Scalable Synthesis of N-Alkyloxy Oxalyl Alanine Esters for Industrial Vitamin B6 Production
The pharmaceutical and fine chemical industries constantly seek robust pathways for synthesizing critical vitamin precursors, particularly those involved in the complex architecture of Vitamin B6. Patent CN102249946B introduces a transformative preparation method for N-alkyloxy oxalyl alanine alkyl ester, a pivotal intermediate in this value chain. This technology leverages a direct reductive amination strategy between pyruvic acid alkyl esters and oxamic acid alkyl esters, bypassing the logistical and safety hurdles associated with traditional acyl chloride chemistry. By enabling the direct formation of the target amine bond under mild acidic or alkaline conditions, this process offers a compelling alternative for manufacturers aiming to optimize their supply chains for high-volume vitamin production. The versatility of the reaction conditions, accommodating various solvents and reducing agents, underscores its potential for widespread industrial adoption.
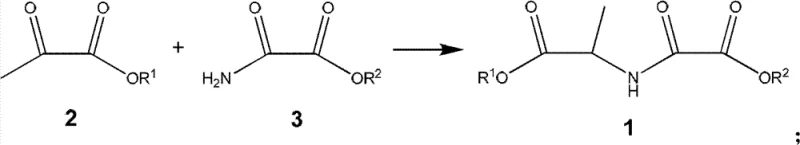
The strategic significance of this patent lies in its ability to streamline the synthesis of N-alkyloxy oxalyl alanine alkyl ester, effectively addressing the bottlenecks of legacy manufacturing protocols. Traditional methods often rely on the coupling of alanine esters with alkoxy oxalyl chlorides or dialkyl oxalates, processes that are fraught with challenges such as the handling of corrosive acid chlorides and the generation of stoichiometric amounts of salt waste. Furthermore, historical literature describes direct esterification routes involving L-alanine and oxalic acid that suffer from excessively long reaction durations, sometimes extending up to one hundred hours, which drastically impacts throughput and energy efficiency. In contrast, the novel approach detailed in the patent utilizes readily available pyruvate and oxamate derivatives, facilitating a rapid condensation-reduction sequence that significantly compresses the production timeline while maintaining exceptional product integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-alkyloxy oxalyl alanine derivatives has been hindered by reliance on reagents that pose significant operational and economic risks. The use of alkoxy oxalyl chloride, while chemically effective, introduces severe safety concerns due to its moisture sensitivity and corrosive nature, necessitating specialized equipment and rigorous containment protocols that drive up capital expenditure. Additionally, alternative routes involving the direct reaction of amino acids with oxalic acid derivatives often require harsh acidic environments and prolonged heating, leading to potential racemization issues and the formation of difficult-to-remove impurities. These legacy processes are characterized by low atom economy and high energy consumption, as the extended reaction times required to drive equilibrium towards the product result in substantial utility costs. For procurement managers, these factors translate into volatile pricing and supply chain fragility, as the dependency on specialized, hazardous reagents limits the pool of qualified suppliers capable of consistent delivery.
The Novel Approach
The methodology disclosed in CN102249946B represents a paradigm shift by employing a reductive amination mechanism that is both operationally simple and chemically elegant. By reacting pyruvic acid alkyl ester with oxamic acid alkyl ester, the process forms an intermediate Schiff base in situ, which is subsequently reduced to the final amine without the need for isolation, although isolation is possible if desired. This one-pot or telescoped approach minimizes unit operations, reducing the physical footprint required for manufacturing and lowering the risk of material loss during transfer. The reaction proceeds efficiently under weakly acidic or alkaline conditions using common organic solvents like dichloroethane, ethanol, or toluene, making it adaptable to existing reactor setups without major retrofitting. The flexibility to use diverse reducing agents, ranging from hydride sources like sodium triacetoxyborohydride to catalytic hydrogenation with palladium on carbon, allows producers to tailor the process to their specific safety and cost profiles, ensuring a robust and scalable supply of this critical intermediate.
Mechanistic Insights into Reductive Amination of Alpha-Keto Esters
The core chemical transformation driving this synthesis is the reductive amination of an alpha-keto ester with an alpha-amino ester derivative, a reaction pathway that offers superior control over stereochemistry and impurity profiles compared to direct acylation. The mechanism initiates with the nucleophilic attack of the amino group from the oxamic acid ester onto the ketone carbonyl of the pyruvic acid ester, facilitated by the presence of a weak acid catalyst such as acetic acid or tosic acid. This condensation step generates an imine or Schiff base intermediate, releasing a molecule of water which can be removed via azeotropic distillation or chemical drying agents to drive the equilibrium forward. The subsequent reduction step involves the addition of hydride equivalents to the electrophilic imine carbon, stereoselectively forming the new C-N bond and yielding the secondary amine structure of the target molecule. This pathway is particularly advantageous because it avoids the high-energy transition states associated with acyl substitution, thereby reducing the formation of side products like di-acylated species or hydrolysis byproducts that often plague traditional methods.
Impurity control in this process is inherently managed through the choice of reducing agent and reaction conditions, which dictates the selectivity of the transformation. For instance, the use of sodium triacetoxyborohydride in dichloroethane provides a mild reducing environment that selectively targets the imine functionality without affecting the ester groups present in the molecule, preserving the structural integrity of the backbone. Alternatively, catalytic hydrogenation offers a clean reduction profile where the only byproduct is the catalyst itself, which can be easily removed via filtration, resulting in a crude product with high GC purity often exceeding 98%. The ability to tune the pH of the reaction medium further enhances selectivity; weakly acidic conditions promote imine formation while preventing the hydrolysis of the sensitive ester linkages, ensuring that the final product stream requires minimal downstream purification. This high level of intrinsic purity is critical for R&D directors who must ensure that downstream Vitamin B6 synthesis is not compromised by trace contaminants that could affect catalyst performance or final drug safety.
How to Synthesize N-Alkyloxy Oxalyl Alanine Alkyl Ester Efficiently
The practical implementation of this synthesis route is designed for seamless integration into standard batch reactor systems, requiring only common laboratory or plant-grade reagents. The process begins with the charging of pyruvic acid alkyl ester and oxamic acid alkyl ester into a reactor equipped with agitation and temperature control, typically under an inert nitrogen atmosphere to prevent oxidation of sensitive intermediates. A solvent system is selected based on solubility and boiling point requirements, with options including halogenated hydrocarbons for low-temperature operations or alcohols for reflux conditions. The addition of a catalytic acid initiates the condensation phase, after which the reducing agent is introduced either in a single portion or via controlled addition to manage exotherms. Following the completion of the reduction, a standard aqueous workup involving pH adjustment and extraction isolates the organic product, which can then be purified by distillation to achieve pharmaceutical-grade specifications.
- Mix pyruvic acid alkyl ester and oxamic acid alkyl ester in a suitable solvent such as dichloroethane or ethanol under nitrogen protection.
- Add a catalytic amount of weak acid like acetic acid or tosic acid to facilitate imine formation, stirring at room temperature or under reflux.
- Introduce a reducing agent such as sodium triacetoxyborohydride or perform catalytic hydrogenation with Pd/C to obtain the target amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this patented methodology offers distinct strategic benefits centered around cost stability and operational reliability. The shift away from hazardous acid chlorides eliminates the need for specialized storage and handling infrastructure, thereby reducing overhead costs associated with safety compliance and waste disposal. Furthermore, the raw materials required—pyruvic acid esters and oxamic acid esters—are commodity chemicals produced on a massive global scale, ensuring a stable and competitive supply base that is less susceptible to the geopolitical or logistical disruptions that often affect niche reagents. The high yields reported in the patent examples, frequently surpassing 90% and approaching quantitative conversion in optimized scenarios, directly correlate to improved material throughput and reduced raw material consumption per kilogram of finished product. This efficiency gain translates into significant margin improvements for manufacturers operating in the highly competitive vitamin market.
- Cost Reduction in Manufacturing: The elimination of expensive activating agents like alkoxy oxalyl chloride removes a major cost driver from the bill of materials, while the use of inexpensive catalysts such as acetic acid or tosic acid further lowers operational expenditures. The ability to run the reaction at ambient temperature or mild reflux reduces energy consumption compared to processes requiring prolonged heating or cryogenic cooling, contributing to a lower overall cost of goods sold. Additionally, the high selectivity of the reductive amination minimizes the formation of byproducts, reducing the burden on purification units and lowering solvent usage rates, which collectively drive down the variable costs associated with production.
- Enhanced Supply Chain Reliability: By utilizing widely available starting materials that are not subject to strict regulatory controls like certain acid chlorides, manufacturers can diversify their supplier base and mitigate the risk of single-source dependency. The robustness of the reaction conditions means that production can be maintained consistently across different facilities without requiring highly specialized operator training or exotic equipment, ensuring business continuity even during periods of workforce fluctuation. The shortened reaction cycle time, dropping from days in older methods to mere hours in this new process, allows for faster inventory turnover and more responsive fulfillment of customer orders, enhancing the agility of the entire supply network.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with various solvent systems and reducing agents that are compatible with large-scale industrial reactors. The reduction in hazardous waste generation, particularly the avoidance of chloride salts and the potential for solvent recycling, aligns with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile not only reduces disposal costs but also enhances the marketability of the final product to downstream customers who are prioritizing environmentally responsible sourcing in their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity for stakeholders evaluating its adoption. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these details is essential for making informed decisions about process validation and capacity planning.
Q: What are the primary advantages of this reductive amination route over traditional acylation methods?
A: This method avoids the use of expensive and hazardous alkoxy oxalyl chlorides. It operates under milder conditions with significantly shorter reaction times compared to direct esterification which can take up to 100 hours, resulting in higher overall yields and lower energy consumption.
Q: Which reducing agents are compatible with this synthesis for large-scale production?
A: The process is highly flexible, supporting chemical reduction using sodium triacetoxyborohydride or sodium borohydride, as well as catalytic hydrogenation using palladium on carbon (Pd/C) or Raney nickel, allowing manufacturers to choose based on available infrastructure and cost constraints.
Q: What purity levels can be achieved with this preparation method?
A: Experimental data indicates that the target compound can be obtained with GC purity exceeding 98%, often reaching above 99% after simple distillation, ensuring the material meets stringent specifications for downstream Vitamin B6 synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Alkyloxy Oxalyl Alanine Alkyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the efficient production of essential vitamins and pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and risk-mitigated. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N-alkyloxy oxalyl alanine alkyl ester meets the exacting standards required for Vitamin B6 synthesis. Our commitment to quality assurance means that our clients can rely on consistent material performance, minimizing batch-to-batch variability and ensuring the integrity of their final drug products.
We invite global partners to engage with us for a Customized Cost-Saving Analysis tailored to their specific production volumes and logistical requirements. By leveraging our expertise in process chemistry and supply chain management, we can help you identify opportunities to optimize your manufacturing costs while securing a stable supply of this vital intermediate. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make data-driven decisions that enhance your competitive position in the marketplace.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
