Revolutionizing Chiral Ligand Production: A Safe and Scalable Route for High-Purity TADDOLs
Revolutionizing Chiral Ligand Production: A Safe and Scalable Route for High-Purity TADDOLs
The landscape of asymmetric synthesis is undergoing a significant transformation driven by the urgent need for safer, more efficient, and cost-effective manufacturing processes for chiral catalysts. Patent CN107721969B, published in early 2020, introduces a groundbreaking preparation method for chiral catalyst ligands known as TADDOLs (α,α,α',α'-tetraaryl-1,3-dioxolane-4,5-dimethanols). This technology addresses critical bottlenecks in the existing supply chain by replacing hazardous reagents and complex purification steps with a streamlined, robust protocol. For R&D directors and procurement managers alike, this innovation represents a pivotal shift towards sustainable and economically viable production of high-value chiral intermediates. By utilizing cheap and easily obtainable raw materials such as chiral diethyl tartrate and isopropyl magnesium bromide, the patent outlines a pathway that drastically lowers the barrier to entry for high-quality ligand manufacturing while ensuring exceptional stereochemical integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of TADDOL ligands has been plagued by severe operational hazards and inefficiencies that hinder large-scale adoption. Traditional methods often rely on the pre-preparation of aromatic Grignard reagents in large excess, typically requiring more than 10 equivalents, which leads to significant waste and difficult post-reaction handling. Furthermore, alternative routes utilizing n-butyllithium necessitate cryogenic conditions as low as -78°C, imposing stringent equipment requirements and posing substantial fire and explosion risks due to the pyrophoric nature of the reagent. The purification processes associated with these legacy methods frequently depend on column chromatography, a technique that is notoriously difficult to scale and results in low overall yields, often ranging from merely 17% to 50% in reported literature. These factors combine to create a supply chain vulnerability characterized by high costs, inconsistent quality, and potential safety incidents during production.
The Novel Approach
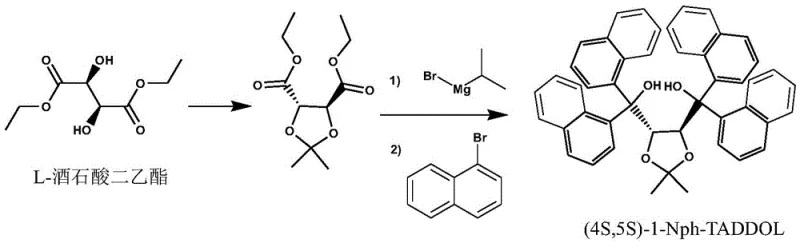
In stark contrast, the methodology disclosed in CN107721969B offers a sophisticated solution that bypasses these traditional pitfalls through an innovative in-situ generation strategy. Instead of isolating dangerous Grignard reagents, the process reacts aromatic hydrocarbon bromides directly with isopropyl magnesium bromide under controlled, mild conditions before introducing the chiral tartrate intermediate. This approach not only simplifies the operational workflow but also significantly enhances safety profiles by eliminating the need for ultra-low temperature reactions and hazardous organolithium species. The result is a highly efficient synthesis that achieves yields exceeding 90% in specific examples, such as the 95.9% yield observed for (4S,5S)-1-Nph-TADDOL. By shifting the paradigm from complex, hazardous batch processes to a streamlined, controllable reaction sequence, this technology enables reliable chiral ligand supplier operations to meet the rigorous demands of the pharmaceutical industry with unprecedented consistency.
Mechanistic Insights into Isopropyl Magnesium Bromide Mediated Arylation
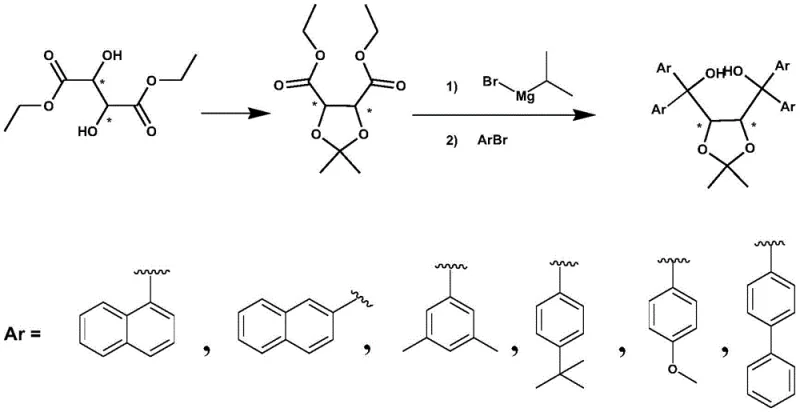
The core of this technological advancement lies in the precise mechanistic control of the nucleophilic addition step, which preserves the chirality of the starting material while installing bulky aryl groups with high fidelity. The reaction initiates with the formation of a reactive aryl-magnesium species via the interaction of the aromatic bromide and isopropyl magnesium bromide in a solvent like tetrahydrofuran. This transmetallation or exchange process generates the active nucleophile in situ, which then attacks the ester carbonyls of the O,O-isopropylidene diethyl tartrate intermediate. The steric environment created by the isopropylidene protecting group plays a crucial role in directing the approach of the nucleophile, ensuring that the stereochemical information encoded in the L- or D-tartrate backbone is faithfully transmitted to the final TADDOL structure. This mechanism avoids the racemization risks often associated with harsher basic conditions or prolonged exposure to reactive organometallics, thereby securing the high enantiomeric excess (ee) values required for effective asymmetric catalysis.
Furthermore, the impurity profile of the final product is meticulously managed through the choice of reagents and workup procedures. The use of isopropyl magnesium bromide minimizes side reactions such as self-coupling of the aryl halide, a common issue in traditional Grignard preparations that leads to difficult-to-remove by-products. Following the reaction, the quenching with aqueous ammonium chloride effectively neutralizes residual magnesium species without inducing exothermic runaways, a frequent hazard in acid quenches of excess Grignard reagents. The subsequent extraction and recrystallization steps leverage the distinct solubility properties of the TADDOL product to remove trace impurities, resulting in a final material with chemical purity greater than 99%. This level of control over the杂质谱 (impurity profile) is essential for R&D teams developing sensitive catalytic processes where trace metals or organic contaminants could poison the catalyst or affect downstream drug substance quality.
How to Synthesize Chiral TADDOL Ligands Efficiently
The practical implementation of this synthesis route involves a straightforward two-step sequence that begins with the protection of the chiral diol and concludes with the double nucleophilic addition. The first step utilizes Lewis acid catalysis to form the acetal protection group, a reaction that is robust and easily monitored by TLC. The second step, the core arylation, requires careful temperature control during the addition of the Grignard component but proceeds smoothly upon heating to moderate temperatures. This operational simplicity makes the technology accessible for both laboratory-scale optimization and pilot plant campaigns. For detailed standard operating procedures and specific parameter settings regarding reagent grades and agitation speeds, please refer to the standardized synthesis guide below.
- Protect chiral diethyl tartrate with 2,2-dimethoxypropane and triethyl orthoformate using a Lewis acid catalyst to form O,O-isopropylidene diethyl tartrate.
- React aromatic hydrocarbon bromide with isopropyl magnesium bromide in THF at low temperature, then add the protected tartrate intermediate and heat to reflux.
- Quench the reaction with ammonium chloride, extract with ethyl acetate, and purify the crude product via recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages that directly impact the bottom line and supply chain resilience for fine chemical manufacturers. By transitioning away from exotic, hazardous reagents like n-butyllithium and complex purification techniques like column chromatography, the process inherently reduces the cost of goods sold (COGS). The reliance on bulk commodities such as diethyl tartrate and simple aromatic bromides ensures that raw material costs remain stable and predictable, shielding the supply chain from the volatility often seen with specialized organometallic reagents. Moreover, the elimination of dangerous unit operations reduces the capital expenditure required for specialized containment and cooling infrastructure, allowing for more flexible manufacturing arrangements.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the drastic simplification of the workflow and the improvement in overall yield. By avoiding the preparation of isolated Grignard reagents and utilizing a one-pot style addition strategy, labor hours and solvent consumption are significantly reduced. The high yields achieved, often surpassing 90%, mean that less raw material is wasted per kilogram of finished product, leading to substantial cost savings in pharmaceutical intermediates manufacturing. Additionally, the replacement of chromatographic purification with simple recrystallization removes a major cost center, as chromatography resins and the associated solvent volumes represent a significant expense in traditional fine chemical production.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly improved by the use of widely available starting materials that are not subject to the same regulatory and logistical constraints as pyrophoric reagents. Isopropyl magnesium bromide is a stable, commercially available solution that can be sourced from multiple suppliers, reducing the risk of single-source dependency. The mild reaction conditions also mean that production is less susceptible to disruptions caused by equipment failure or utility fluctuations, as the process does not rely on maintaining cryogenic temperatures. This robustness ensures that delivery schedules for high-purity chiral ligands can be met consistently, supporting the just-in-time manufacturing models of modern pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than conventional methods, aligning with the increasing demand for green chemistry solutions. The reduction in solvent usage, particularly the avoidance of large volumes required for column chromatography, simplifies waste treatment and lowers disposal costs. The process is inherently safer, minimizing the risk of thermal runaways or fires, which facilitates easier permitting and regulatory compliance for commercial scale-up of complex polymer additives or pharmaceutical intermediates. This scalability ensures that the technology can grow with market demand, from kilogram quantities for R&D to multi-ton production for commercial API synthesis without requiring fundamental process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this TADDOL synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on safety, quality, and operational feasibility. Understanding these details is crucial for technical teams evaluating the integration of this method into their existing manufacturing portfolios.
Q: Why is this new TADDOL preparation method safer than conventional routes?
A: This method eliminates the need for preparing dangerous aromatic Grignard reagents separately or using highly flammable n-butyllithium at cryogenic temperatures (-78°C), significantly reducing industrial safety risks.
Q: What purity levels can be achieved with this synthesis route?
A: Through optimized reaction conditions and simple recrystallization, the process consistently delivers chiral TADDOL ligands with chemical purity exceeding 99% and ee values greater than 99%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the use of bulk chemicals, mild reaction temperatures, and the avoidance of complex column chromatography purification makes this method highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral TADDOL Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthesis route for the global supply of asymmetric catalysts. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with the necessary safety systems and reactor configurations to handle Grignard chemistry efficiently while maintaining stringent purity specifications. With our rigorous QC labs and commitment to quality assurance, we guarantee that every batch of chiral ligand meets the exacting standards required for sensitive asymmetric transformations.
We invite forward-thinking organizations to collaborate with us to leverage this advanced technology for their specific applications. Whether you require custom derivatives of TADDOLs or support in optimizing the process for your unique substrate, our technical team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to reach out for specific COA data and route feasibility assessments to see how we can drive value and efficiency in your supply chain together.
