High-Purity S-3-Cyclohexenecarboxylic Acid: Scalable Purification for Edoxaban Synthesis
Introduction to Advanced Chiral Resolution Technology
The pharmaceutical industry faces relentless pressure to secure high-purity intermediates for complex small molecule drugs, particularly anticoagulants like Edoxaban. A pivotal advancement in this domain is detailed in patent CN111099989A, which outlines a robust purification method for S-3-cyclohexenecarboxylic acid. This technology addresses the critical bottleneck of separating chiral enantiomers, transforming crude material with an ee value of less than 97% into a highly refined product exceeding 99% ee. For R&D directors and procurement strategists, this represents a significant leap forward in process reliability, offering a pathway to eliminate impurities that could compromise downstream synthesis efficiency. The method leverages classical diastereomeric salt formation but optimizes it through precise solvent systems and thermal controls, ensuring that the final intermediate meets the rigorous specifications required for global regulatory submission.
Furthermore, the economic implications of this purification strategy extend beyond mere purity metrics. By integrating a closed-loop recovery system for the chiral resolving agent, the process mitigates one of the most substantial cost drivers in chiral chemistry: the consumption of expensive amines. This approach not only enhances the sustainability profile of the manufacturing route but also stabilizes the supply chain against fluctuations in raw material pricing. As a reliable pharmaceutical intermediates supplier, understanding these nuanced process improvements is essential for evaluating long-term vendor partnerships. The ability to consistently deliver >99% ee material without resorting to costly preparative HPLC or enzymatic kinetic resolutions positions this technology as a cornerstone for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the acquisition of single-enantiomer carboxylic acids has relied on three primary strategies, each fraught with distinct operational challenges that hinder large-scale commercial viability. Chiral pool synthesis, while elegant, is severely constrained by the availability and cost of natural chiral starting materials, often limiting the structural diversity accessible to process chemists. Asymmetric synthesis, although powerful, frequently struggles to achieve optical yields greater than 90% without extensive optimization of ligand libraries and reaction conditions, leading to prolonged development timelines. Moreover, biological asymmetric synthesis, despite its high selectivity, often suffers from slow reaction kinetics and complex product isolation protocols that are difficult to translate from the laboratory to multi-ton production scales.
Perhaps the most common historical approach, standard racemate resolution via fractional crystallization, often fails to provide the necessary purity uplift in a single pass. Conventional resolution processes are highly sensitive to minor variations in cooling rates, solvent composition, and seeding protocols, resulting in batch-to-batch inconsistency. In many existing methods, achieving an ee value above 97% requires multiple recrystallization cycles that drastically reduce overall yield, sometimes rendering the process economically unfeasible. Additionally, the failure to efficiently recover the resolving agent in traditional workflows leads to significant waste generation and inflated raw material costs, creating a dual burden on both the P&L statement and environmental compliance metrics.
The Novel Approach
The methodology described in the patent introduces a streamlined, three-stage protocol that overcomes these historical inefficiencies through precise control of thermodynamic parameters. The process initiates with a salification step using R-a-phenylethylamine in ethyl acetate, where the molar ratio is carefully tuned between 1:1 and 1:1.5 to maximize the formation of the target diastereomeric salt while minimizing the co-precipitation of the undesired enantiomer. This is followed by a rigorous recrystallization phase in ethanol, where temperature gradients are managed between 60-100°C for dissolution and 30-50°C for crystallization, effectively purging impurities through repeated solid-liquid equilibria. The final dissociation step utilizes a biphasic system of dichloromethane and water with pH control, ensuring clean liberation of the free acid.
What truly distinguishes this novel approach is its inherent design for circularity and scalability. Unlike methods that discard the mother liquor or the resolving agent, this protocol explicitly includes a recovery loop where the aqueous phase is basified to reclaim the R-a-phenylethylamine. This feature transforms a linear, wasteful process into a cyclical, efficient operation that aligns with modern green chemistry principles. For supply chain heads, this means a reduction in the volume of hazardous waste requiring disposal and a decreased dependency on external suppliers for chiral amines. The simplicity of the unit operations—reflux, filtration, and distillation—ensures that the process can be easily scaled from pilot plants to commercial reactors without requiring specialized equipment or exotic catalysts.
Mechanistic Insights into Diastereomeric Salt Resolution
At the heart of this purification technology lies the fundamental principle of diastereomeric salt formation, where the physical properties of the resulting salts differ sufficiently to allow separation. When the racemic or enriched crude S-3-cyclohexenecarboxylic acid reacts with the chiral base R-a-phenylethylamine, two distinct salts are formed: the S-acid/R-amine salt and the R-acid/R-amine salt. These salts are diastereomers, meaning they possess different crystal lattice energies and solubility profiles in specific solvents like ethyl acetate and ethanol. The patent exploits these subtle differences by optimizing the solvent environment to favor the precipitation of the less soluble S-acid/R-amine complex, while the more soluble R-acid/R-amine species remains in the mother liquor. This selective crystallization is the primary driver for the enrichment of optical purity from <97% to >99%.
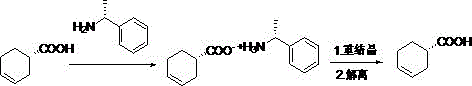
The recrystallization step further refines this separation through Ostwald ripening and defect correction within the crystal lattice. By dissolving the crude salt at elevated temperatures (up to 100°C) and slowly cooling to 30-50°C, the system allows for the growth of highly ordered crystals that exclude impurity molecules. Repeating this operation three times, as suggested in the examples, acts as a multi-stage distillation equivalent for solids, progressively stripping away trace amounts of the opposite enantiomer. The final dissociation step is equally critical; by adjusting the pH to 1-3 using hydrochloric acid, the ammonium salt is protonated, releasing the free amine into the aqueous phase and the free carboxylic acid into the organic dichloromethane phase. This clean phase separation ensures that no amine residues contaminate the final API intermediate, a crucial factor for passing rigorous heavy metal and residual solvent tests.
From an impurity control perspective, this mechanism offers superior predictability compared to chromatographic methods. In column chromatography, band broadening and tailing can lead to cross-contamination that is difficult to model. In contrast, crystallization-based purification relies on well-defined phase diagrams, allowing process engineers to map out the eutectic points and solubility curves with high precision. This determinism reduces the risk of unexpected impurity spikes during scale-up. Furthermore, the use of common organic solvents like ethyl acetate, ethanol, and dichloromethane ensures that residual solvent levels can be easily managed to meet ICH Q3C guidelines. The robustness of this mechanistic pathway provides R&D teams with a high degree of confidence in the reproducibility of the final product quality.
How to Synthesize S-3-Cyclohexenecarboxylic Acid Efficiently
Implementing this purification route requires strict adherence to the thermal and stoichiometric parameters defined in the patent to ensure optimal yield and purity. The process begins with the preparation of the diastereomeric salt, followed by iterative recrystallization to boost enantiomeric excess, and concludes with acid-base extraction to isolate the free acid. Operators must pay close attention to the cooling rates during crystallization and the pH control during dissociation to prevent emulsion formation or oiling out. The following guide outlines the standardized operational sequence derived from the patent examples, serving as a foundational reference for process validation and technology transfer activities.
- Mix crude S-3-cyclohexenecarboxylic acid (ee < 97%) with R-a-phenylethylamine in ethyl acetate, reflux at 50-100°C, and filter to obtain the diastereomeric salt.
- Dissolve the salt in ethanol at 60-100°C, cool to 30-50°C to induce crystallization, and filter repeatedly to enhance optical purity.
- Treat the recrystallized material with dichloromethane, water, and hydrochloric acid (pH 1-3) to dissociate the salt, then separate and distill the organic layer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this purification technology translates directly into tangible operational efficiencies and risk mitigation. The primary advantage lies in the drastic simplification of the downstream processing workflow. By eliminating the need for complex chromatographic columns or expensive enzymatic reactors, the capital expenditure (CAPEX) required for setting up production lines is significantly lowered. The reliance on standard chemical engineering unit operations such as filtration and distillation means that existing multipurpose facilities can be utilized without major retrofitting. This flexibility allows for faster time-to-market for new drug applications and provides the agility to respond to sudden spikes in demand for Edoxaban intermediates without the lead time associated with installing specialized equipment.
- Cost Reduction in Manufacturing: The economic model of this process is heavily fortified by the recovery and reuse of the chiral resolving agent, R-a-phenylethylamine. In traditional resolution processes, the resolving agent is often treated as a consumable, representing a substantial portion of the raw material bill. By implementing the recovery step described in the patent, where the aqueous layer is basified and extracted, the net consumption of this expensive reagent is minimized. This circular approach leads to substantial cost savings over the lifecycle of the product. Additionally, the high yield reported in the examples (82-86%) ensures that the input cost of the crude acid is amortized over a larger quantity of saleable product, further driving down the cost per kilogram.
- Enhanced Supply Chain Reliability: The reagents utilized in this synthesis—ethyl acetate, ethanol, dichloromethane, and hydrochloric acid—are commodity chemicals with robust, global supply chains. Unlike specialized chiral ligands or biocatalysts that may have single-source suppliers and long lead times, these solvents and reagents are readily available from multiple vendors. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Furthermore, the process operates under relatively mild conditions (reflux temperatures rather than cryogenic cooling), reducing the energy load and the dependency on specialized utility infrastructure, thereby enhancing the overall resilience of the manufacturing site.
- Scalability and Environmental Compliance: Scaling a crystallization process is generally more predictable than scaling a kinetic resolution, as it relies on thermodynamic equilibrium rather than reaction rate constants. This makes the transition from 100 kgs to multi-ton production smoother and less prone to unforeseen exotherms or mixing issues. From an environmental standpoint, the ability to recycle the amine reduces the chemical oxygen demand (COD) of the waste stream. The use of distillation for solvent recovery also aligns with green manufacturing initiatives, allowing for the minimization of volatile organic compound (VOC) emissions. These factors collectively simplify the permitting process and ensure long-term compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. They are derived from a detailed analysis of the patent specifications and are intended to clarify the operational boundaries and potential benefits for stakeholders. Understanding these nuances is critical for making informed decisions about process adoption and vendor qualification.
Q: What is the final optical purity achieved by this purification method?
A: The patented process consistently achieves an enantiomeric excess (ee) value of greater than 99%, meeting the stringent requirements for anticoagulant synthesis.
Q: Can the chiral resolving agent be recovered in this process?
A: Yes, the process includes a specific recovery step where the aqueous layer is treated with sodium hydroxide and extracted to recover R-a-phenylethylamine for reuse, significantly reducing material costs.
Q: Why is S-3-cyclohexenecarboxylic acid critical for Edoxaban production?
A: S-3-cyclohexenecarboxylic acid serves as a key chiral building block for Edoxaban; the R-enantiomer is inactive for this specific therapeutic application, making high-purity separation essential.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-3-Cyclohexenecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceuticals like Edoxaban depends on the unwavering quality and availability of key chiral intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We leverage advanced purification technologies, such as the resolution method discussed herein, to deliver S-3-cyclohexenecarboxylic acid with stringent purity specifications that exceed industry standards. Our rigorous QC labs employ state-of-the-art chiral HPLC methods to verify ee values and impurity profiles, guaranteeing that every batch supports your regulatory filings without delay.
We invite you to collaborate with us to optimize your supply chain for anticoagulant synthesis. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can drive efficiency and reduce total landed costs for your organization. Let us be your strategic partner in delivering high-performance chemical solutions.
