Scalable Aqueous Synthesis of N-Alkylsulfonamides for Global Pharmaceutical Supply Chains
Introduction to Green Sulfonamide Manufacturing
The pharmaceutical and fine chemical industries are currently undergoing a significant paradigm shift towards sustainable manufacturing practices, driven by both regulatory pressure and the economic necessity of reducing waste. Patent CN112047865B represents a critical advancement in this domain, disclosing a novel method for synthesizing N-alkylsulfonamide derivatives directly in an aqueous medium. This technology leverages a specialized water-soluble iridium complex to catalyze the coupling of sulfonamide derivatives with alcohols, effectively replacing hazardous alkylating agents with benign alternatives. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this patent offers a pathway to high-purity compounds with drastically simplified downstream processing. The core innovation lies in the ability to conduct these transformations in pure water, eliminating the need for volatile organic compounds (VOCs) and ensuring that the only byproduct generated is water itself, thereby achieving near-perfect atom economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial synthesis of N-alkylsulfonamides has relied heavily on the reaction between N-alkylamines and sulfonyl chlorides. While chemically effective, this classical approach presents severe logistical and safety challenges that impact the total cost of ownership for chemical manufacturers. Sulfonyl chlorides are notoriously unstable, highly toxic, and corrosive, requiring specialized storage infrastructure and rigorous safety protocols to prevent exposure and degradation. Furthermore, the reaction typically generates stoichiometric amounts of acidic byproducts, such as hydrogen chloride, which necessitate the use of excess base and create significant salt waste streams that complicate purification and wastewater treatment. When conducted in organic solvents like toluene or dichloromethane, these processes also incur high costs related to solvent recovery, fire safety compliance, and environmental discharge limits, making them increasingly untenable for modern green chemistry standards.
The Novel Approach
In stark contrast, the methodology described in CN112047865B utilizes alcohols as the alkylating source, which are inherently safer, more stable, and often less expensive than their halide counterparts. By employing a water-soluble Cp*Ir catalyst, the reaction proceeds efficiently in pure water at moderate temperatures, typically around 130°C, without the need for toxic organic co-solvents. This aqueous protocol not only mitigates the risks associated with handling hazardous reagents but also streamlines the workup procedure; since the catalyst and inorganic salts remain in the aqueous phase, the organic product can often be isolated with minimal contamination. The use of weak bases such as cesium carbonate or potassium carbonate further enhances the mildness of the reaction conditions, preserving sensitive functional groups that might otherwise degrade under the harsh acidic conditions of traditional sulfonyl chloride couplings. This shift from organic to aqueous media represents a fundamental improvement in process safety and environmental compatibility.

Mechanistic Insights into Water-Soluble Iridium Catalysis
The efficacy of this synthetic route is underpinned by the unique properties of the water-soluble Cp*Ir complex, which acts as a robust hydrogen-borrowing catalyst. Mechanistically, the iridium center facilitates the dehydrogenation of the alcohol substrate to generate a reactive aldehyde intermediate in situ, which then condenses with the sulfonamide nitrogen to form a hemiaminal species. Subsequent dehydration and reduction steps, mediated by the metal-hydride species formed during the initial dehydrogenation, yield the final N-alkylated product while regenerating the active catalyst. This borrowing hydrogen mechanism ensures that no external reducing agents are required, as the hydrogen removed from the alcohol is returned to the intermediate, resulting in water as the sole stoichiometric byproduct. The water-solubility of the catalyst, often achieved through specific ligand design such as bipyridine derivatives with polar substituents, ensures that the catalytic cycle occurs homogeneously within the aqueous phase, maximizing contact between the reagents and the active metal center.
From an impurity control perspective, this mechanism offers distinct advantages over nucleophilic substitution pathways. Because the reaction avoids the formation of highly reactive sulfonyl chloride intermediates, the risk of over-alkylation or side reactions with other nucleophilic sites on the molecule is significantly reduced. The mild basic conditions prevent the hydrolysis of the sulfonamide bond, a common degradation pathway in acidic environments. Furthermore, the high selectivity of the iridium catalyst minimizes the formation of regio-isomers or elimination byproducts that are frequently observed when using strong bases or high-temperature thermal methods. For quality control teams, this translates to a cleaner crude reaction profile, reducing the burden on purification columns and ensuring that the final API intermediate meets stringent purity specifications with higher overall yields, often exceeding 80-90% as demonstrated in the patent examples.
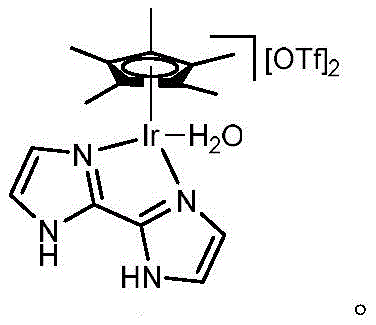
How to Synthesize N-Alkylsulfonamides Efficiently
Implementing this technology requires precise control over reaction parameters to maximize the turnover number of the precious metal catalyst. The standard protocol involves charging a reaction vessel with the sulfonamide derivative, the water-soluble iridium catalyst at a loading of approximately 1.0 mol%, a weak base like cesium carbonate at 0.1 equivalents, and the alcohol substrate at 1.2 equivalents. Water is added as the sole solvent, and the mixture is heated to 130±10°C, typically in a sealed microwave tube or pressurized reactor to maintain the liquid phase of water above its boiling point. After a reaction time of roughly 2 hours, the mixture is cooled, and the solvent is removed via rotary evaporation. The detailed standardized synthesis steps for specific derivatives are outlined below.
- Charge a reaction vessel with sulfonamide derivative, water-soluble iridium complex catalyst (1.0 mol%), weak base (0.1 equiv.), alcohol (1.2 equiv.), and water as the solvent.
- Heat the reaction mixture to 130±10°C and maintain for approximately 2 hours to ensure complete conversion.
- Cool the mixture to room temperature, remove water via rotary evaporation, and purify the target compound using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous catalytic method offers substantial strategic benefits beyond mere technical feasibility. The transition from hazardous sulfonyl chlorides to stable alcohols fundamentally alters the raw material risk profile, ensuring greater supply continuity and reducing the costs associated with hazardous material transport and storage. Since alcohols are commodity chemicals with robust global supply chains, manufacturers are less vulnerable to the price volatility and availability issues that often plague specialized acid chlorides. Additionally, the elimination of organic solvents removes a major cost center related to solvent purchase, recovery, and disposal, directly contributing to a lower cost of goods sold (COGS). The simplified workup procedure, which avoids complex extraction and neutralization steps, further reduces labor and utility consumption, making the process highly attractive for large-scale commercial production.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the drastic simplification of the process workflow and the elimination of expensive waste treatment protocols. By removing the need for toxic sulfonyl chlorides and organic solvents, facilities can avoid the capital expenditure associated with corrosion-resistant reactors and advanced solvent recovery distillation columns. The high atom economy of the reaction means that raw material costs are optimized, as nearly all input mass is converted into the desired product or benign water. Furthermore, the use of a low-loading catalyst (1.0 mol%) ensures that the cost of the precious metal does not erode profit margins, especially when considering the high yields achieved. These factors combine to create a manufacturing process that is significantly more cost-efficient than traditional methods, allowing for competitive pricing in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of widely available and stable starting materials. Alcohols and simple sulfonamides are produced on a massive scale globally, reducing the risk of supply disruptions that can occur with niche reagents. The mild reaction conditions also mean that the process is less sensitive to minor fluctuations in temperature or pressure, leading to more consistent batch-to-batch quality and reducing the rate of failed batches. This reliability is crucial for maintaining just-in-time inventory levels and meeting the strict delivery schedules demanded by pharmaceutical clients. The ability to produce high-purity intermediates with a streamlined process ensures that downstream API synthesis is not delayed by quality issues, thereby strengthening the entire value chain.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of water, which has excellent heat transfer properties and is non-flammable, reducing the safety risks associated with large-scale exothermic reactions. The absence of VOC emissions aligns perfectly with increasingly stringent environmental regulations, minimizing the need for expensive scrubbing systems and permitting delays. Waste streams are predominantly aqueous and contain minimal organic load, simplifying wastewater treatment and reducing environmental fees. This green profile not only future-proofs the manufacturing site against regulatory changes but also enhances the brand reputation of the supplier as a responsible partner in sustainable chemistry, a factor that is becoming increasingly important in vendor selection criteria for multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous iridium-catalyzed synthesis. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: What are the primary advantages of using water as a solvent in this sulfonamide synthesis?
A: Using water eliminates the need for toxic organic solvents like toluene, significantly reducing environmental impact and waste disposal costs while improving operator safety.
Q: How does this method compare to traditional sulfonyl chloride routes in terms of safety?
A: This method avoids highly toxic and corrosive sulfonyl chlorides, utilizing stable alcohols instead, which simplifies storage and handling requirements.
Q: Is the iridium catalyst recoverable in this aqueous process?
A: The process utilizes a water-soluble Cp*Ir complex which facilitates the reaction in the aqueous phase, and while specific recovery rates depend on downstream processing, the homogeneous nature allows for efficient catalytic turnover.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkylsulfonamides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous iridium-catalyzed synthesis described in CN112047865B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green chemistry are realized at an industrial level. Our state-of-the-art facilities are equipped to handle water-based reactions safely and efficiently, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for drug substance manufacturing. We are committed to leveraging this advanced technology to deliver superior quality N-alkylsulfonamides that support your drug development timelines.
We invite you to collaborate with us to explore how this innovative synthetic route can optimize your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating the tangible economic benefits of switching to this aqueous protocol. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Let us help you build a more sustainable and cost-effective supply chain for your critical intermediates.
