Scalable Manufacturing of 4,4'-Disubstituted-2,2'-Diaminobiphenyl via Novel Reductive Coupling
Introduction to Advanced Reductive Coupling Technology
The chemical industry is constantly seeking more efficient pathways to synthesize complex biaryl amines, which serve as critical building blocks for high-performance polymers and pharmaceutical agents. Patent CN111574380A introduces a groundbreaking method for preparing 4,4'-disubstituted-2,2'-diaminobiphenyl and its hydrochloride salts through a novel reductive coupling strategy. This technology represents a significant departure from traditional cross-coupling methods by utilizing readily available 4-substituted nitrobenzenes as the primary feedstock. The process leverages a sophisticated catalytic hydrogenation step followed by an acid-catalyzed rearrangement to construct the biphenyl backbone with exceptional atom economy. For R&D directors and procurement specialists, this patent offers a compelling alternative to expensive palladium-catalyzed cross-couplings, promising substantial reductions in raw material costs while maintaining rigorous purity standards. The ability to produce these valuable intermediates under relatively mild conditions (45-120°C) and moderate pressures (1.0-2.0 MPa) underscores the industrial viability of this approach for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,2'-diaminobiphenyl derivatives has relied heavily on transition-metal catalyzed cross-coupling reactions such as the Suzuki-Miyaura coupling or oxidative homocoupling of aryl halides. These conventional routes often necessitate the use of expensive and sensitive aryl boronic acids or aryl iodides/bromides as starting materials, which significantly inflates the overall production cost. Furthermore, these methods typically generate substantial amounts of stoichiometric metal waste and require rigorous purification steps to remove trace palladium residues, which is a critical concern for pharmaceutical applications. The multi-step nature of synthesizing the requisite coupling partners adds complexity to the supply chain, increasing lead times and introducing potential points of failure in large-scale manufacturing. Additionally, the use of strong bases and high temperatures in some oxidative coupling protocols can lead to poor selectivity and the formation of difficult-to-remove regioisomers, compromising the quality of the final active ingredient or polymer precursor.
The Novel Approach
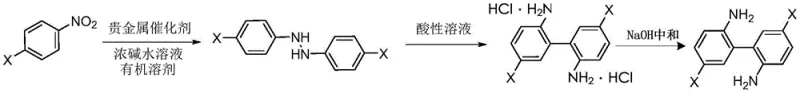
In stark contrast, the method disclosed in CN111574380A streamlines the synthesis by directly coupling two molecules of 4-substituted nitrobenzene through a reductive pathway. This innovative route bypasses the need for pre-functionalized coupling partners, utilizing simple nitro compounds that are commodity chemicals available in bulk quantities. The core transformation involves a catalytic hydrogenation that generates a 1,2-diphenylhydrazine intermediate, which subsequently undergoes a benzidine-type rearrangement in an acidic medium to form the desired biphenyl structure. This telescoped approach not only reduces the number of unit operations but also improves the overall mass balance of the process. By integrating the reduction and rearrangement steps, the technology minimizes solvent usage and waste generation, aligning perfectly with modern green chemistry principles. The result is a robust, scalable process that delivers high-purity products with a simplified workflow, making it an ideal candidate for cost reduction in fine chemical intermediate manufacturing.
Mechanistic Insights into Catalytic Hydrogenation and Rearrangement
The heart of this synthetic strategy lies in the precise control of the catalytic hydrogenation step, where 4-substituted nitrobenzene is converted into a 1,2-diphenylhydrazine derivative. This transformation is facilitated by a supported precious metal catalyst, such as platinum, palladium, ruthenium, or rhodium, dispersed on carriers like activated carbon or alumina. The presence of a strong base, specifically sodium hydroxide, is crucial for promoting the condensation of the reduced nitro species into the hydrazine linkage rather than stopping at the aniline stage. The reaction is conducted in organic solvents like toluene or xylene under hydrogen pressure ranging from 1.0 to 2.0 MPa, ensuring complete conversion while maintaining safety. Following the hydrogenation, the reaction mixture is subjected to an acidic environment containing hydrochloric or sulfuric acid along with an ammonium salt. This acidic medium triggers a [5,5]-sigmatropic rearrangement, effectively cleaving the N-N bond of the hydrazine and reforming C-C bonds between the aromatic rings to yield the 2,2'-diaminobiphenyl skeleton. This mechanistic pathway is highly selective, minimizing the formation of ortho-para isomers that often plague electrophilic aromatic substitution routes.
Impurity control is meticulously managed through the subsequent workup and purification stages described in the patent. After the rearrangement, the crude product exists as a hydrochloride salt, which is isolated by filtration. This salt formation step inherently purifies the molecule by excluding neutral organic impurities that remain in the mother liquor. The free base is then regenerated by neutralization with sodium hydroxide to a pH of 7-8.5, followed by a dual-solvent purification system. Dissolution in ethyl acetate removes insoluble inorganic salts and polymeric byproducts, while subsequent recrystallization from toluene ensures the removal of closely related structural analogues. This rigorous purification protocol guarantees that the final 4,4'-disubstituted-2,2'-diaminobiphenyl achieves a purity level exceeding 99%, as confirmed by NMR analysis in the patent examples. Such high purity is essential for downstream applications in polyimide synthesis where trace impurities can adversely affect the thermal and mechanical properties of the resulting polymer films.
How to Synthesize 4,4'-Disubstituted-2,2'-Diaminobiphenyl Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and safety. The process begins with the loading of the nitrobenzene substrate, solvent, and base into a pressure vessel, followed by the addition of the heterogeneous catalyst. Strict exclusion of oxygen via nitrogen purging is mandatory before introducing hydrogen to prevent explosive mixtures. The temperature must be ramped gradually to the target range of 45-120°C depending on the specific substituent, as electron-withdrawing groups may require different activation energies compared to electron-donating alkyl groups. Once the hydrogenation is complete, the catalyst is filtered off for regeneration, and the filtrate is transferred to the rearrangement reactor. Here, the controlled addition of the acid-ammonium mixture is critical to manage the exotherm and ensure uniform crystal formation of the hydrochloride salt. For detailed operational parameters and specific stoichiometric ratios tailored to your specific substrate, please refer to the standardized synthesis steps provided below.
- Perform catalytic hydrogenation of 4-substituted nitrobenzene using a precious metal catalyst (Pt, Pd, Ru, etc.) in organic solvent with NaOH at 45-120°C and 1.0-2.0 MPa pressure to form 1,2-diphenylhydrazine.
- React the hydrazine intermediate in a mixed acidic solution of strong acid (HCl/H2SO4) and ammonium salt at 20-60°C to induce rearrangement into the crude diaminobiphenyl hydrochloride.
- Neutralize the crude salt with NaOH, purify the free base via ethyl acetate dissolution and toluene recrystallization, and finally convert to the hydrochloride salt using concentrated HCl.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this reductive coupling technology offers transformative benefits for supply chain stability and cost management. The primary driver for cost reduction is the substitution of expensive aryl halides and boronic acids with commodity nitrobenzenes, which are produced on a massive global scale and exhibit stable pricing trends. This shift in raw material sourcing significantly de-risks the supply chain against fluctuations in the palladium market or shortages of specialized coupling partners. Furthermore, the heterogeneous nature of the catalyst allows for straightforward filtration and reuse, eliminating the need for costly metal scavenging resins or complex extraction processes typically associated with homogeneous catalysis. The ability to recycle the catalyst multiple times without loss of activity translates directly into lower operating expenses and a reduced environmental footprint, which is increasingly important for meeting corporate sustainability goals. Additionally, the solvents used, such as toluene and ethyl acetate, are easily recoverable through distillation, further enhancing the economic efficiency of the process by minimizing waste disposal costs.
- Cost Reduction in Manufacturing: The elimination of stoichiometric organometallic reagents and the use of cheap nitro-starting materials drastically lower the bill of materials. The recovery and reuse of the precious metal catalyst mean that the effective cost of the catalyst per kilogram of product is negligible over time. Moreover, the simplified purification train reduces solvent consumption and energy usage associated with multiple chromatographic separations, leading to substantial overall cost savings in the manufacturing of high-purity fine chemical intermediates.
- Enhanced Supply Chain Reliability: By relying on widely available 4-substituted nitrobenzenes, manufacturers can secure long-term supply contracts with multiple vendors, reducing dependency on single-source suppliers of exotic reagents. The robustness of the reaction conditions, which tolerate a variety of substituents including halogens and alkyl groups, allows for flexible production scheduling across different product grades. This flexibility ensures consistent delivery timelines and mitigates the risk of production stoppages due to raw material unavailability, thereby strengthening the reliability of the supply chain for critical downstream customers.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard hydrogenation equipment and acid-resistant reactors found in most multipurpose chemical plants. The aqueous workup and salt precipitation steps simplify the isolation of the product, avoiding the need for complex distillation of high-boiling intermediates. From an environmental standpoint, the reduction in heavy metal waste and the ability to recycle organic solvents align with strict regulatory frameworks regarding effluent discharge. This compliance facilitates smoother permitting processes for capacity expansion and ensures long-term operational continuity without regulatory interruptions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this reductive coupling technology. These answers are derived directly from the experimental data and claims presented in patent CN111574380A, providing a reliable foundation for feasibility assessments. Understanding these details is crucial for R&D teams evaluating the transfer of this technology from the laboratory to pilot or commercial scale. The responses cover aspects of catalyst life, purity profiles, and substrate scope, offering a comprehensive overview of the process capabilities.
Q: What are the key advantages of the reductive coupling method over Suzuki coupling for diaminobiphenyls?
A: The reductive coupling method utilizes inexpensive 4-substituted nitrobenzenes as starting materials instead of complex aryl halides required for Suzuki coupling. It eliminates the need for stoichiometric organometallic reagents and reduces heavy metal waste, significantly lowering raw material costs and simplifying the purification process while maintaining high yields above 90%.
Q: Can the precious metal catalyst be recovered and reused in this process?
A: Yes, the patent explicitly states that the solid precious metal catalyst (such as Pt/C, Pd/C, or Rh/Al2O3) can be filtered off after the hydrogenation step and reused multiple times without significant loss of catalytic activity, which is a critical factor for reducing production costs in large-scale manufacturing.
Q: What purity levels can be achieved with the described recrystallization protocol?
A: By employing a specific purification sequence involving ethyl acetate dissolution followed by toluene recrystallization, the process consistently achieves final product purity greater than 99%, meeting the stringent quality specifications required for high-performance polymer and pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Disubstituted-2,2'-Diaminobiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes for complex biaryl amines in the development of next-generation materials and therapeutics. Our technical team has extensively analyzed the reductive coupling methodology described in CN111574380A and possesses the expertise to adapt this chemistry for diverse substrates beyond those explicitly listed in the patent. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from gram-scale optimization to tonnage manufacturing is seamless and risk-mitigated. Our facilities are equipped with state-of-the-art high-pressure hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, including residual metal analysis and detailed impurity profiling. We are committed to delivering high-purity fine chemical intermediates that meet the exacting standards of the global pharmaceutical and specialty polymer industries.
We invite you to collaborate with us to leverage this cost-effective technology for your specific project needs. Whether you require custom synthesis of novel derivatives or reliable supply of standard grades, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner dedicated to innovation, quality, and long-term value creation through advanced chemical manufacturing solutions.
