Advanced Manufacturing of Dihydroisoxazole Intermediates for High-Efficiency Herbicide Production
The global agrochemical sector is continuously demanding more efficient and sustainable pathways for producing high-performance herbicides, particularly those based on the sulfonylurea and topramezone classes. A pivotal breakthrough in this domain is documented in patent CN114644602A, which details a novel preparation method for dihydroisoxazole compounds. These specific chemical structures serve as critical building blocks, or key intermediates, in the synthesis of advanced herbicidal agents that exhibit superior activity compared to legacy products like acetochlor. For R&D Directors and Technical Procurement Managers, understanding the nuances of this patent is essential, as it offers a pathway to significantly enhance the purity and yield of these vital intermediates while simultaneously addressing long-standing safety and cost concerns inherent in traditional manufacturing processes. This report analyzes the technical merits and commercial implications of adopting this refined synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-halogenated 5,5-dimethyl-4,5-dihydroisoxazole compounds has been plagued by significant economic and safety inefficiencies that burden the supply chain. Conventional literature often describes routes starting from glyoxylic acid, which requires reaction with hydroxylamine hydrochloride followed by substitution and decarboxylation using bromine. This traditional approach is fundamentally flawed because it necessitates the use of more than two equivalents of expensive bromine, yet the utilization rate of this hazardous reagent is shockingly low, often hovering around only twenty-five percent. Furthermore, alternative methods disclosed in earlier patents involve the use of 3-methyl-2-butenal, a raw material that itself requires high-temperature isomerization of 3-methyl-3-butenal, thereby adding unnecessary energy costs and complexity to the upstream supply chain. These legacy processes not only inflate the cost of goods sold but also introduce substantial environmental and safety liabilities due to the handling of elemental halogens and harsh reaction conditions.
The Novel Approach
In stark contrast, the methodology outlined in the subject patent introduces a paradigm shift by utilizing 3-methyl-3-butenal and hydroxylamine compounds as the primary raw materials under the influence of a specialized catalyst system. This new route bypasses the need for dangerous elemental chlorine or bromine gas, opting instead for safer hypochlorite salts which are easier to handle and store in an industrial setting. The process is designed to achieve high conversion rates and selectivity, directly translating to reduced waste generation and higher overall throughput for manufacturing facilities. By streamlining the synthetic steps and employing more accessible starting materials, this approach effectively decouples the production cost from the volatility of expensive halogen markets. For procurement teams, this represents a strategic opportunity to stabilize the cost base of agrochemical intermediate manufacturing while improving the safety profile of the production site.
Mechanistic Insights into Acid-Base Composite Catalyzed Cyclization
The core innovation of this synthesis lies in the sophisticated use of an acid-base composite catalyst to drive the cyclization reaction between 3-methyl-3-butenal and hydroxamine derivatives. Unlike simple acid or base catalysis which may lead to side reactions or polymerization of the aldehyde, this dual-system utilizes specific pairings such as trifluoroacetic acid combined with 2,3-indoline or diphenyl phosphoric acid with N-methylaniline. This precise balancing of acidity and basicity facilitates the nucleophilic attack and subsequent ring closure to form the 5,5-dimethyl-4,5-dihydroisoxazole structure with exceptional efficiency. The reaction proceeds smoothly at mild temperatures ranging from zero to thirty degrees Celsius, which minimizes thermal degradation of sensitive intermediates and reduces the energy load on the reactor systems. Such mechanistic control ensures that the impurity profile remains clean, a critical factor for downstream processing in pharmaceutical and agrochemical applications where strict regulatory limits on byproducts are enforced.
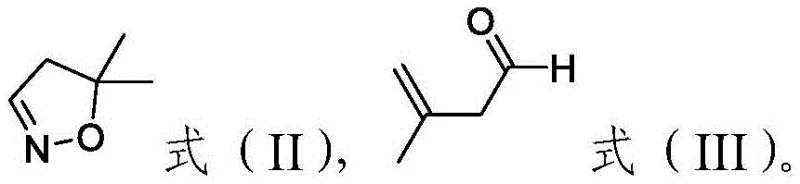
Following the formation of the dihydroisoxazole ring, the subsequent halogenation step is equally critical for achieving the final target molecule required for herbicide synthesis. The patent specifies the use of hypochlorite salts, such as sodium hypochlorite or calcium hypochlorite, in the presence of an inorganic acid like hydrochloric acid. This in situ generation of the halogenating species allows for a controlled reaction environment that avoids the violent exotherms associated with direct halogen gas addition. The reaction is typically conducted in solvents such as alcohol nitriles or aromatic hydrocarbons at temperatures between zero and ten degrees Celsius to maximize selectivity. This careful modulation of reaction conditions ensures that the chlorination occurs specifically at the desired position on the isoxazole ring without over-chlorination or ring opening, thereby preserving the structural integrity of the valuable intermediate.
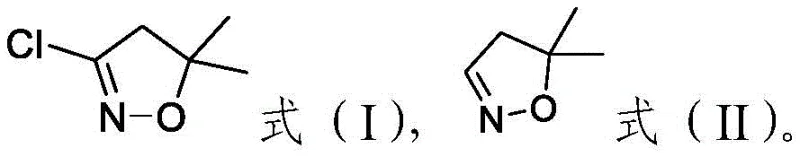
How to Synthesize 3-chloro-5,5-dimethyl-4,5-dihydroisoxazole Efficiently
Implementing this synthesis route requires precise adherence to the molar ratios and temperature controls specified in the technical disclosure to ensure optimal yield and purity. The process begins with the preparation of the intermediate through the catalyzed reaction of the aldehyde and hydroxylamine source, followed by purification via rectification to remove unreacted starting materials. Once the high-purity intermediate is secured, it is subjected to the halogenation protocol using aqueous hypochlorite solutions under acidic conditions. Detailed standard operating procedures regarding mixing speeds, addition rates, and quenching protocols are essential for maintaining safety and consistency at scale. For a comprehensive breakdown of the standardized synthesis steps and specific parameter settings, please refer to the technical guide below.
- React 3-methyl-3-butenal with hydroxylamine compounds using an acid-base composite catalyst at 0-30°C to form the dihydroisoxazole ring.
- Purify the intermediate 5,5-dimethyl-4,5-dihydroisoxazole via rectification to ensure high purity before halogenation.
- Perform halogenation using sodium hypochlorite and inorganic acid at controlled low temperatures to yield the final chlorinated product.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the bottom line and supply continuity, the adoption of this patented methodology offers profound strategic benefits that extend beyond mere technical feasibility. The shift away from elemental bromine and complex isomerization feedstocks directly addresses the volatility often seen in the pricing of specialty halogenated intermediates. By utilizing commodity-grade hypochlorites and more stable aldehyde precursors, manufacturers can insulate their production costs from the fluctuations of the broader halogen market. This stability is crucial for long-term contract negotiations with agrochemical formulators who demand consistent pricing over multi-year periods. Furthermore, the improved safety profile reduces the insurance and compliance overhead associated with handling hazardous gases, contributing to a leaner operational expenditure model.
- Cost Reduction in Manufacturing: The elimination of expensive bromine reagents, which historically suffered from poor atom economy, results in a drastic reduction in raw material expenditures. Additionally, the high conversion rates achieved through the acid-base catalyst system minimize the loss of valuable starting materials into waste streams, further enhancing the overall process efficiency. The ability to operate at near-ambient temperatures also translates to significant energy savings compared to high-temperature isomerization routes required by older technologies. These cumulative factors create a robust framework for substantial cost savings without compromising on the quality of the final agrochemical intermediate.
- Enhanced Supply Chain Reliability: Sourcing elemental bromine or chlorine gas often involves complex logistics and stringent regulatory permits that can bottleneck production schedules. By switching to aqueous hypochlorite solutions and stable organic acids, the supply chain becomes more resilient and less susceptible to transportation disruptions or regulatory tightening. The raw materials required for this process are widely available from multiple global suppliers, reducing the risk of single-source dependency. This diversification of the supply base ensures continuous operation even during regional shortages, providing a competitive edge in meeting delivery commitments to downstream herbicide manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic gas emissions make this process inherently easier to scale from pilot plant to full commercial production. Waste treatment is simplified as the effluent primarily contains salt solutions rather than heavy metal contaminants or persistent organic halides, aligning with increasingly strict environmental regulations. The high selectivity of the reaction reduces the burden on downstream purification units, allowing for faster batch turnover and higher facility utilization rates. This scalability ensures that the technology can meet growing global demand for high-efficiency herbicides while maintaining a sustainable environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dihydroisoxazole synthesis pathway. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on safety, cost, and scalability. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios. The answers reflect a commitment to transparency and technical accuracy based on the available intellectual property.
Q: Why is the hypochlorite method safer than traditional bromination?
A: Traditional methods often utilize elemental bromine or chlorine gas, which pose significant safety hazards and require specialized containment. The patented method employs aqueous hypochlorite solutions, drastically reducing operational risks and simplifying waste treatment protocols.
Q: What represents the primary cost advantage of this synthesis route?
A: The process eliminates the need for expensive bromine reagents, which historically suffered from low utilization rates. Additionally, it utilizes 3-methyl-3-butenal, a more economically accessible raw material compared to isomerized alternatives used in older pathways.
Q: Can this process be scaled for industrial herbicide production?
A: Yes, the reaction conditions operate within mild temperature ranges (0-30°C for cyclization, 0-10°C for halogenation) and utilize standard solvents like alcohol nitriles or aromatic hydrocarbons, making it highly amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-chloro-5,5-dimethyl-4,5-dihydroisoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of this patent can be realized in a real-world industrial setting. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of dihydroisoxazole intermediate meets the exacting standards required for modern agrochemical formulations. Our commitment to quality assurance means that your supply chain is backed by data-driven consistency and reliability.
We invite you to collaborate with us to optimize your sourcing strategy for these critical herbicide intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to reach out to request specific COA data and route feasibility assessments to verify how this advanced synthesis method can enhance your product portfolio. Let us help you secure a competitive advantage through superior chemistry and supply chain excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
