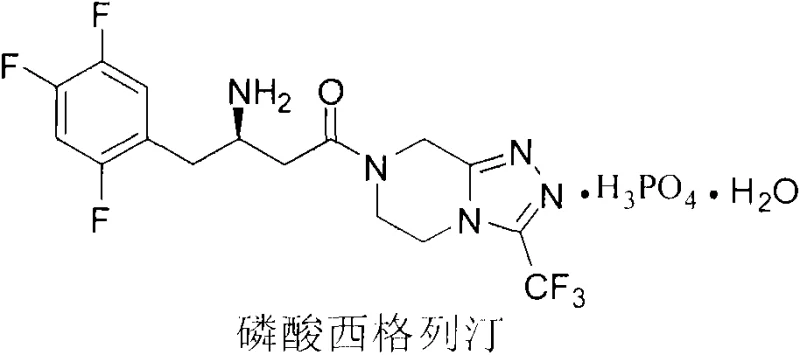
Revolutionizing Sitagliptin Base Production: A Noble-Metal-Free Asymmetric Synthesis Strategy
Revolutionizing Sitagliptin Base Production: A Noble-Metal-Free Asymmetric Synthesis Strategy
The pharmaceutical landscape for Type II diabetes treatment has been dominated by DPP-IV inhibitors, with Sitagliptin phosphate standing as a cornerstone therapy due to its efficacy and safety profile. However, the manufacturing of its core intermediate, Sitagliptin base, has historically relied on complex and capital-intensive methodologies involving precious metal catalysis. Patent CN103387577A introduces a transformative approach to this challenge, detailing an asymmetric synthesis method that utilizes di-p-toluoyl-L-tartaric acid (L-DTTA) as a chiral control source. This innovation represents a significant paradigm shift in pharmaceutical intermediates manufacturing, moving away from the dependency on scarce noble metals towards more accessible organocatalytic systems. By leveraging this technology, manufacturers can achieve high optical purity in a single step while drastically simplifying the operational workflow. The strategic implementation of this route offers a compelling value proposition for global supply chains seeking resilience and cost efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of optically pure Sitagliptin base has been tethered to transition metal-catalyzed asymmetric hydrogenation, a technique pioneered in early patents such as WO2004/085378 and subsequent literature. These conventional routes typically employ rhodium complexes with chiral ligands like Josiphos or palladium on carbon under high-pressure hydrogen atmospheres. While effective, these methods impose severe constraints on production scalability and economic viability. The reliance on rhodium and palladium introduces significant volatility in raw material costs, as these metals are subject to fluctuating global market prices and supply chain bottlenecks. Furthermore, the requirement for high-pressure hydrogenation necessitates specialized reactor infrastructure and rigorous safety protocols, increasing both capital expenditure (CapEx) and operational risk. The removal of trace metal residues from the final API also adds complex purification steps, further eroding profit margins and extending production lead times.
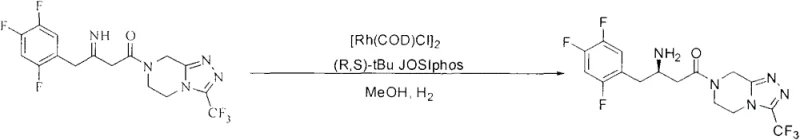
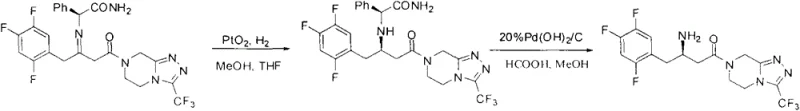
The Novel Approach
In stark contrast to the metal-dependent legacy processes, the methodology disclosed in Patent CN103387577A utilizes a chiral acid-mediated reduction strategy that operates under mild, atmospheric conditions. This novel approach replaces the expensive transition metal catalysts with L-di-p-toluoyl-L-tartaric acid (L-DTTA), a commercially available and cost-effective chiral resolving agent. The reaction proceeds via the reduction of a Sitagliptin imine intermediate using sodium borohydride in the presence of the chiral acid. This shift eliminates the need for high-pressure hydrogen gas, thereby removing a major safety hazard and allowing the reaction to be performed in standard glass-lined or stainless steel reactors without specialized pressure ratings. The result is a streamlined process that not only reduces the direct cost of goods sold (COGS) but also enhances the overall safety profile of the manufacturing facility, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into L-DTTA Mediated Asymmetric Reduction
The core of this technological breakthrough lies in the stereoselective interaction between the chiral controller, L-DTTA, and the reducing agent, sodium borohydride. In this system, L-DTTA likely functions by forming a transient chiral complex with the imine substrate or by modifying the reactivity of the borohydride species to create a chiral environment for hydride transfer. The patent data indicates that temperature control is a critical parameter, with optimal results observed between -10°C and 30°C. At lower temperatures, the conformational flexibility of the transition state is restricted, favoring the formation of one enantiomer over the other with high fidelity. The molar ratio of L-DTTA to sodium borohydride is also pivotal; experimental data suggests that a ratio of approximately 1.8:1 maximizes both yield and optical purity. This precise tuning of reaction conditions allows for the direct generation of Sitagliptin base with optical purity exceeding 99%, effectively bypassing the need for downstream chiral chromatography or multiple recrystallization cycles that are often required in less selective processes.
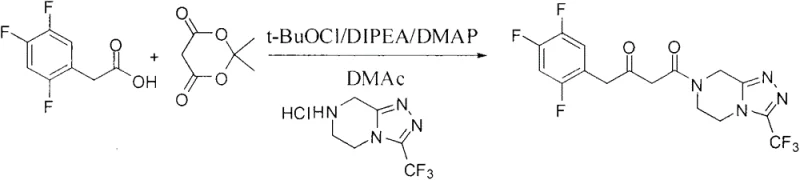
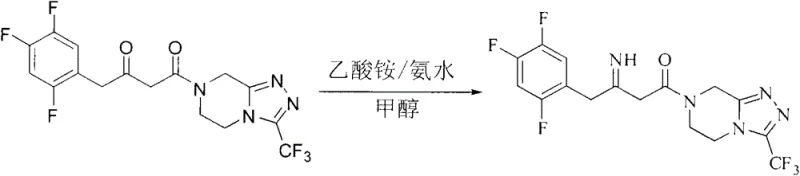
Furthermore, the impurity profile generated by this organic reduction method is significantly cleaner compared to metal-catalyzed routes. In traditional hydrogenation, side reactions such as over-reduction or metal-induced decomposition can generate difficult-to-remove impurities that compromise the quality of the final API. The L-DTTA mediated reduction is highly chemoselective, targeting specifically the C=N bond of the imine while leaving other sensitive functional groups, such as the trifluoromethyl-substituted phenyl ring and the triazolopyrazine core, intact. This selectivity minimizes the formation of by-products, simplifying the workup procedure which involves a straightforward acid-base extraction followed by recrystallization. The ability to achieve high purity (99.6%) and high optical purity (99.7%) directly from the reaction mixture underscores the robustness of this mechanism, providing R&D teams with a reliable pathway to meet stringent regulatory specifications for chiral drugs.
How to Synthesize Sitagliptin Base Efficiently
The synthesis protocol outlined in the patent provides a clear, four-step roadmap for producing high-quality Sitagliptin base, beginning with the construction of the carbon skeleton and culminating in the chiral reduction. The process initiates with the coupling of 2,4,5-trifluorophenylacetic acid to the triazolopyrazine fragment to form the ketone intermediate, followed by condensation with ammonium acetate to generate the crucial imine species. The detailed standardized synthetic steps for the key asymmetric reduction and isolation are provided in the technical guide below, ensuring reproducibility across different manufacturing sites.
- Synthesize the ketone intermediate by reacting 2,4,5-trifluorophenylacetic acid with the triazolopyrazine derivative using pivaloyl chloride activation.
- Convert the ketone to the corresponding imine intermediate using ammonium acetate in methanol under reflux conditions.
- Perform the key asymmetric reduction by adding the imine to a mixture of sodium borohydride and L-DTTA in toluene at controlled low temperatures.
- Isolate the final product through acidic workup, basification, extraction, and recrystallization from toluene to achieve high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this L-DTTA based synthesis route offers tangible strategic advantages that extend beyond simple chemistry. The primary benefit is the decoupling of production costs from the volatile precious metals market. By substituting rhodium and palladium with organic acids and common reducing agents, the direct material costs are significantly reduced, providing a buffer against commodity price spikes. Additionally, the elimination of high-pressure hydrogenation removes the need for specialized infrastructure maintenance and safety certifications, lowering the barrier to entry for contract manufacturing organizations (CMOs) and increasing the pool of potential suppliers. This diversification of the supply base enhances supply chain resilience, reducing lead time for high-purity pharmaceutical intermediates and mitigating the risk of production stoppages due to equipment failure or regulatory audits related to high-pressure operations.
- Cost Reduction in Manufacturing: The economic impact of switching to this noble-metal-free process is profound, driven primarily by the removal of expensive catalyst recovery and recycling loops. In traditional routes, the loss of even small amounts of rhodium represents a significant financial drain, and the infrastructure required to recover these metals adds complexity and cost. By utilizing L-DTTA and sodium borohydride, the process eliminates these heavy metal costs entirely, resulting in substantial cost savings per kilogram of produced intermediate. Furthermore, the simplified workup procedure reduces solvent consumption and energy usage associated with extensive purification steps, contributing to a leaner and more cost-effective manufacturing operation that improves overall margin potential.
- Enhanced Supply Chain Reliability: From a logistics and sourcing perspective, the raw materials required for this synthesis are commodity chemicals with stable and abundant global supplies. Unlike specialized chiral ligands or noble metals which may have limited suppliers and long lead times, L-DTTA and sodium borohydride are widely available from multiple vendors. This abundance ensures consistent availability of starting materials, preventing production delays caused by raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in utility supply or environmental conditions, ensuring a steady and reliable output of product that can meet the demanding schedules of downstream API synthesis and formulation.
- Scalability and Environmental Compliance: The environmental and safety profile of this method aligns perfectly with modern green chemistry principles and increasingly strict regulatory standards. The absence of heavy metals simplifies waste stream management, as there is no need for costly treatment of metal-contaminated effluents. This reduces the environmental footprint of the manufacturing process and lowers the costs associated with waste disposal and regulatory compliance. Moreover, the operation at atmospheric pressure and moderate temperatures makes the process inherently safer and easier to scale from pilot plant to multi-ton commercial production, facilitating rapid capacity expansion to meet market demand without the need for massive capital investment in high-pressure reactor farms.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making.
Q: How does the L-DTTA method compare to Rh-catalyzed hydrogenation in terms of cost?
A: The L-DTTA method significantly reduces costs by eliminating the need for expensive rhodium or palladium catalysts and specialized high-pressure hydrogenation equipment, relying instead on readily available sodium borohydride.
Q: What optical purity can be achieved with this asymmetric synthesis route?
A: By strictly controlling the reaction temperature between -10°C and 30°C and optimizing the molar ratio of L-DTTA to sodium borohydride, optical purity exceeding 99% can be consistently achieved.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable as it avoids dangerous high-pressure operations and uses common solvents like toluene and ethyl acetate, simplifying safety protocols and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Base Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively evaluated the L-DTTA mediated reduction pathway and confirmed its potential for delivering high-quality Sitagliptin base with exceptional optical purity. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for final API production.
We invite forward-thinking procurement leaders to collaborate with us on optimizing their supply chains for diabetes therapeutics. By leveraging our expertise in this novel synthesis method, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate the superior economics and reliability of our supply offering before making any commitment.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
