Revolutionizing Fluorinated Intermediate Production via Electrochemical Defluorinative Carboxylation
The landscape of fluorinated organic synthesis is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective manufacturing protocols. Patent CN111909031B introduces a groundbreaking electrochemical strategy for the synthesis of geminal difluoroalkenyl-substituted alkyl carboxylic acids and (Z)-alpha-fluoroacrylic acid derivatives. This technology leverages carbon dioxide as a C1 building block, directly addressing the challenges associated with the inertness of CO2 and the strength of carbon-fluorine bonds. For R&D directors and procurement specialists in the fine chemical sector, this represents a pivotal shift away from reliance on precious metal catalysts towards a more atom-economical and environmentally benign electrochemical paradigm. The ability to construct complex fluorinated scaffolds directly from simple olefinic precursors under mild conditions opens new avenues for the rapid development of bioactive molecules used in pharmaceuticals and agrochemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of fluorinated carboxylic acids has relied heavily on transition metal catalysis, often involving copper or palladium complexes, coupled with stoichiometric amounts of reducing agents such as organoboron compounds or silanes. These conventional pathways present substantial drawbacks for large-scale manufacturing, including the high cost of noble metals, the generation of significant metal-containing waste streams, and the requirement for rigorous purification steps to meet stringent pharmaceutical impurity specifications. Furthermore, many thermal methods necessitate elevated temperatures and pressures to overcome the kinetic barrier of CO2 insertion, which can lead to poor functional group tolerance and safety concerns in industrial reactors. The reliance on external chemical reductants not only increases the raw material cost but also complicates the reaction mass balance, making the process less attractive for green chemistry initiatives.
The Novel Approach
In stark contrast, the electrochemical defluorinative carboxylation described in the patent utilizes electrons as clean reagents to drive the transformation, effectively replacing chemical reductants. By employing an undivided cell configuration with inexpensive electrode materials like platinum or nickel, the process achieves the activation of strong C-F bonds and the subsequent fixation of CO2 under remarkably mild conditions, typically at room temperature and atmospheric pressure. This methodology not only simplifies the operational setup but also enhances the safety profile by avoiding high-pressure reactors and pyrophoric reagents. The electrochemical system demonstrates excellent compatibility with a wide range of functional groups, including esters, halides, and ethers, which is critical for the late-stage functionalization of complex drug candidates. This shift to electro-organic synthesis aligns perfectly with modern sustainability goals while offering a robust platform for cost reduction in pharmaceutical intermediate manufacturing.
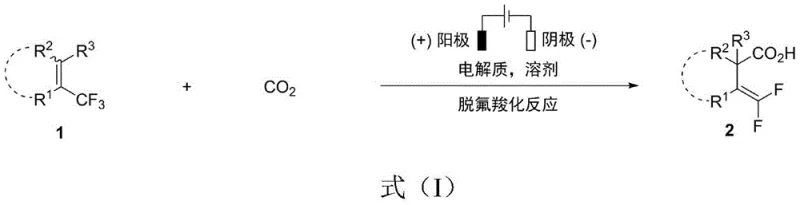
Mechanistic Insights into Electrochemical Defluorinative Carboxylation
The core of this innovation lies in the unique mechanism of electrochemical activation, which differs fundamentally from thermal radical processes. At the cathode, the reduction of the fluoro-olefin substrate generates a radical anion intermediate, triggering the elimination of a fluoride ion to form a carbon-centered radical. This highly reactive species then undergoes rapid addition to carbon dioxide, forming a carboxylate radical anion. Subsequent electron transfer and protonation steps yield the final carboxylic acid product. The precise control of potential and current density allows for the selective activation of the C-F bond without affecting other sensitive moieties within the molecule. This level of control is instrumental in achieving high regioselectivity, producing specific isomers that are otherwise inaccessible. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for different substrate classes, ensuring consistent quality and yield across diverse chemical spaces.
Furthermore, the choice of electrolyte and solvent plays a critical role in stabilizing the reactive intermediates and facilitating ion transport within the cell. The patent highlights the effectiveness of quaternary ammonium salts like nBu4NClO4 in polar aprotic solvents such as DMF, which provide the necessary conductivity and solvation power. The absence of transition metals eliminates the risk of metal leaching, a common concern in API synthesis that often requires additional scavenging steps. By relying on electricity rather than chemical oxidants or reductants, the process minimizes the formation of side products derived from reagent decomposition. This mechanistic clarity provides a solid foundation for scaling up the reaction, as the parameters can be finely tuned to maintain efficiency even as the reactor volume increases, ensuring high-purity pharmaceutical intermediates are produced consistently.
How to Synthesize Geminal Difluoroalkenyl Alkyl Carboxylic Acids Efficiently
The practical implementation of this electrochemical protocol is straightforward, requiring standard laboratory equipment that can be easily adapted for pilot plant operations. The procedure involves dissolving the alpha-trifluoromethyl olefin substrate and the supporting electrolyte in anhydrous DMF, followed by the introduction of carbon dioxide gas. The reaction is driven by applying a constant current through platinum electrodes, maintaining the system at room temperature to preserve sensitive functional groups. After the electrolysis is complete, a simple aqueous workup involving acidification and extraction isolates the crude product, which can be further purified by column chromatography if necessary. This streamlined workflow reduces the number of unit operations compared to traditional multi-step syntheses. For detailed operational parameters and specific substrate examples, please refer to the standardized synthesis guide below.
- Prepare the electrolytic cell by adding supporting electrolyte (e.g., nBu4NClO4), solvent (DMF), and the alpha-trifluoromethyl olefin substrate into a reaction vessel equipped with platinum electrodes.
- Saturate the solution with carbon dioxide gas at atmospheric pressure or under slight pressurization, ensuring continuous bubbling throughout the reaction process.
- Apply a constant current (typically 8 mA) at room temperature for approximately 7 hours, followed by acidic workup, extraction, and purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical technology offers compelling advantages for supply chain stability and cost management. By removing the dependency on volatile precious metal markets and expensive stoichiometric reagents, manufacturers can achieve significant cost reduction in fine chemical manufacturing. The simplified process flow reduces energy consumption associated with heating and cooling, contributing to a lower overall carbon footprint. Additionally, the use of CO2 as a feedstock adds value to a greenhouse gas, aligning production with corporate sustainability targets. The robustness of the method across various substrates ensures a reliable supply of diverse intermediates, mitigating the risk of production delays caused by reagent shortages. This reliability is crucial for maintaining continuous manufacturing lines in the competitive pharmaceutical and agrochemical sectors.
- Cost Reduction in Manufacturing: The elimination of noble metal catalysts and stoichiometric reducing agents drastically lowers the bill of materials. Since electrons serve as the primary reagent, the ongoing operational costs are primarily driven by electricity, which is generally more stable and predictable than the pricing of specialized chemical reagents. This structural change in cost drivers allows for better long-term financial planning and margin protection.
- Enhanced Supply Chain Reliability: The starting materials, such as alpha-trifluoromethyl olefins and CO2, are readily available commodity chemicals, reducing the risk of supply bottlenecks. The mild reaction conditions also mean that the process can be implemented in a wider range of facilities without requiring specialized high-pressure infrastructure, thereby expanding the potential supplier base and enhancing supply chain resilience.
- Scalability and Environmental Compliance: Electrochemical reactions are inherently scalable by increasing electrode surface area or using flow cell reactors, facilitating the commercial scale-up of complex fluorinated intermediates. The reduction in hazardous waste generation simplifies regulatory compliance and waste disposal logistics, making the process more environmentally sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of fluorinated carboxylic acids. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of this electrochemical method over traditional metal catalysis?
A: This method eliminates the need for expensive noble metal catalysts and stoichiometric reducing agents like borates, significantly reducing raw material costs and simplifying downstream purification processes.
Q: Is this process scalable for industrial manufacturing of pharmaceutical intermediates?
A: Yes, the use of undivided cells and standard electrolytes suggests high scalability. The mild reaction conditions (room temperature, ambient pressure CO2) facilitate easier transition from laboratory to commercial production scales.
Q: What is the regioselectivity profile of this defluorinative carboxylation?
A: The electrochemical approach offers unique regioselectivity distinct from existing metal-catalyzed methods, allowing access to specific geminal difluoroalkenyl isomers that are difficult to obtain via conventional thermal pathways.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Geminal Difluoroalkenyl Alkyl Carboxylic Acid Supplier
As the demand for fluorinated building blocks continues to surge in the life sciences sector, partnering with an experienced CDMO is essential for successful project execution. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped to handle electrochemical processes safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality. We understand the critical nature of supply continuity and are committed to delivering high-performance intermediates that meet your exact requirements.
We invite you to collaborate with our technical team to explore how this innovative electrochemical route can optimize your specific synthesis challenges. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and route feasibility assessments to support your R&D and supply chain strategies, ensuring you stay ahead in the competitive market.
