Advanced Synthesis of 2-Bromo-4,5-Dimethoxyphenylpropionitrile for Commercial API Production
Advanced Synthesis of 2-Bromo-4,5-Dimethoxyphenylpropionitrile for Commercial API Production
The pharmaceutical industry's relentless pursuit of effective cardiovascular treatments has placed significant spotlight on Ivabradine, a groundbreaking anti-ischemic agent approved for clinical use in Europe. At the heart of manufacturing this complex molecule lies the critical intermediate, 2-bromo-4,5-dimethoxyphenylpropionitrile, whose efficient synthesis is paramount for supply chain stability. Patent CN101407474A introduces a transformative preparation method that addresses longstanding inefficiencies in producing this key building block. By leveraging a streamlined three-step sequence starting from readily available 3,4-dimethoxybenzaldehyde, this technology offers a robust pathway that significantly enhances yield while simplifying operational complexity. For R&D directors and procurement specialists alike, understanding the nuances of this patented route is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting the rigorous demands of modern API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-bromo-4,5-dimethoxyphenylpropionitrile has been plagued by cumbersome multi-step processes that erode overall efficiency and drive up manufacturing costs. Traditional literature, such as the work by Kametani, describes a four-step route involving condensation with cyanoacetic acid followed by double bond reduction and decarboxylation, which reportedly achieves a meager total yield of merely 50.1%. Furthermore, alternative approaches attempting to brominate 3,4-dimethoxyphenylpropionitrile directly often suffer from poor regioselectivity, leading to the formation of undesirable by-products where bromine is incorrectly introduced at the alpha-carbon position adjacent to the benzene ring. These side reactions not only complicate downstream purification but also necessitate expensive chromatographic separations that are impractical for large-scale industrial application. Additionally, some legacy methods rely on hazardous reagents like liquid ammonia and sodium amide for subsequent cyclization steps, posing significant safety and environmental compliance challenges for modern chemical plants.
The Novel Approach
In stark contrast, the methodology outlined in patent CN101407474A presents a highly optimized strategy that fundamentally restructures the synthetic logic to maximize atom economy and operational simplicity. This novel approach initiates with the direct bromination of 3,4-dimethoxybenzaldehyde in acetic acid, a reaction that proceeds with excellent selectivity to form 2-bromo-4,5-dimethoxybenzaldehyde crystals in high yield. Following this, a base-catalyzed condensation with acetonitrile efficiently constructs the carbon skeleton, yielding the cinnamonnitrile intermediate without the need for complex protecting group strategies. The final step employs a versatile reduction technique, utilizing either sodium borohydride in a methanol-pyridine system or catalytic hydrogenation with palladium on carbon, to saturate the double bond gently. This strategic reordering of reactions eliminates the decarboxylation step entirely and avoids the formation of alpha-brominated impurities, resulting in a shorter reaction route that is inherently more suitable for commercial scale-up of complex pharmaceutical intermediates.
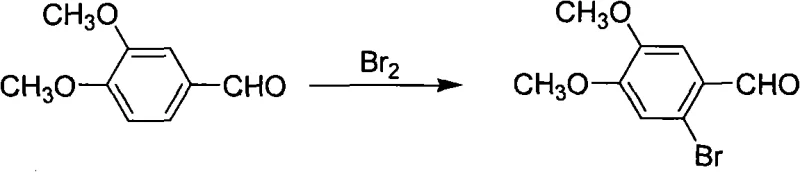
Mechanistic Insights into Electrophilic Aromatic Substitution and Reduction
The success of this synthesis hinges on the precise control of electrophilic aromatic substitution during the initial bromination phase. When 3,4-dimethoxybenzaldehyde is dissolved in acetic acid, the electron-donating methoxy groups activate the aromatic ring, directing the incoming bromine electrophile specifically to the position ortho to one methoxy group and meta to the aldehyde, which is sterically and electronically favored. The use of acetic acid as a solvent is critical here, as it moderates the reactivity of molecular bromine, preventing poly-bromination and ensuring the formation of the mono-brominated product with high purity. As the reaction progresses at temperatures between 0°C and 110°C, the product precipitates as a solid, driving the equilibrium forward and facilitating easy isolation via filtration. This mechanistic clarity allows process chemists to tightly control impurity profiles, ensuring that the resulting aldehyde meets the stringent purity specifications required for downstream pharmaceutical applications.
Following the formation of the aldehyde, the subsequent condensation and reduction steps demonstrate remarkable chemical versatility. The condensation with acetonitrile, catalyzed by strong bases like potassium hydroxide or sodium hydride, proceeds through an anionic mechanism to form the alpha,beta-unsaturated nitrile. The final reduction of the carbon-carbon double bond is particularly noteworthy for its flexibility; the patent describes two distinct pathways. One utilizes sodium borohydride in the presence of a Lewis base like pyridine, which facilitates a 1,4-reduction (conjugate addition) to yield the saturated nitrile without affecting the sensitive nitrile group itself. Alternatively, catalytic hydrogenation using palladium on carbon offers a green chemistry alternative that generates no stoichiometric waste. Both mechanisms effectively suppress the formation of over-reduced amines or other side products, thereby maintaining the integrity of the nitrile functionality which is crucial for the subsequent cyclization into the benzocyclobutane core of Ivabradine.
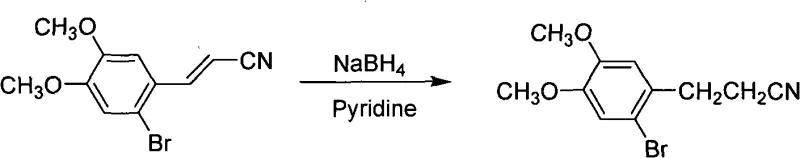
How to Synthesize 2-Bromo-4,5-Dimethoxyphenylpropionitrile Efficiently
Implementing this patented synthesis requires strict adherence to the specified reaction parameters to ensure reproducibility and optimal yield. The process begins with the careful addition of bromine to a solution of the aldehyde in glacial acetic acid, maintaining temperature control to manage exothermicity. Once the brominated aldehyde is isolated, it is subjected to reflux conditions with acetonitrile and a selected base, where reaction time and solvent ratios are adjusted to maximize the conversion to the cinnamonnitrile intermediate. Finally, the reduction step offers operators a choice between chemical reduction with borohydrides or catalytic hydrogenation, allowing facilities to select the method best aligned with their existing infrastructure and safety protocols. For a detailed breakdown of the specific molar ratios, temperature ranges, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- React 3,4-dimethoxybenzaldehyde with bromine in acetic acid at 0-110°C to obtain 2-bromo-4,5-dimethoxybenzaldehyde.
- Condense the resulting aldehyde with acetonitrile in an organic solvent using a basic catalyst like KOH or NaOH under reflux.
- Reduce the double bond of the intermediate cinnamonnitrile using sodium borohydride in methanol/pyridine or via catalytic hydrogenation with Pd/C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound benefits for procurement managers and supply chain heads tasked with optimizing the cost structure of API manufacturing. By eliminating the need for a four-step sequence and replacing it with a concise three-step process, the technology drastically reduces the consumption of solvents, reagents, and energy, leading to substantial cost savings in raw material procurement. The avoidance of hazardous reagents like liquid ammonia and the ability to operate the reduction step at atmospheric pressure using sodium borohydride significantly lowers the barrier for entry regarding equipment investment and safety compliance costs. Furthermore, the high regioselectivity of the bromination step minimizes the generation of difficult-to-separate impurities, which translates to reduced waste disposal costs and higher throughput in the purification units. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The streamlined nature of this process directly impacts the bottom line by removing entire unit operations associated with decarboxylation and complex purification found in older methods. By achieving higher yields at each individual step—reported as high as 86.5% for bromination and over 80% for reduction—the overall material efficiency is significantly improved, meaning less starting material is wasted per kilogram of final product. Additionally, the use of commodity chemicals like acetic acid, methanol, and acetonitrile ensures that raw material costs remain stable and predictable, shielding the project from volatility associated with exotic or specialized reagents. This economic efficiency makes the process highly attractive for generic drug manufacturers seeking to optimize their margin structures.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as 3,4-dimethoxybenzaldehyde ensures that the supply chain is not vulnerable to bottlenecks caused by scarce precursors. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-low temperature cryogenic setups for the main transformations, allows for flexible production scheduling across different manufacturing sites globally. This flexibility is crucial for maintaining continuity of supply in the face of unexpected market disruptions or logistical challenges. Moreover, the simplified workflow reduces the lead time for batch completion, enabling faster response times to fluctuating demand from downstream API producers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is facilitated by the absence of extreme pressure or temperature requirements that often complicate reactor design. The option to use sodium borohydride for reduction avoids the need for high-pressure hydrogenation reactors, which require specialized licensing and safety infrastructure, thus accelerating the timeline for technology transfer. From an environmental standpoint, the reduction in step count inherently lowers the E-factor (mass of waste per mass of product), aligning with increasingly strict global regulations on chemical waste discharge. The ability to recycle solvents like acetic acid and methanol further enhances the sustainability profile of the manufacturing process, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety protocols, and impurity management. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: How does this new synthesis route compare to traditional methods in terms of yield?
A: Traditional methods, such as the four-step process reported by Kametani, often suffer from low overall yields around 50.1%. In contrast, the method described in patent CN101407474A streamlines the process into three efficient steps, significantly improving the overall yield and reducing material waste, which is critical for cost-effective API manufacturing.
Q: What are the safety advantages of the reduction step in this protocol?
A: Conventional routes sometimes require harsh conditions like liquid ammonia and sodium amide for cyclization precursors, or high-pressure hydrogenation which poses safety risks. This patent offers a flexible reduction strategy using sodium borohydride in methanol/pyridine at atmospheric pressure, or mild catalytic hydrogenation, greatly enhancing operational safety and ease of scale-up.
Q: Does this method avoid specific impurities common in bromination reactions?
A: Yes. Previous methods involving the bromination of 3,4-dimethoxyphenylpropionitrile often resulted in unwanted by-products where bromine was introduced at the alpha-carbon position. By performing the bromination early on the aldehyde substrate (3,4-dimethoxybenzaldehyde) in acetic acid, this route ensures high regioselectivity for the aromatic ring, minimizing difficult-to-remove side products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Bromo-4,5-Dimethoxyphenylpropionitrile Supplier
As the global demand for cardiovascular therapeutics continues to rise, securing a partner with deep technical expertise in intermediate synthesis is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced process chemistry to deliver high-quality intermediates that meet the rigorous standards of the international pharmaceutical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs whether you are in the clinical trial phase or full-scale commercial manufacturing. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-bromo-4,5-dimethoxyphenylpropionitrile we supply is free from the critical impurities that can derail downstream synthesis.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your project's economics and timeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can become a cornerstone of your supply chain strategy.
