Revolutionizing Cyclobutane Synthesis: A Gold Carbene Template Strategy for Commercial Scale-Up
Revolutionizing Cyclobutane Synthesis: A Gold Carbene Template Strategy for Commercial Scale-Up
The landscape of organic synthesis is constantly evolving, driven by the need for more efficient, selective, and scalable methodologies to access complex molecular architectures. Among these, cyclobutane derivatives hold a position of immense strategic value, serving as critical scaffolds in the development of bioactive pharmaceutical ingredients, agrochemicals, and advanced functional materials. However, the construction of the four-membered ring has historically been plagued by thermodynamic instability and kinetic barriers associated with ring strain. A groundbreaking approach detailed in patent CN109111392B offers a transformative solution to these challenges by leveraging the unique properties of gold carbene chemistry. This patent discloses a novel synthetic method that utilizes a rectangular gold carbene compound as a self-assembling template to facilitate the [2+2] photochemical cycloaddition reaction. By integrating the template directly into the reactant structure, this innovation bypasses the stochastic limitations of traditional solution-phase photochemistry, offering a pathway to high-purity cyclobutane derivatives with exceptional conversion efficiency.
For research and development directors overseeing complex molecule synthesis, the implications of this technology are profound. The method described in CN109111392B moves beyond the trial-and-error nature of conventional photochemical setups. Instead, it employs a rational design where the metal-ligand coordination geometry dictates the outcome of the reaction. The use of binuclear gold carbene complexes allows for the precise pre-organization of olefinic substrates, ensuring that the reactive double bonds are held in the optimal parallel orientation required by Schmidt's rule. This level of control not only enhances the yield but also drastically reduces the formation of regioisomers and byproducts, addressing one of the most persistent pain points in the manufacturing of strained ring systems. As we delve deeper into the technical specifics, it becomes clear that this represents a significant leap forward in the reliable production of high-value chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclobutane rings via [2+2] photocycloaddition has been fraught with significant technical hurdles that impede its widespread adoption in industrial settings. The fundamental challenge lies in the probabilistic nature of molecular collisions in solution. According to Schmidt's rule, for a [2+2] cycloaddition to occur successfully, the two reacting carbon-carbon double bonds must be arranged in a parallel fashion with an inter-planar distance of less than 4.2 angstroms. In a standard liquid-phase reaction without constraints, molecules move in a disorderly, Brownian motion, making the probability of two specific olefins meeting in this precise geometric alignment exceedingly low. Consequently, traditional methods often suffer from poor conversion rates, requiring prolonged irradiation times that can lead to substrate degradation or the formation of complex mixtures of oligomers and polymers.
To mitigate these issues, previous strategies have relied heavily on the use of external host matrices or template agents, such as cyclodextrins, cucurbiturils, or metal-organic frameworks (MOFs). While these hosts can provide the necessary confined space to align the reactants, they introduce their own set of complications. The inclusion of these bulky auxiliary substances adds significant complexity to the downstream processing, as the product must be separated from the host matrix, often requiring extensive chromatography or recrystallization steps. Furthermore, the loading capacity of these hosts is frequently limited, and the diffusion of reactants into the host cavities can be slow, creating bottlenecks in throughput. For a procurement manager focused on cost reduction in fine chemical manufacturing, these inefficiencies translate directly into higher operational expenditures and longer lead times, rendering many photochemical routes economically unviable for large-scale production.
The Novel Approach
The methodology presented in patent CN109111392B fundamentally reimagines the templating strategy by internalizing the template within the reactant structure itself. Rather than relying on an external host to capture and align free-floating olefins, this approach utilizes a binuclear gold carbene complex that spontaneously assembles with the olefin ligands to form a discrete, rectangular supramolecular structure. In this "gold carbene template" system, the rigid coordination geometry of the gold centers acts as a molecular ruler, locking the olefinic double bonds into the exact parallel arrangement required for photocyclization. This pre-organization eliminates the entropic penalty associated with bringing two independent molecules together in the correct orientation, thereby accelerating the reaction kinetics significantly.
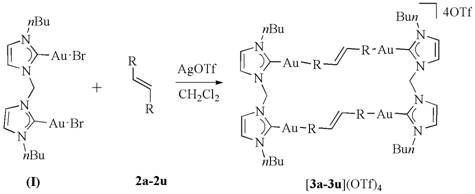
This internal templating mechanism offers a distinct advantage in terms of product selectivity and ease of isolation. Because the reaction occurs within the confines of the well-defined rectangular complex, the formation of unwanted regioisomers or polymeric byproducts is effectively suppressed. The result is a reaction profile characterized by high conversion efficiency and the generation of a single, dominant product. Moreover, the subsequent release of the cyclobutane product is driven by the relief of ring strain, which triggers the cleavage of the gold-nitrogen bonds, allowing the product to dissociate from the metal framework. This self-releasing mechanism simplifies the workup procedure, as the cyclobutane derivative can often be isolated through simple precipitation or filtration, avoiding the need for complex separation of host-guest complexes. For supply chain heads concerned with the commercial scale-up of complex intermediates, this streamlined workflow represents a robust and scalable alternative to traditional photochemical methods.
Mechanistic Insights into Gold Carbene Templated Photocyclization
The mechanistic elegance of this synthesis lies in the dual role of the gold carbene complex as both a structural scaffold and a reactive precursor. The process begins with the reaction of a binuclear gold carbene complex, typically featuring N-heterocyclic carbene (NHC) ligands, with a bis-olefinic substrate in the presence of a silver salt promoter such as AgOTf. The silver ion facilitates the abstraction of halide ligands from the gold centers, generating cationic gold species that are highly electrophilic and prone to coordination with the electron-rich olefin double bonds. This coordination event drives the self-assembly of the rectangular intermediate, denoted as [3a-3u](OTf)4 in the patent literature. The rigidity of the NHC-gold backbone ensures that the two olefin arms are held at a fixed distance and angle, creating an ideal microenvironment for the subsequent photochemical transformation.
Upon exposure to ultraviolet light, specifically at a wavelength of 365 nm, the pre-aligned olefins within the rectangular complex undergo a concerted [2+2] cycloaddition. This photo-excitation promotes an electron from the bonding pi-orbital to the anti-bonding pi*-orbital, allowing the formation of new sigma bonds between the carbon atoms of the opposing double bonds. The formation of the cyclobutane ring introduces significant angle strain into the molecular framework. Interestingly, this strain energy is harnessed to drive the final step of the mechanism: the fragmentation of the gold template. The formation of the strained four-membered ring destabilizes the coordination environment, leading to the cleavage of the gold-nitrogen bonds that tether the substrate to the carbene ligands. This results in the release of the free cyclobutane derivative and the regeneration of the gold species, which can potentially be recycled.
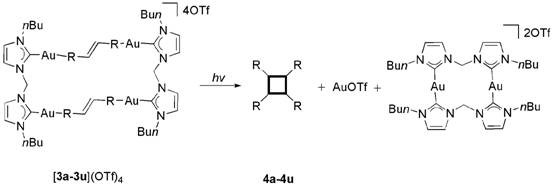
From an impurity control perspective, this mechanism offers superior predictability compared to free-solution photochemistry. In traditional methods, the random collision of excited state molecules can lead to a variety of head-to-head, head-to-tail, or tail-to-tail dimerization products. However, the topological constraints imposed by the rectangular gold cage enforce a specific head-to-head (or defined) alignment, ensuring that only one stereoisomer is formed. This high degree of stereocontrol is critical for pharmaceutical applications where the biological activity is often dependent on the specific three-dimensional arrangement of the molecule. By minimizing the generation of stereoisomeric impurities, this method reduces the burden on analytical quality control and simplifies the regulatory filing process for new drug candidates containing these motifs.
How to Synthesize Cyclobutane Derivatives Efficiently
The practical implementation of this gold carbene templated synthesis involves a straightforward two-stage protocol that balances high yield with operational simplicity. The first stage focuses on the quantitative assembly of the rectangular gold precursor under dark conditions to prevent premature photocyclization, while the second stage utilizes controlled UV irradiation to trigger the ring-closing event. This separation of steps allows for rigorous quality control of the intermediate before committing to the photochemical transformation. Detailed standardized synthetic steps for this process are provided in the guide below.
- Preparation of the rectangular gold carbene precursor by reacting a binuclear gold carbene complex with C=C olefin ligands in dichloromethane using AgOTf.
- Isolation of the intermediate gold complex through filtration and washing to ensure high purity before the photochemical step.
- Execution of the [2+2] photochemical cycloaddition under 365 nm UV irradiation to induce ring closure and release the final cyclobutane derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of the gold carbene template method described in CN109111392B offers compelling economic and logistical benefits that extend beyond mere chemical novelty. The primary value proposition lies in the drastic simplification of the purification workflow. Traditional photochemical syntheses often require extensive chromatographic separation to isolate the desired cyclobutane from a soup of oligomers and unreacted starting materials. In contrast, the high selectivity of the gold-templated reaction means that the crude product is already of high purity, often allowing for isolation via simple crystallization or precipitation. This reduction in downstream processing time translates directly into lower solvent consumption, reduced waste generation, and decreased labor costs, contributing to a significantly reduced overall cost of goods sold (COGS).
- Cost Reduction in Manufacturing: The elimination of external template agents such as cyclodextrins or specialized MOFs removes a major cost driver from the raw material bill. These auxiliary substances are often expensive and difficult to recover, representing a sunk cost in every batch. By using the gold carbene complex as a self-contained reactor, the process avoids these expenditures entirely. Furthermore, the high conversion efficiency minimizes the amount of unreacted starting material that needs to be recovered and recycled, streamlining the material flow. The ability to operate at room temperature also reduces energy consumption associated with heating or cooling reactors, further enhancing the economic viability of the process for large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes to greater supply chain stability. The reagents involved, including the gold carbene precursors and common olefin ligands, are chemically stable and can be sourced from established suppliers or synthesized in-house with high reproducibility. Unlike enzymatic or biocatalytic processes that may be sensitive to minor fluctuations in pH or temperature, this chemical method is tolerant of standard industrial operating variances. This reliability ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results. For a reliable pharmaceutical intermediate supplier, this consistency is key to maintaining long-term contracts with global partners.
- Scalability and Environmental Compliance: Scaling photochemical reactions is notoriously difficult due to the limited penetration depth of UV light in large vessels. However, the high efficiency of this templated reaction means that shorter irradiation times are required, making it more amenable to continuous flow processing or the use of thin-film reactors which solve the light penetration issue. Additionally, the process generates less chemical waste due to the absence of auxiliary hosts and the high selectivity of the reaction. This aligns well with modern green chemistry principles and environmental regulations, reducing the burden on waste treatment facilities and lowering the environmental compliance costs associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this gold carbene templated synthesis. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and advantages of this approach for industrial applications.
Q: What is the primary advantage of the gold carbene template method over traditional photochemical synthesis?
A: The primary advantage is the elimination of external auxiliary templates like cyclodextrins or MOFs. The gold carbene complex itself acts as a rigid rectangular scaffold that pre-aligns the olefin ligands perfectly for [2+2] cycloaddition, resulting in single-product formation and significantly easier separation.
Q: How does this method impact the purity profile of the final cyclobutane derivative?
A: Because the reaction relies on a highly ordered supramolecular assembly within the gold complex, side reactions such as polymerization or random oligomerization are effectively suppressed. This leads to a much cleaner crude reaction mixture and simplifies downstream purification processes.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction conditions (room temperature) and common solvents like dichloromethane and acetonitrile. The high conversion efficiency and the ability to recover the gold species suggest strong potential for commercial scale-up with optimized recycling protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclobutane Derivative Supplier
The technological breakthroughs encapsulated in patent CN109111392B highlight the immense potential of gold carbene chemistry in accessing valuable strained ring systems. At NINGBO INNO PHARMCHEM, we recognize the strategic importance of such advanced synthetic methodologies in the development of next-generation therapeutics. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from discovery to market is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of cyclobutane intermediate meets the highest industry standards.
We invite you to explore how our capabilities can optimize your supply chain for complex organic intermediates. Whether you are looking to secure a stable source of high-purity building blocks or need assistance in adapting this gold-templated route for your specific API candidates, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your project requirements. Let us provide you with specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in delivering high-quality chemical solutions efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
