies associated with traditional tetrabenazine precursor manufacturing. This innovation shifts the paradigm from hazardous, multi-step organometallic processes to a streamlined, green chemistry approach utilizing accessible raw materials. For R&D directors and procurement strategists, this patent represents a critical opportunity to optimize the supply chain for VMAT2 inhibitors, ensuring both economic viability and regulatory compliance in an increasingly stringent global market.
![Chemical structure of the target pharmaceutical intermediate 9,10-dimethoxy-3-isobutyl-benzo[a]quinolizidine-2-one](/insights/img/tetrabenazine-intermediate-synthesis-green-catalysis-pharma-supplier-20260305033326-02.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this benzo[a]quinolizidine core has been dominated by legacy methodologies, such as those described in US Patent 2830993 by Hoffmann-La Roche, which present significant operational and economic bottlenecks. These conventional routes typically rely on the condensation of 1-ethoxycarbonyl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline with isobutyl-dimethyl malonate, necessitating the use of highly reactive and dangerous sodium metal in methanol. The process requires prolonged reflux periods extending up to 16 hours at elevated temperatures around 120°C in toluene, creating severe safety hazards and energy consumption issues. Furthermore, the reliance on toxic aromatic solvents like toluene complicates waste management and increases the environmental footprint, making these methods increasingly untenable for modern GMP-compliant facilities seeking to minimize their ecological impact and operational risk profiles.
The Novel Approach
In stark contrast, the methodology disclosed in CN102260255B offers a radically simplified two-step sequence that bypasses the need for hazardous reagents and harsh conditions. By utilizing 5-methyl-2-hexanone, a cheap and readily available commodity chemical, as the starting material, the process fundamentally alters the cost structure of the synthesis. The reaction conditions are remarkably mild, proceeding in ethanol for the initial step and transitioning to an aqueous medium for the critical cyclization, thereby eliminating the bulk of organic solvent usage. This approach not only reduces the reaction time significantly, often completing the key transformation in just 4 hours, but also simplifies the isolation procedure to basic filtration and recrystallization, offering a robust pathway for industrial scale-up that aligns perfectly with green chemistry principles.
Mechanistic Insights into Mannich Condensation and Aqueous Cyclization
The core of this synthetic breakthrough lies in the strategic application of a Mannich reaction followed by a phase-transfer catalyzed cyclization. The first stage involves the condensation of 5-methyl-2-hexanone with formaldehyde and dimethylamine hydrochloride in ethanol, generating the key intermediate 3-dimethylaminomethyl-5-methyl-hexan-2-one. This step effectively installs the necessary carbon framework and amine functionality required for the subsequent ring closure. The precision of this step is critical, as the stoichiometry of formaldehyde and dimethylamine must be carefully controlled to prevent over-alkylation or polymerization, ensuring a clean profile for the downstream reaction. The use of ethanol as a solvent here provides an optimal balance of solubility for the reactants while facilitating the removal of water formed during the condensation through azeotropic distillation if necessary.
The second and most innovative stage involves the cyclization of the isolated intermediate with 6,7-dimethoxy-3,4-dihydroisoquinoline hydrochloride in an aqueous medium. This reaction is facilitated by triethylbenzylammonium chloride (TEBAC), which acts as a phase transfer catalyst to bridge the solubility gap between the organic intermediate and the aqueous salt. Operating at a moderate temperature of 90°C, this system promotes the nucleophilic attack and subsequent ring closure to form the rigid benzo[a]quinolizidine skeleton. The choice of water as the reaction medium is particularly advantageous for impurity control, as many organic byproducts remain insoluble or can be easily washed away, leading to a crude product of high purity that requires minimal purification effort beyond simple recrystallization.
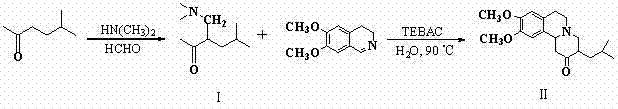
How to Synthesize 9,10-dimethoxy-3-isobutyl-benzo[a]quinolizidine-2-one Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to maximize yield and purity. The process begins with the preparation of the Mannich base, followed by its direct utilization in the cyclization step without extensive derivatization. Operators must ensure precise pH control during the workup of the first step, alkalizing the aqueous layer to pH 8-10 to ensure complete extraction of the free amine intermediate. For the second step, maintaining the reaction temperature at 90°C for approximately 4 hours is crucial to drive the equilibrium towards the desired cyclic product while minimizing thermal degradation. Detailed standard operating procedures regarding reagent addition rates and stirring speeds are essential to replicate the laboratory success on a pilot or commercial scale.
- Perform Mannich reaction of 5-methyl-2-hexanone with formaldehyde and dimethylamine in ethanol under reflux.
- Isolate the intermediate 3-dimethylaminomethyl-5-methyl-hexan-2-one via extraction and column chromatography.
- React the intermediate with 6,7-dimethoxy-3,4-dihydroisoquinoline hydrochloride in water using TEBAC catalyst at 90°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers profound strategic benefits that extend far beyond simple yield improvements. The substitution of exotic, high-cost starting materials with commodity ketones like 5-methyl-2-hexanone drastically reduces the raw material cost basis, insulating the production budget from the volatility often seen in specialized fine chemical markets. Additionally, the elimination of sodium metal and the reduction of flammable organic solvents like toluene significantly lower the costs associated with hazardous material handling, storage, and disposal. This shift not only improves the bottom line but also enhances the overall safety profile of the manufacturing site, reducing insurance premiums and regulatory scrutiny.
- Cost Reduction in Manufacturing: The economic advantage of this process is driven primarily by the replacement of expensive precursors with widely available bulk chemicals. By removing the need for complex protecting group strategies and hazardous reductants, the process intensity is lowered, leading to substantial savings in utility consumption and labor hours. The simplified purification protocol, which relies on recrystallization rather than complex chromatographic separations for the final step, further reduces the consumption of silica gel and elution solvents, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Sourcing reliability is significantly improved as the key raw materials, such as 5-methyl-2-hexanone and formaldehyde, are produced on a massive global scale with stable supply lines. Unlike specialized intermediates that may be subject to single-source bottlenecks or long lead times, these commodity chemicals can be procured from multiple qualified vendors, ensuring business continuity. The robustness of the aqueous reaction system also means that production is less susceptible to disruptions caused by solvent shortages or quality variations in organic reagents.
- Scalability and Environmental Compliance: The transition to water-based chemistry for the critical cyclization step aligns perfectly with modern environmental regulations and sustainability goals. This green synthesis route minimizes the generation of hazardous waste streams, simplifying the permitting process for new manufacturing lines and reducing the burden on wastewater treatment facilities. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, facilitating rapid scale-up from kilogram to tonne quantities with minimal capital expenditure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in CN102260255B, providing a clear understanding of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements based on this superior manufacturing method.
Q: What are the primary cost drivers eliminated in this new synthesis route?
A: The new route eliminates the need for expensive 1-ethoxycarbonyl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline and hazardous sodium metal, replacing them with commodity chemicals like 5-methyl-2-hexanone.
Q: How does the aqueous reaction medium impact environmental compliance?
A: By utilizing water as the solvent for the critical cyclization step instead of toluene, the process drastically reduces volatile organic compound (VOC) emissions and simplifies wastewater treatment protocols.
Q: Is the process scalable for commercial API production?
A: Yes, the mild reaction conditions (90°C) and simplified workup procedures (filtration and recrystallization) are highly conducive to large-scale manufacturing without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9,10-dimethoxy-3-isobutyl-benzo[a]quinolizidine-2-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of reliable supply chains for neurological therapeutics and possess the technical expertise to bring this advanced synthesis route to commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 9,10-dimethoxy-1,3,4,6,7,11b-hexahydro-3-isobutyl-2H-benzo[a]quinolizidine-2-one meets the highest global pharmacopoeia standards.
We invite potential partners to engage with our technical procurement team to discuss how this green synthesis technology can be integrated into your existing supply network. By requesting a Customized Cost-Saving Analysis, you can gain a detailed projection of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance the competitiveness and sustainability of your pharmaceutical portfolio.
