Advanced Synthesis of R-L-Prolinol Derivatives for Commercial Scale-Up of Complex Chiral Catalysts
Advanced Synthesis of R-L-Prolinol Derivatives for Commercial Scale-Up of Complex Chiral Catalysts
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access high-purity chiral building blocks that can serve as effective organocatalysts. Patent CN103450074A introduces a groundbreaking approach to synthesizing an R-L-prolinol chiral organic small molecule compound featuring a rigid cyclopropane structure. This innovation addresses critical limitations in existing organocatalyst design by enhancing steric hindrance and rigidity without compromising catalytic activity. By integrating a cyclopropane ring into the pyrrole scaffold of prolinol, the resulting compound offers superior stereocontrol in asymmetric reactions. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering next-generation chiral auxiliaries with improved performance metrics.
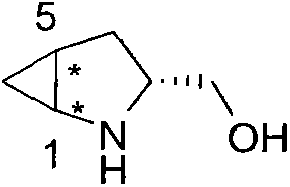
The core breakthrough lies in the ability to produce enantiomerically pure products without the need for subsequent chiral preparation or resolution steps, which traditionally inflate costs and extend lead times. The patent details a streamlined three-step synthesis that begins with a highly selective cyclopropanation, followed by reduction and deprotection. This methodology not only simplifies the operational workflow but also drastically reduces the environmental footprint by avoiding toxic heavy metal reagents often associated with older cyclopropanation techniques. As we delve deeper into the technical specifics, it becomes evident that this process is ideally suited for the commercial scale-up of complex polymer additives and pharmaceutical intermediates where purity and consistency are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of proline derivatives with cyclopropane structures has been plagued by significant technical and economic hurdles. Prior art, such as the methods developed by H. Stephen's group, relied heavily on toxic and expensive cyclization reagents like trimethyltin methylide (Me3SnCH2). These reagents not only pose severe safety and environmental risks but also result in poor diastereoselectivity, often yielding enantiomeric mixtures with ratios as low as 1:4. Such low selectivity necessitates extensive downstream purification and chiral resolution processes, which are resource-intensive and detrimental to overall process efficiency. Furthermore, the harsh reaction conditions required for these traditional routes often lead to lower yields of the desired olefin products, creating bottlenecks in production capacity and driving up the cost of goods sold for the final chiral catalyst.
The Novel Approach
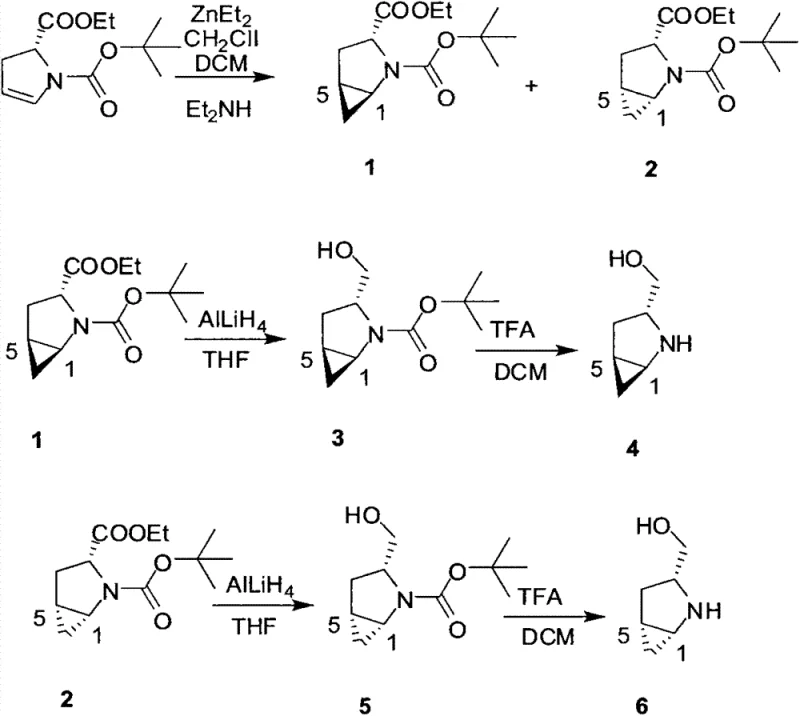
In stark contrast, the novel approach outlined in CN103450074A leverages a modified Simmons-Smith cyclopropane reaction that fundamentally transforms the selectivity profile of the synthesis. By utilizing diethylzinc (ZnEt2) and chloroiodomethane (CH2ClI) under controlled low-temperature conditions, the process achieves a remarkable diastereomeric ratio exceeding 25:1. This dramatic improvement in selectivity effectively eliminates the need for costly and time-consuming chiral separation steps, directly translating to substantial cost savings in chiral catalyst manufacturing. The route is designed for operational simplicity, utilizing standard laboratory equipment and readily available reagents, which enhances its viability for industrial adoption. This shift from toxic tin chemistry to zinc-based carbenoid chemistry exemplifies a move towards greener, more sustainable chemical manufacturing practices.
Mechanistic Insights into Simmons-Smith Cyclopropanation
The success of this synthesis hinges on the precise mechanistic control of the Simmons-Smith reaction, which involves the generation of a zinc carbenoid species that adds across the double bond of the pyrrole ring. The existing chirality at the C-2 position of the starting material directs the facial selectivity of the cyclopropanation, ensuring that the cyclopropane ring forms with the desired stereochemistry. The use of specific amine additives during the workup phase further enhances the enantioselectivity by stabilizing the transition state or facilitating the removal of unwanted isomers. This level of mechanistic understanding allows for fine-tuning of reaction parameters, such as temperature (-20°C to -15°C) and reagent stoichiometry, to maximize the yield of the target (1R,3R,5R) isomer. For technical teams, understanding these nuances is crucial for replicating the high purity standards required for GMP-grade intermediate production.
Following the cyclopropanation, the ester moiety is reduced to a primary alcohol using lithium aluminum hydride (LiAlH4), a transformation that proceeds with high fidelity under mild conditions. The final step involves the removal of the tert-butoxycarbonyl (Boc) protecting group using trifluoroacetic acid (TFA), revealing the free amine necessary for catalytic activity. This sequence ensures that the rigid cyclopropane scaffold is preserved throughout the synthesis, maintaining the structural integrity required for high-performance organocatalysis. The resulting compound exhibits enhanced steric bulk compared to unmodified prolinol, which is critical for achieving high enantioselectivity in downstream applications like asymmetric Michael additions.
How to Synthesize R-L-Prolinol Efficiently
The synthesis of this high-value chiral intermediate is structured around three distinct chemical transformations that can be easily scaled from gram to kilogram quantities. The process begins with the cyclopropanation of the protected pyrrole ester, followed by a reduction step to install the hydroxymethyl group, and concludes with acidic deprotection. Each step has been optimized to minimize byproduct formation and maximize isolation yields, ensuring a robust and reproducible workflow. Detailed standardized operating procedures for each stage, including specific quenching protocols and purification methods, are essential for maintaining batch-to-batch consistency. For a comprehensive guide on executing these steps with precision, please refer to the technical breakdown below.
- Perform Simmons-Smith cyclopropane reaction on (R)-1-N-tert-butoxycarbonyl-2,3-dihydro-2-pyrrole ethyl formate using ZnEt2 and CH2ClI.
- Conduct ester reduction reaction on the cyclopropane intermediate using LiAlH4 in THF to obtain the alcohol derivative.
- Execute amino-removal protecting action using trifluoroacetic acid (TFA) to yield the final R-L-prolinol chiral compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers compelling advantages for procurement managers and supply chain heads looking to optimize their sourcing strategies for chiral materials. The elimination of toxic tin reagents not only reduces raw material costs but also simplifies waste disposal compliance, leading to significant operational expenditure reductions. By removing the need for chiral resolution, the process shortens the overall production cycle time, thereby enhancing supply chain reliability and reducing the risk of stockouts for critical catalyst inventories. Furthermore, the use of common reagents like zinc and iodomethane ensures a stable supply base, mitigating the risks associated with sourcing specialized or regulated chemicals.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the exceptional diastereoselectivity achieved during the initial cyclopropanation step. By producing the desired isomer with a ratio greater than 25:1, the need for expensive chromatographic separation or recrystallization of enantiomers is effectively removed. This streamlining of the purification workflow significantly lowers solvent consumption and labor hours, resulting in a leaner manufacturing cost structure. Additionally, the replacement of costly tin-based reagents with more economical zinc reagents further contributes to the overall reduction in direct material costs, making the final catalyst more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available and stable chemical feedstocks. Unlike specialized organometallic reagents that may have long lead times or limited suppliers, diethylzinc and chloroiodomethane are commodity chemicals with robust global supply networks. This accessibility ensures that production schedules can be maintained without interruption due to raw material shortages. Moreover, the simplified process flow reduces the number of unit operations required, decreasing the potential for equipment downtime and logistical bottlenecks, thus guaranteeing a more consistent and reliable delivery of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind from the outset. The reaction conditions are mild and do not require extreme pressures or temperatures, facilitating safe scale-up in standard stainless steel reactors. From an environmental standpoint, the avoidance of heavy metal contaminants like tin aligns with increasingly stringent global regulations on chemical manufacturing emissions. This compliance reduces the burden on wastewater treatment facilities and minimizes the environmental liability associated with production, supporting corporate sustainability goals and enhancing the brand reputation of manufacturers who adopt this green chemistry approach.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical specifications and commercial viability of this technology, we have compiled a set of frequently asked questions based on the patent data. These answers provide clarity on the performance metrics, safety profile, and application scope of the new R-L-prolinol derivative. Understanding these details is vital for stakeholders evaluating the integration of this catalyst into their existing synthetic workflows. The following section clarifies key aspects of the technology to support informed decision-making.
Q: How does this new synthesis method improve upon conventional prolinol catalyst production?
A: Unlike conventional methods that often suffer from low diastereoselectivity (e.g., 1:4 ratio) and require toxic tin reagents, this patent utilizes a modified Simmons-Smith reaction achieving a molar ratio greater than 25:1, eliminating the need for complex chiral resolution.
Q: What are the key advantages for supply chain stability when adopting this route?
A: The process avoids expensive and hazardous cyclization reagents like Me3SnCH2, relying instead on more accessible zinc and iodomethane derivatives. This simplifies procurement and reduces regulatory hurdles associated with heavy metal waste.
Q: Can this R-L-prolinol derivative be used for large-scale asymmetric catalysis?
A: Yes, the compound demonstrates high catalytic activity and enantioselectivity (up to 92% ee) in asymmetric Michael addition reactions, making it suitable for scaling up complex pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-L-Prolinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced synthesis technology for the production of high-performance chiral catalysts. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of R-L-prolinol derivative meets the highest standards required for sensitive asymmetric catalysis applications. We are committed to delivering quality and consistency that empowers your R&D and production teams.
We invite you to collaborate with us to leverage this innovative route for your specific chemical needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this process can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage through superior chiral intermediate supply.
