Advanced Cage-Shaped Supramolecular Catalysts for Efficient Sulfoxide Manufacturing
Advanced Cage-Shaped Supramolecular Catalysts for Efficient Sulfoxide Manufacturing
The landscape of fine chemical synthesis is undergoing a significant transformation driven by the need for more sustainable and efficient catalytic systems. Patent CN112774733B introduces a groundbreaking cage-shaped supramolecular catalyst designed specifically for the selective oxidation of thioethers to sulfoxides and sulfones. This technology addresses critical bottlenecks in the production of pharmaceutical intermediates by utilizing a robust covalent cage structure derived from tridentate aldehydes and 1,2-cyclohexanediamine. The resulting material, when complexed with transition metals like Nickel(II), exhibits exceptional catalytic performance, achieving conversion rates exceeding 95% under remarkably mild conditions. For R&D directors and process engineers, this represents a pivotal shift away from harsh, wasteful traditional methods toward a more controlled, green chemistry approach that aligns with modern regulatory and environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfoxides and sulfones has relied heavily on stoichiometric oxidants or homogeneous transition metal catalysts that suffer from significant drawbacks in industrial settings. Traditional methods often involve the use of expensive and toxic heavy metals that are difficult to remove from the final product, necessitating complex purification steps that drive up costs and extend lead times. Furthermore, many existing catalytic systems, such as certain chiral Salen-Ti complexes or scandium-based catalysts referenced in prior art, exhibit limited stability and low conversion rates, often requiring prolonged reaction times and large volumes of organic solvents. These inefficiencies not only increase the environmental footprint through excessive waste generation but also compromise the economic viability of scaling these processes for commercial production of high-purity pharmaceutical intermediates.
The Novel Approach
The innovative strategy outlined in CN112774733B overcomes these challenges by employing a unique cage-like supramolecular architecture that encapsulates the active metal centers within a rigid organic framework. This structural design prevents the aggregation of metal ions and protects the active sites from deactivation, thereby enhancing both the stability and longevity of the catalyst. By utilizing a "tridentate aldehyde" precursor to form a covalent organic cage, the technology ensures a well-defined coordination environment that promotes high selectivity for the desired sulfoxide products while minimizing over-oxidation to sulfones. This approach allows for the use of greener solvents and milder temperatures, drastically simplifying the downstream processing requirements and offering a clear pathway for cost reduction in pharmaceutical intermediate manufacturing without sacrificing yield or purity.
Mechanistic Insights into Ni(II)-SNC Catalyzed Oxidation
The core of this technological advancement lies in the precise molecular engineering of the Ni(II)-SNC catalyst, where the nickel centers are securely anchored within the Salen-type ligand cage. This supramolecular confinement effect creates a microenvironment that facilitates the activation of oxidants such as iodosobenzene diacetate or hydrogen peroxide, enabling efficient oxygen transfer to the sulfur atom of the substrate. The rigid cage structure restricts the conformational freedom of the reacting species, which is crucial for achieving the high regioselectivity and chemoselectivity observed in the experimental data. Unlike free metal salts that may leach into the solution or form inactive clusters, the caged nickel species maintain their integrity throughout the catalytic cycle, ensuring consistent performance across multiple batches and providing a reliable foundation for reproducible process chemistry.
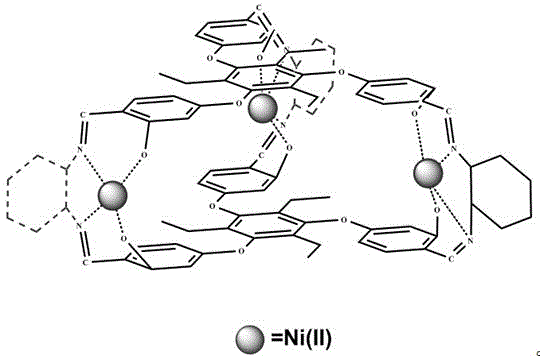
Furthermore, the impurity profile of the reaction is significantly improved due to the steric hindrance provided by the ethyl groups on the benzene rings of the cage ligand. This steric bulk effectively shields the active sites from bulky impurities or side-reactants that could otherwise lead to the formation of unwanted by-products. The mechanism involves a coordinated interaction between the axial ligands, such as 4-methylpyridine or imidazole derivatives, and the metal center, which fine-tunes the electronic properties of the catalyst to match the specific oxidation potential of various thioether substrates. This level of control is essential for R&D teams aiming to synthesize complex API intermediates where even trace impurities can impact the safety and efficacy of the final drug product, making this catalyst a superior choice for high-specification applications.
How to Synthesize Ni(II)-SNC Efficiently
The preparation of this advanced catalyst follows a streamlined two-step protocol that is highly amenable to scale-up. First, the covalent cage ligand is synthesized via a condensation reaction between the tridentate aldehyde and 1,2-cyclohexanediamine in a suitable organic solvent, followed by isolation and drying. Subsequently, the ligand is metallated with a nickel salt under inert atmosphere to yield the final active catalyst. This straightforward synthetic route minimizes the need for exotic reagents or specialized equipment, facilitating rapid technology transfer from the laboratory to pilot plant operations. For detailed procedural specifics regarding stoichiometry, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below.
- Condense 2,4,6-triethyl-1,3,5-tris(4-oxomethyl-1-formyl)benzene with 1,2-cyclohexanediamine in organic solvent at 30-70°C to form the Salen ligand cage.
- React the isolated Salen ligand with a transition metal salt (e.g., Nickel acetate) under inert gas protection with heating reflux for 10-25 hours.
- Purify the resulting M-SNC catalyst by washing with water and vacuum drying, ready for use in sulfide oxidation reactions.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this cage-shaped supramolecular catalyst offers transformative benefits that directly impact the bottom line and operational resilience. The ability to recycle the catalyst multiple times without significant degradation in activity means that the effective cost per kilogram of product is substantially lowered, as the expensive metal component does not need to be replenished for every batch. Additionally, the use of milder reaction conditions reduces energy consumption and lowers the safety risks associated with high-temperature or high-pressure operations, leading to reduced insurance and compliance costs. The compatibility with greener solvents also simplifies waste management protocols, allowing facilities to minimize their environmental liability and adhere to increasingly stringent global regulations regarding chemical discharge and solvent usage.
- Cost Reduction in Manufacturing: The elimination of expensive noble metals and the ability to recover and reuse the catalyst significantly lower the raw material costs associated with sulfide oxidation processes. By avoiding the need for complex metal scavenging steps typically required for homogeneous catalysts, manufacturers can streamline their purification workflows, reducing both solvent usage and processing time. This efficiency translates into direct savings on operational expenditures, making the production of high-value sulfoxide intermediates more economically competitive in the global market.
- Enhanced Supply Chain Reliability: The robustness of the Ni(II)-SNC catalyst against air and moisture ensures consistent performance even under less-than-ideal storage or handling conditions, reducing the risk of batch failures due to catalyst degradation. Since the precursor materials are commercially available and the synthesis does not rely on scarce or geopolitically sensitive resources, supply chain continuity is greatly enhanced. This reliability allows procurement managers to forecast production schedules with greater confidence, minimizing the risk of delays that could disrupt the delivery of critical pharmaceutical ingredients to downstream customers.
- Scalability and Environmental Compliance: The synthetic pathway described in the patent is inherently scalable, utilizing standard reactor configurations and common chemical reagents that are readily accessible in bulk quantities. The reduction in hazardous waste generation, achieved through higher selectivity and solvent recycling, aligns perfectly with corporate sustainability goals and regulatory requirements. This environmental advantage not only future-proofs the manufacturing process against tightening regulations but also enhances the brand reputation of the supplier as a responsible partner in the green chemistry initiative.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating its suitability for your specific production needs. Understanding these details is crucial for making informed decisions about process integration and vendor selection.
Q: What are the key advantages of the Ni(II)-SNC catalyst over traditional methods?
A: The Ni(II)-SNC catalyst offers superior stability, insensitivity to air and moisture, and high selectivity (>95%) for sulfoxides. Unlike traditional homogeneous catalysts, it can be recycled multiple times without significant loss of activity.
Q: What reaction conditions are required for sulfide oxidation using this catalyst?
A: The reaction proceeds under mild conditions, typically between 0°C and 80°C, using green solvents such as methanol or acetone. It utilizes common oxidants like iodosobenzene diacetate or hydrogen peroxide.
Q: Is this catalyst suitable for large-scale industrial production?
A: Yes, the catalyst demonstrates excellent potential for industrial application due to its high conversion rates, recyclability, and the use of commercially available raw materials, significantly reducing operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ni(II)-SNC Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge catalytic technologies to maintain a competitive edge in the fine chemical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging innovations like the cage-shaped supramolecular catalyst, we empower our partners to achieve superior process economics and environmental performance.
We invite you to engage with our technical procurement team to discuss how this advanced oxidation technology can be integrated into your supply chain. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible value this catalyst can bring to your manufacturing operations.
