Advanced Catalytic Strategy for Diarylmethyl Phosphonates: Scalable Manufacturing for Global Pharma Supply Chains
The landscape of organophosphorus chemistry is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic routes in the pharmaceutical and fine chemical sectors. A pivotal advancement in this domain is detailed in patent CN109456362B, which discloses a novel method for the highly efficient and selective preparation of diarylmethyl-substituted organic phosphonate derivatives. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a robust base-catalyzed 1,6-addition strategy. By leveraging stable P(O)-H compounds and 4-arylmethylene-2,6-di-tert-butyl-2,5-cyclohexadien-1-one derivatives as key building blocks, this methodology addresses critical bottlenecks regarding substrate stability, reaction safety, and environmental impact. For R&D directors and procurement managers seeking reliable pharmaceutical intermediate suppliers, understanding the mechanistic elegance and operational simplicity of this patent is crucial for optimizing supply chains and reducing the cost of goods sold (COGS) in complex API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diarylmethyl-substituted organophosphonates has been plagued by significant technical and economic hurdles that hinder large-scale commercial adoption. Traditional pathways, such as Friedel-Crafts reactions utilizing ferric chloride or nucleophilic couplings dependent on palladium, copper, and nickel catalysts, impose severe constraints on process safety and purity profiles. These legacy methods frequently necessitate the use of air-sensitive reagents like trialkyl phosphites or phosphorus oxychloride, which are not only highly corrosive but also pose substantial handling risks in industrial settings. Furthermore, the reliance on precious transition metals introduces complex downstream purification challenges, as residual metal levels must be strictly controlled to meet stringent pharmacopeial standards. The cumulative effect of these factors—harsh reaction conditions, expensive catalyst loading, difficult recycling protocols, and poor atom economy—results in inflated production costs and extended lead times, making conventional routes increasingly untenable for modern, cost-conscious fine chemical manufacturing.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the innovative strategy outlined in the patent data employs a direct 1,6-addition reaction that fundamentally simplifies the synthetic architecture. By utilizing readily available P(O)-H compounds reacting with sterically hindered quinone methide precursors, the process achieves exceptional chemoselectivity without the need for exotic ligands or sensitive organometallic species. The reaction proceeds smoothly under a nitrogen atmosphere at mild temperatures ranging from 25°C to 80°C, utilizing inexpensive inorganic bases like cesium carbonate as the catalytic driver. This approach not only eliminates the toxicity associated with heavy metal catalysts but also ensures that the target product is formed with selectivity approaching 100%, drastically reducing the burden on purification units. The versatility of this method is further evidenced by its broad substrate scope, accommodating various electron-donating and electron-withdrawing groups on both the phosphorus and aryl moieties, thereby offering a universal platform for the commercial scale-up of complex pharmaceutical intermediates.
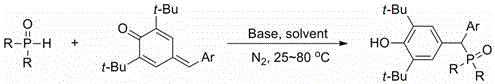
Mechanistic Insights into Base-Catalyzed 1,6-Addition
The core of this technological breakthrough lies in the unique reactivity profile of the 4-arylmethylene-2,6-di-tert-butyl-2,5-cyclohexadien-1-one system, which acts as a highly activated Michael acceptor. Under the influence of a mild base such as cesium carbonate, the P(O)-H compound undergoes deprotonation to generate a nucleophilic phosphorus anion. This anion then executes a conjugate addition at the exocyclic methylene carbon of the quinone methide derivative. Unlike traditional electrophilic aromatic substitutions that often suffer from regioselectivity issues, this 1,6-addition is driven by the thermodynamic stability gained upon rearomatization of the cyclohexadienone ring to a phenolic system. The presence of the bulky tert-butyl groups at the 2 and 6 positions plays a critical steric role, preventing unwanted side reactions at the ortho-positions and effectively channeling the nucleophilic attack exclusively to the exocyclic double bond. This precise control over the reaction trajectory is what enables the observed near-quantitative selectivity, ensuring that the final high-purity organic phosphonates are generated with minimal formation of regioisomeric impurities.
From an impurity control perspective, the mechanism offers distinct advantages over transition-metal catalyzed cross-couplings. In Pd or Cu-catalyzed systems, common impurities often arise from homocoupling of the phosphorus species or incomplete halide conversion, which are notoriously difficult to separate due to similar polarity. However, in this base-mediated protocol, the primary byproduct is simply the protonated base or unreacted starting material, both of which possess significantly different physicochemical properties compared to the target phosphonate. The driving force of aromatization ensures that once the addition occurs, the reaction is essentially irreversible under the stated conditions, pushing the equilibrium towards completion. This kinetic and thermodynamic favorability allows for the use of stoichiometric or near-stoichiometric ratios of reactants, minimizing waste generation. For quality assurance teams, this translates to a much cleaner crude reaction profile, facilitating simpler work-up procedures and reducing the number of chromatographic passes required to achieve the stringent purity specifications demanded by global regulatory bodies.
How to Synthesize Diarylmethyl Phosphonates Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic cooling systems typically associated with organometallic chemistry. The standard protocol involves charging the reaction vessel with the P(O)-H substrate, the quinone methide derivative, and the cesium carbonate catalyst in a suitable organic solvent such as acetonitrile or dichloromethane. Maintaining an inert nitrogen atmosphere is critical to prevent oxidation of the phosphorus species, although the reagents themselves are significantly more stable than their trivalent counterparts. Detailed standard operating procedures (SOPs) regarding mixing rates, temperature ramping, and quenching strategies are essential for reproducibility. For a comprehensive guide on the exact molar ratios and specific solvent optimizations validated in the patent examples, please refer to the standardized synthesis steps provided below.
- Mix P(O)-H compound, 4-arylmethylene-2,6-di-tert-butyl-2,5-cyclohexadien-1-one, cesium carbonate catalyst, and organic solvent in a reaction vessel under nitrogen atmosphere.
- Stir the reaction mixture at a temperature range of 25°C to 80°C for a duration of 3 to 6 hours to ensure complete conversion.
- Upon completion, purify the resulting diarylmethyl-substituted organic phosphonate derivatives using standard column chromatography techniques.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic methodology offers profound strategic benefits that extend far beyond simple yield improvements. The elimination of precious metal catalysts such as palladium and platinum removes a major variable cost component and mitigates supply risk associated with volatile metal markets. Furthermore, the use of stable, shelf-stable P(O)-H compounds instead of corrosive phosphorus halides or air-sensitive phosphites significantly enhances workplace safety and reduces the costs associated with hazardous waste disposal and specialized storage infrastructure. The mild reaction conditions (25-80°C) imply lower energy consumption for heating and cooling, contributing to a reduced carbon footprint and aligning with corporate sustainability goals. These factors collectively contribute to a more resilient and cost-effective supply chain, enabling manufacturers to offer competitive pricing while maintaining robust inventory levels of critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the substitution of expensive transition metal catalysts and specialized ligands with commodity inorganic bases like cesium carbonate. In traditional cross-coupling workflows, the cost of the catalyst system combined with the extensive purification steps required to remove trace metals (often involving scavenger resins or additional crystallization cycles) can account for a significant portion of the total manufacturing cost. By adopting this metal-free approach, manufacturers can drastically simplify the downstream processing train, reducing solvent usage, filtration time, and resin consumption. Additionally, the high selectivity minimizes the loss of valuable starting materials to side products, improving the overall mass balance and effective yield. This streamlined process flow directly translates to substantial cost savings per kilogram of finished product, allowing for more aggressive pricing strategies in competitive tender scenarios without compromising margin integrity.
- Enhanced Supply Chain Reliability: Supply chain continuity is often jeopardized by the reliance on specialized reagents that have limited global suppliers or long lead times. The reagents utilized in this patented method, specifically the P(O)-H compounds and the quinone methide precursors, are derived from widely available commodity chemicals and can be sourced from multiple vendors globally. This diversification of the raw material base reduces the risk of supply disruptions caused by geopolitical issues or single-source manufacturing failures. Moreover, the stability of these reagents allows for bulk purchasing and long-term storage without significant degradation, enabling companies to build strategic stockpiles during favorable market conditions. The robustness of the reaction conditions also means that production is less susceptible to minor fluctuations in utility availability (such as chilled water or steam pressure), ensuring consistent output even during periods of infrastructure stress, thereby guaranteeing reliable delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often exposes hidden inefficiencies, particularly regarding heat management and waste treatment. The exothermic nature of many traditional phosphorylation reactions requires careful thermal control to prevent runaway scenarios, limiting batch sizes and reactor utilization. In contrast, the mild thermal profile of this 1,6-addition reaction facilitates safer scale-up, allowing for larger batch sizes and higher throughput in existing reactor fleets without the need for capital-intensive retrofitting. From an environmental compliance standpoint, the absence of heavy metals and halogenated phosphorus reagents significantly reduces the toxicity of the effluent stream. This simplifies wastewater treatment protocols and lowers the fees associated with hazardous waste disposal. The alignment with Green Chemistry principles not only satisfies regulatory requirements but also enhances the brand reputation of the manufacturer as a sustainable partner, which is increasingly becoming a key selection criterion for multinational pharmaceutical corporations evaluating their vendor networks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps R&D and procurement teams evaluate the feasibility of integrating this route into their existing portfolios. Whether the concern is about catalyst recovery, substrate scope limitations, or purity profiles, the answers below reflect the verified capabilities of the described method.
Q: What catalyst system is utilized in this novel synthesis method?
A: The process utilizes inexpensive inorganic bases such as cesium carbonate (Cs2CO3), sodium carbonate, or potassium carbonate, eliminating the need for expensive transition metal catalysts.
Q: How does this method improve upon traditional Friedel-Crafts or Arbuzov reactions?
A: Unlike traditional methods requiring air-sensitive reagents or harsh Lewis acids, this approach operates under mild conditions (25-80°C) with near 100% selectivity and avoids toxic byproducts.
Q: What yields can be expected from this 1,6-addition reaction?
A: Experimental data indicates isolated yields ranging from 67% to 99%, with selectivity approaching 100% for the target diarylmethyl phosphonate derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diarylmethyl Phosphonate Supplier
As the global demand for high-performance organophosphorus compounds continues to rise in the pharmaceutical and agrochemical sectors, having a manufacturing partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced catalytic technologies like the one described in patent CN109456362B to deliver superior value. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from benchtop discovery to full-scale industrial supply. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for GMP environments. Our commitment to technical excellence ensures that we can handle complex molecular architectures with the precision and reliability that top-tier pharmaceutical companies demand.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your specific project requirements. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to this metal-free protocol can impact your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data for similar structures and to discuss route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access not just to a chemical supplier, but to a strategic ally dedicated to driving efficiency, quality, and innovation in your supply chain.
