Advanced Manufacturing of 1,1-Dimethyl-4,4'-Bipyridine Dichloride via Green Methylation and Ion Exchange
Advanced Manufacturing of 1,1-Dimethyl-4,4'-Bipyridine Dichloride via Green Methylation and Ion Exchange
The chemical industry is currently witnessing a pivotal shift towards greener, more sustainable synthetic pathways, particularly for high-value heterocyclic compounds used in pharmaceutical and agrochemical sectors. A recent technological breakthrough, documented in patent CN115611800A, introduces a highly efficient preparation method for 1,1-dimethyl-4,4'-bipyridine dichloride, a critical intermediate often utilized in redox applications and specialized material synthesis. This innovation departs radically from legacy manufacturing techniques by employing a mild, two-step sequence that begins with the methylation of 4,4'-bipyridine using methyl iodide, followed by a strategic anion exchange. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediates supplier partnerships, this patent represents a significant leap forward in process safety and environmental compliance. By eliminating the need for hazardous high-pressure reactors and toxic cyanide reagents, this method not only simplifies the operational workflow but also drastically reduces the ecological footprint associated with large-scale production. The ability to achieve high conversion rates and yields under such benign conditions underscores the potential for this technology to redefine the supply chain dynamics for complex bipyridyl derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 1,1-dimethyl-4,4'-bipyridyl dichloride has been dominated by the ammonia cyanogen method, a process fraught with significant operational hazards and environmental liabilities. This traditional route necessitates the alkylation of pyridine with methyl chloride followed by a dimerization reaction in the presence of ammonia and cyanide ions, a combination that inherently demands high-pressure reaction vessels to drive the chemistry to completion. The reliance on sodium cyanide introduces severe toxicity risks, requiring elaborate containment systems and expensive waste treatment protocols to neutralize cyanide-laden effluents before discharge. Furthermore, the harsh reaction conditions often lead to the formation of complex impurity profiles that are difficult to separate, thereby compromising the overall purity of the final API intermediate. For supply chain managers, these factors translate into elevated insurance costs, stringent regulatory scrutiny, and potential disruptions due to the handling of controlled hazardous substances. The inability to easily recycle reagents in this legacy process further exacerbates the cost structure, making it increasingly uncompetitive in a market that prioritizes green chemistry principles.
The Novel Approach
In stark contrast, the methodology outlined in patent CN115611800A offers a streamlined, catalyst-free alternative that operates under remarkably mild conditions, effectively bypassing the pitfalls of the cyanogen route. By utilizing 4,4'-bipyridine as the starting scaffold and reacting it directly with methyl iodide in acetonitrile, the process achieves quaternization without the need for extreme pressures or toxic catalysts. The subsequent anion exchange step allows for the precise replacement of iodide counterions with chloride ions using regenerable ion-exchange resins, ensuring the final product meets stringent purity specifications required for high-end applications. This approach not only simplifies the equipment requirements—allowing for the use of standard glass-lined or stainless steel reactors—but also facilitates the recovery and recycling of valuable iodine species and solvents. For companies focused on cost reduction in pharmaceutical intermediates manufacturing, this transition represents a strategic opportunity to lower both capital expenditure on safety infrastructure and operational expenditure on waste management. The robustness of this two-step sequence ensures consistent quality and yield, making it an ideal candidate for commercial scale-up of complex fine chemicals.
Mechanistic Insights into Quaternization and Anion Exchange
The core of this innovative synthesis lies in the nucleophilic substitution mechanism governing the initial methylation step, where the lone pair electrons on the nitrogen atoms of the 4,4'-bipyridine ring attack the electrophilic methyl carbon of methyl iodide. This reaction proceeds efficiently in polar aprotic solvents like acetonitrile, which stabilize the transition state and facilitate the formation of the quaternary ammonium iodide salt. The absence of external catalysts is a critical feature, as it eliminates the risk of metal contamination in the final product, a common concern in pharmaceutical synthesis that often necessitates costly purification steps. Following the formation of the deep red iodized salt intermediate, the process leverages the principles of ion exchange chromatography to swap the iodide anions for chloride anions. This is achieved by passing an aqueous solution of the iodized salt through a column packed with a chloride-type strong base anion exchange resin, such as Tulsimer A-62MP. The resin preferentially binds the iodide ions while releasing chloride ions into the solution, driven by the concentration gradient and the specific selectivity of the resin matrix.
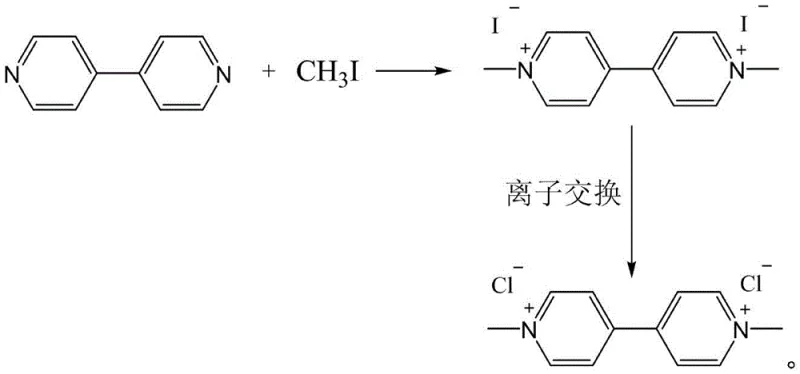
Controlling the impurity profile in this process is inherently managed by the specificity of the reactants and the physical separation methods employed. Since the methylation reaction is highly selective for the nitrogen centers of the bipyridine ring, side reactions such as ring alkylation or polymerization are minimal, especially when stoichiometry is carefully controlled within the preferred molar ratios of 1:1 to 1:5. The solid iodized salt precipitates out of the acetonitrile solution upon completion, allowing for a simple filtration step that physically separates the product from soluble organic impurities and unreacted starting materials. Furthermore, the anion exchange step acts as an additional purification barrier; the resin column can effectively trap trace organic contaminants or colored bodies that might have co-precipitated, resulting in a white, high-purity dichloride salt after evaporation. This dual-mechanism of chemical selectivity followed by physical and chromatographic purification ensures that the final high-purity 1,1-dimethyl-4,4'-bipyridine dichloride meets the rigorous standards demanded by downstream applications in electronics and medicine.
How to Synthesize 1,1-Dimethyl-4,4'-Bipyridine Dichloride Efficiently
Implementing this synthesis route requires careful attention to solvent quality and resin capacity to maximize the efficiency of the ion exchange cycle. The process begins by dissolving 4,4'-bipyridine in dry acetonitrile and adding methyl iodide under controlled temperature conditions, typically ranging from 30°C to 120°C depending on the desired reaction kinetics. Once the reddish-brown iodized salt has formed and been isolated via filtration, it is redissolved in water and passed through the ion exchange column at a flow rate optimized for mass transfer, generally between 5 BV/h and 20 BV/h. The detailed standardized operating procedures, including specific work-up techniques and resin regeneration protocols, are critical for maintaining batch-to-batch consistency.
- React 4,4'-bipyridine with methyl iodide in acetonitrile solvent at temperatures between 30°C and 120°C to form the iodized salt intermediate.
- Filter the resulting reddish-brown iodized salt and dry the solvent for recycling purposes.
- Perform anion exchange using a chloride-type ion resin (such as Tulsimer A-62MP) to convert the iodized salt into the final 1,1-dimethyl-4,4'-bipyridine dichloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic benefits that extend far beyond simple chemical transformation. The elimination of high-pressure reactors and toxic cyanide reagents fundamentally alters the risk profile of the manufacturing site, leading to reduced insurance premiums and fewer regulatory hurdles. Moreover, the ability to recycle the acetonitrile solvent and regenerate the ion exchange resin creates a closed-loop system that minimizes raw material consumption and waste generation. This circular approach not only aligns with corporate sustainability goals but also insulates the supply chain from volatility in raw material pricing. By securing a reliable pharmaceutical intermediates supplier who utilizes this technology, buyers can ensure a steady flow of material without the disruption risks associated with hazardous waste disposal or strict environmental crackdowns on older, dirtier technologies.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the reaction setup and the recyclability of key inputs. By removing the need for expensive high-pressure equipment and the associated safety monitoring systems, capital investment is significantly lowered. Additionally, the recovery of iodine in the form of sodium iodide solution during the resin regeneration phase allows for the potential reuse of this valuable halogen, effectively reducing the net consumption of methyl iodide over time. The catalyst-free nature of the reaction further contributes to cost savings by eliminating the expense of purchasing and subsequently removing heavy metal catalysts, streamlining the downstream processing workflow.
- Enhanced Supply Chain Reliability: Operational continuity is greatly improved due to the mild reaction conditions and the stability of the reagents involved. Unlike processes that rely on gaseous methyl chloride or unstable cyanide salts, the use of liquid methyl iodide and solid 4,4'-bipyridine simplifies logistics and storage requirements. The robustness of the ion exchange step ensures that even if minor variations occur in the upstream methylation, the final product quality can be maintained through the purification capability of the resin. This resilience translates to reducing lead time for high-purity intermediates, as there is less need for extensive re-processing or batch rejection due to safety incidents or equipment failures common in high-pressure systems.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward because it relies on unit operations that are well-understood and easily manageable, such as filtration and column chromatography. The absence of toxic cyanide waste means that effluent treatment is less complex and costly, facilitating easier compliance with increasingly stringent global environmental regulations. The regenerable nature of the ion exchange resin means that solid waste generation is minimal, supporting a greener manufacturing footprint that appeals to environmentally conscious stakeholders and end-users in the pharmaceutical and agrochemical industries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 1,1-dimethyl-4,4'-bipyridine dichloride using this advanced methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear picture of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their own supply chains or product formulations.
Q: How does this new process improve safety compared to traditional methods?
A: Unlike the conventional ammonia cyanogen method which requires high pressure and generates toxic sodium cyanide waste, this novel process operates under mild conditions without catalysts and eliminates the use of highly toxic cyanide reagents, significantly enhancing operational safety.
Q: Can the raw materials and byproducts be recycled in this synthesis?
A: Yes, the process is designed for sustainability; the acetonitrile solvent can be recovered after filtration, the iodine element participating in the reaction can be recycled as sodium iodide solution during resin regeneration, and the anion exchange resin itself is regenerable using aqueous NaCl solutions.
Q: What are the typical reaction conditions for the methylation step?
A: The methylation of 4,4'-bipyridine with methyl iodide typically proceeds in acetonitrile at temperatures ranging from 30°C to 120°C, with reaction times varying between 2 hours and 24 hours depending on the desired conversion rate and molar ratios used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Dimethyl-4,4'-Bipyridine Dichloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting next-generation synthetic routes to meet the evolving demands of the global fine chemical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless and efficient. We are committed to delivering stringent purity specifications for all our intermediates, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch against the highest industry standards. By leveraging the green chemistry principles outlined in recent innovations, we provide our partners with a sustainable and cost-effective source of essential building blocks.
We invite you to collaborate with us to explore how this optimized synthesis can benefit your specific application needs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals.
