Optimizing Gimeracil Production: Advanced Synthetic Routes for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for critical anticancer agents, and the preparation of 5-chloro-4-hydroxyl-2(1H)-pyridone, widely known as Gimeracil, represents a significant focal point in medicinal chemistry. As detailed in patent CN1915976B, a novel methodology has been established that addresses the longstanding inefficiencies associated with traditional heterocyclic synthesis. This compound serves as a vital biological regulator and dihydropyrimidine dehydrogenase inhibitor, playing an indispensable role in the efficacy of fluorouracil-based chemotherapy regimens. The technical breakthrough described herein offers a streamlined approach that not only enhances reaction yields but also significantly improves the impurity profile of the final product. For R&D teams evaluating potential manufacturing partners, understanding the nuances of this specific patent data is crucial for ensuring the consistent supply of high-quality active pharmaceutical ingredients. The structural integrity of the target molecule, as depicted below, requires precise control over substitution patterns to maintain biological activity.
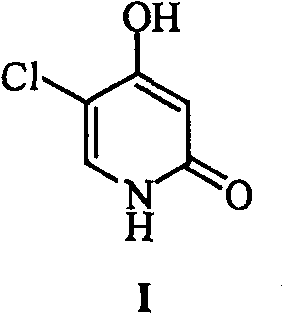
Historically, the synthesis of this pyridone derivative has been plagued by cumbersome multi-step procedures that result in suboptimal overall recovery rates and operational hazards. Conventional methods, such as those utilizing 4-nitropyridine N-oxide or 3-oxo-1,5-ethyl glutarate as starting materials, often necessitate sealed tube reactions and involve up to seven distinct chemical transformations. These legacy processes frequently suffer from total recovery rates as low as 12% to 26%, creating substantial material waste and driving up the cost of goods sold for downstream drug manufacturers. Furthermore, the reliance on harsh conditions and complex purification steps, such as column chromatography, renders these methods impractical for ton-scale commercial production. In contrast, the novel approach outlined in the patent data leverages a more direct cyclization and functionalization strategy that drastically reduces the number of unit operations required. By shifting the synthetic logic towards a selective chlorination followed by a controlled hydrolysis, the new route eliminates several bottleneck steps that traditionally limit throughput and scalability in fine chemical manufacturing.
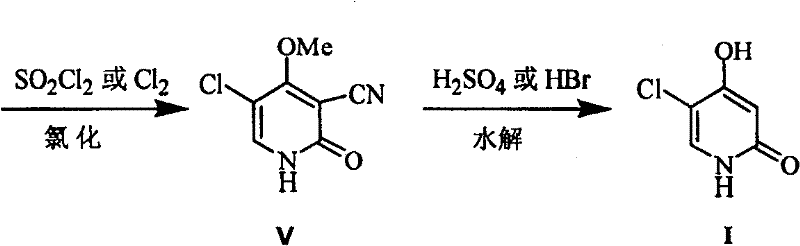
Mechanistic Insights into Sulfuryl Chloride-Mediated Cyclization
The core innovation of this synthetic pathway lies in the precise mechanistic control exerted during the chlorination of the 3-cyano-4-methoxy-2(1H)-pyridone precursor. The reaction utilizes sulfuryl chloride as both a chlorinating agent and an oxidant, operating effectively within a glacial acetic acid solvent system to facilitate the formation of the 2,5-dichloro-3-cyano-4-methoxypyridine intermediate. Critical to the success of this transformation is the maintenance of light-shielded conditions, as exposure to ambient fluorescence can accelerate the decomposition of sulfuryl chloride, leading to increased reagent consumption and the formation of undesirable by-products. The temperature must be rigorously maintained between 45°C and 50°C to balance reaction kinetics with selectivity, ensuring that the chlorine atoms are installed at the correct positions on the pyridone ring without causing ring degradation. This level of mechanistic precision allows for the isolation of the intermediate in yields exceeding 80%, providing a robust foundation for the subsequent hydrolysis step that generates the final active moiety.
Following the chlorination, the hydrolysis mechanism is engineered to simultaneously achieve demethylation and decarboxylation while preserving the sensitive chloro-substituent at the 5-position. The process employs concentrated sulfuric acid or hydrobromic acid at elevated temperatures ranging from 100°C to 125°C to cleave the methoxy group and convert the cyano functionality into the requisite hydroxyl and carbonyl structures. A key advantage of this specific acidic environment is its ability to suppress the formation of colored impurities that often plague hydrolysis reactions involving nitrogen-containing heterocycles. By optimizing the acid concentration to between 60% and 80% for sulfuric acid, the reaction drives the equilibrium towards the desired 5-chloro-4-hydroxyl-2(1H)-pyridone product with a purity greater than 99%. This rigorous control over the hydrolysis parameters ensures that related substances remain below 0.5%, meeting the stringent quality standards required for pharmaceutical intermediates intended for human consumption.
How to Synthesize 5-chloro-4-hydroxyl-2(1H)-pyridone Efficiently
Implementing this synthesis requires careful attention to the addition rates and thermal profiles described in the experimental embodiments to maximize safety and yield. The procedure begins with the preparation of the chlorinating solution, followed by the controlled addition of the solid precursor to manage exothermicity and ensure homogeneous mixing throughout the reaction vessel. Operators must adhere to the specified molar ratios, typically favoring a slight excess of sulfuryl chloride to drive the reaction to completion without generating excessive waste. The subsequent workup involves solvent recovery and crystallization steps that are designed to be easily integrated into standard filtration and drying equipment found in modern chemical plants. Detailed standardized synthetic steps see the guide below for exact operational parameters.
- Prepare a reaction mixture of sulfuryl chloride and glacial acetic acid, maintaining light-shielded conditions to prevent reagent decomposition.
- Slowly add 3-cyano-4-methoxy-2(1H)-pyridone to the chlorinating solution at 45-50°C to form the dichloro intermediate with high selectivity.
- Hydrolyze the resulting intermediate using concentrated sulfuric acid or hydrobromic acid at elevated temperatures to achieve final deprotection and cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits regarding cost stability and vendor reliability. The elimination of multiple synthetic steps and the avoidance of expensive transition metal catalysts fundamentally alter the cost structure of producing this key anticancer intermediate. By simplifying the process flow, manufacturers can reduce the burden on quality control laboratories and minimize the risk of batch failures that often disrupt supply continuity in complex chemical manufacturing. This efficiency gain allows suppliers to offer more competitive pricing models while maintaining healthy margins, which is essential for long-term partnership viability in the volatile pharmaceutical market. Furthermore, the use of commodity chemicals like glacial acetic acid and sulfuric acid reduces dependency on specialized reagents that may be subject to geopolitical supply constraints or price volatility.
- Cost Reduction in Manufacturing: The streamlined nature of this synthetic route significantly lowers the operational expenditure associated with producing Gimeracil intermediates by removing the need for costly purification techniques like column chromatography. By achieving high crude yields directly from the reaction, the consumption of solvents and energy for separation processes is drastically reduced, leading to substantial overall cost savings. Additionally, the high selectivity of the chlorination step minimizes the generation of waste streams that require expensive treatment and disposal, further enhancing the economic viability of the process. These cumulative efficiencies enable a more aggressive pricing strategy for the final API, benefiting the entire value chain from raw material suppliers to end-product manufacturers.
- Enhanced Supply Chain Reliability: Utilizing widely available and stable reagents such as sulfuryl chloride and acetic acid ensures that production schedules are not held hostage by the scarcity of exotic catalysts or specialized starting materials. The robustness of the reaction conditions means that manufacturing can proceed with minimal downtime for equipment cleaning or maintenance, thereby increasing the overall asset utilization rate of the production facility. This reliability is critical for pharmaceutical companies that require just-in-time delivery of intermediates to meet their own regulatory filing deadlines and market launch windows. Consequently, partners adopting this technology can guarantee more consistent lead times and higher fill rates for their global customer base.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, moving seamlessly from kilogram-level laboratory batches to multi-ton annual commercial production without requiring fundamental changes to the reaction engineering. The absence of heavy metal residues simplifies the environmental compliance landscape, reducing the regulatory burden associated with wastewater treatment and residual solvent limits in the final product. This green chemistry advantage aligns with the increasing corporate sustainability goals of major pharmaceutical buyers who prioritize suppliers with lower carbon footprints and safer operational profiles. As a result, this method future-proofs the supply chain against tightening environmental regulations while supporting sustainable growth in the production of essential medicines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and sourcing of this specific pyridone derivative. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance for industry stakeholders. Understanding these details helps decision-makers evaluate the feasibility of integrating this intermediate into their existing drug development pipelines. Please review the specific answers below for clarification on process capabilities and quality metrics.
Q: What are the critical reaction conditions for the chlorination step?
A: The chlorination reaction requires strict light-shielding conditions and temperature control between 45°C and 50°C to prevent sulfuryl chloride decomposition and ensure high yield of the dichloro intermediate.
Q: How does this method improve product purity compared to conventional routes?
A: By utilizing a specific hydrolysis protocol with concentrated sulfuric or hydrobromic acid, this method effectively removes methoxy and cyano groups while minimizing side reactions, achieving purity levels greater than 99%.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the process avoids expensive catalysts and uses common solvents like glacial acetic acid, making it highly scalable and cost-effective for commercial manufacturing of anticancer intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-chloro-4-hydroxyl-2(1H)-pyridone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of Gimeracil in the global oncology market and have invested heavily in mastering the complex chemistry required for its efficient production. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the fluctuating demands of international pharmaceutical clients with ease. We operate state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of intermediate we supply meets the highest standards for safety and efficacy. Our commitment to technical excellence allows us to navigate the intricacies of heterocyclic synthesis with precision, delivering products that facilitate smoother regulatory approvals for our partners.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements and timeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into how our optimized processes can reduce your overall cost of goods. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique formulation needs. Let us collaborate to bring life-saving medications to patients faster and more efficiently through superior chemical manufacturing solutions.
