Advanced Synthesis of High-Purity 1-Bromopyrene for Commercial OLED Material Production
Advanced Synthesis of High-Purity 1-Bromopyrene for Commercial OLED Material Production
The rapid expansion of the organic light-emitting diode (OLED) industry has created an urgent demand for high-performance intermediates that meet stringent purity specifications. Patent CN108299149B introduces a groundbreaking synthesis method for 1-bromopyrene, a critical building block in the fabrication of advanced optoelectronic materials. This technology addresses the longstanding challenges associated with traditional bromination processes, specifically targeting the issues of environmental toxicity and low regioselectivity that have plagued manufacturers for years. By utilizing dibromohydantoin as a controlled brominating agent instead of hazardous elemental bromine, the process achieves a remarkable yield of greater than 95% while maintaining exceptional operational safety. Furthermore, the integration of a novel picric acid complexation purification step ensures that the final product reaches a purity level of ≥99.5%, which is essential for preventing defects in high-resolution display panels. For global procurement teams and R&D directors, this patent represents a pivotal shift towards more sustainable and efficient electronic chemical manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 1-bromopyrene has relied heavily on direct bromination using elemental bromine or indirect methods involving hydrobromic acid and oxidants. These conventional pathways are fraught with significant technical and environmental drawbacks that hinder efficient commercial scale-up of complex organic intermediates. The use of liquid bromine generates substantial amounts of corrosive hydrogen bromide gas, necessitating expensive scrubbing systems and posing severe safety risks to plant personnel. Moreover, the high reactivity of elemental bromine often leads to over-bromination, resulting in a crude product contaminated with significant quantities of 1,6-dibromopyrene and 1,8-dibromopyrene isomers. These structural impurities are notoriously difficult to separate due to their similar physical properties, ultimately capping the purity of the final material at approximately 85%. Such low purity levels are unacceptable for high-end OLED applications, where trace impurities can drastically reduce device lifespan and efficiency.
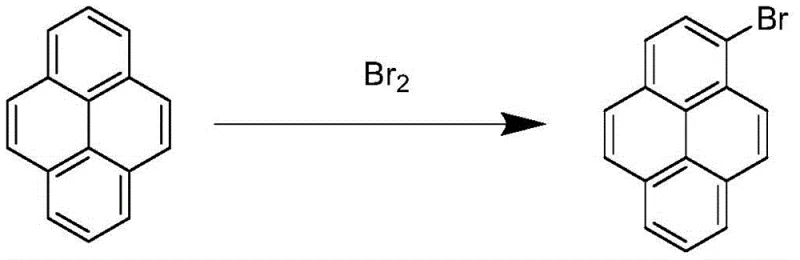
The Novel Approach
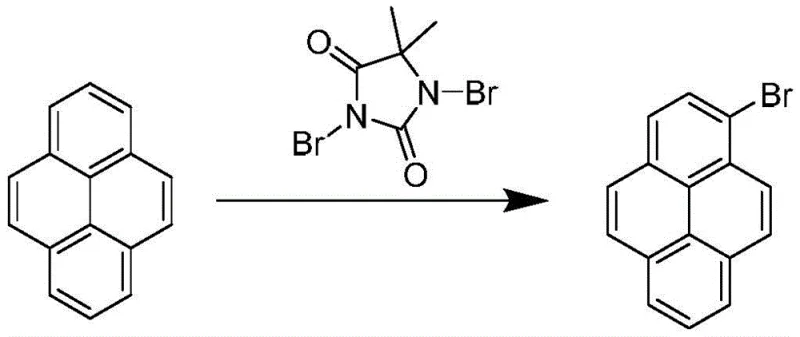
In stark contrast to the hazardous traditional routes, the methodology disclosed in CN108299149B employs dibromohydantoin as a solid, easy-to-handle brominating source that offers superior control over the reaction kinetics. This reagent releases bromine in situ under mild conditions, typically between 20°C and 25°C, which effectively suppresses the formation of poly-brominated byproducts and ensures high regioselectivity for the 1-position. The reaction environment is significantly cleaner, eliminating the release of toxic gases and reducing the burden on waste treatment facilities. Following the initial synthesis, the crude product undergoes a sophisticated purification sequence involving the formation of a charge-transfer complex with picric acid. This step selectively crystallizes the target molecule as a brick-red solid, physically separating it from unreacted starting materials and minor isomers. The subsequent decomposition of this complex using an alkaline solution yields 1-bromopyrene with a purity exceeding 99.5%, establishing a new benchmark for quality in the supply chain of high-purity OLED intermediates.
Mechanistic Insights into Dibromohydantoin-Mediated Bromination
The core chemical transformation in this patented process is an electrophilic aromatic substitution where dibromohydantoin acts as a source of electrophilic bromine species. Unlike the chaotic radical mechanisms often triggered by elemental bromine under harsh conditions, the reaction with dibromohydantoin proceeds through a more controlled pathway that favors mono-substitution at the most electron-rich position of the pyrene ring. The solvent system, which can include dichloromethane, ethyl acetate, or toluene, plays a crucial role in stabilizing the transition state and facilitating the smooth progression of the reaction within a short timeframe of approximately 30 minutes. This kinetic control is vital for minimizing the residence time of reactive intermediates that could otherwise lead to side reactions. The stoichiometry is carefully managed, with a molar ratio of pyrene to dibromohydantoin maintained between 1:0.5 and 1:0.6, ensuring that the brominating agent is the limiting reagent to prevent di-bromination. This precise balancing act is the fundamental reason behind the drastic reduction in impurity profiles compared to legacy methods.
Beyond the primary reaction, the purification mechanism via picric acid complexation is a masterclass in supramolecular chemistry applied to industrial separation. Picric acid, being a strong electron acceptor, forms a stable donor-acceptor complex specifically with the electron-rich 1-bromopyrene molecule. This interaction alters the solubility characteristics of the target compound, causing it to precipitate out of the solution as distinct brick-red crystals while leaving impurities in the mother liquor. The selectivity of this complexation is high enough to exclude the troublesome 1,6- and 1,8-dibromo isomers that plague other methods. Once isolated, the complex is treated with a sodium hydroxide solution, which deprotonates the picric acid, breaking the complex and releasing the free 1-bromopyrene into the organic phase. This clever use of reversible chemical binding allows for the recovery of both the high-value product and the picric acid reagent, which can be recycled after acidification, thereby enhancing the overall atom economy of the process.
How to Synthesize 1-Bromopyrene Efficiently
Implementing this synthesis route requires careful attention to temperature control and stoichiometric addition to maximize the benefits of the dibromohydantoin reagent. The process begins with the suspension of pyrene in a suitable solvent, followed by the portion-wise addition of the brominating agent to manage the slight exotherm and maintain the reaction temperature below 25°C. After the reaction is complete, a simple aqueous workup removes hydantoin byproducts, and the crude material is isolated via concentration and crystallization. The subsequent purification step involves dissolving the crude solid, adding picric acid, and heating gently to induce the formation of the characteristic complex crystals. For detailed operational parameters, including specific solvent volumes, stirring rates, and drying conditions, please refer to the standardized protocol below which outlines the exact execution of this patented technology.
- React pyrene with dibromohydantoin in a solvent like dichloromethane at 20-25°C to form crude 1-bromopyrene.
- Purify the crude product by forming a complex with picric acid in ethanol or dichloromethane at 30-40°C.
- Decompose the complex using sodium hydroxide solution, separate the organic layer, and recrystallize to obtain pure 1-bromopyrene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method translates into tangible strategic advantages regarding cost stability and operational reliability. The shift from hazardous elemental bromine to solid dibromohydantoin simplifies logistics and storage requirements, as the latter is non-volatile and safer to transport, reducing insurance and handling costs significantly. Furthermore, the ability to recover and recycle picric acid within the process creates a closed-loop system that minimizes raw material consumption and waste disposal fees. This efficiency directly contributes to cost reduction in electronic chemical manufacturing by lowering the variable cost per kilogram of the final API intermediate. The robustness of the process also means fewer batch failures and less downtime for equipment maintenance caused by corrosion from acidic gases, ensuring a more consistent flow of goods to downstream customers.
- Cost Reduction in Manufacturing: The elimination of expensive corrosion-resistant equipment required for handling liquid bromine results in substantial capital expenditure savings. Additionally, the high yield of over 95% and the recyclability of the picric acid reagent mean that less raw material is wasted per unit of output. This improved material efficiency drives down the overall production cost, allowing suppliers to offer more competitive pricing without sacrificing margins. The simplified workup procedure also reduces labor hours and energy consumption associated with extensive purification steps, further enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: By utilizing stable, solid reagents like dibromohydantoin, the supply chain becomes less vulnerable to disruptions caused by the strict transportation regulations governing hazardous liquids. The mild reaction conditions reduce the risk of thermal runaways or safety incidents that could halt production lines. This stability ensures a predictable production schedule, which is critical for meeting the just-in-time delivery requirements of major display panel manufacturers. Consequently, partners can rely on a steady supply of high-purity OLED intermediates without the fear of unexpected shortages or quality deviations.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, operating at near-ambient temperatures and atmospheric pressure, which simplifies the engineering requirements for scaling from pilot plants to multi-ton reactors. The absence of toxic HBr gas emissions aligns perfectly with increasingly stringent global environmental regulations, reducing the need for complex abatement systems. This eco-friendly profile not only mitigates regulatory risk but also enhances the brand reputation of manufacturers as sustainable producers. The ability to scale efficiently ensures that reducing lead time for high-purity electronic chemicals is achievable even as market demand surges.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this route into their existing manufacturing frameworks. The answers are derived directly from the experimental data and technical disclosures within the patent, providing a reliable foundation for decision-making. We encourage technical stakeholders to review these points to fully appreciate the operational improvements offered by this method.
Q: Why is dibromohydantoin preferred over elemental bromine for synthesizing 1-bromopyrene?
A: Dibromohydantoin offers a safer, environmentally friendly alternative to elemental bromine, eliminating hazardous HBr gas emissions and reducing the formation of difficult-to-remove dibromo impurities, thereby significantly improving product purity from 85% to over 99.5%.
Q: How does the picric acid complexation method improve purification efficiency?
A: The picric acid complexation method selectively binds with 1-bromopyrene to form brick-red crystals, allowing for the physical separation of impurities. Subsequent treatment with alkali releases the pure product, ensuring a final purity of ≥99.5% suitable for high-end OLED applications.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process operates under mild conditions (20-25°C) without requiring extreme temperatures or pressures, making it highly suitable for commercial scale-up. Additionally, the recovery of picric acid enhances the economic viability and sustainability of large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Bromopyrene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of next-generation display technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of the global OLED market. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify every batch. By leveraging the advanced synthesis methods described in patents like CN108299149B, we can provide a reliable OLED material supplier partnership that guarantees both quality and consistency.
We invite you to engage with our technical procurement team to discuss how our capabilities align with your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data to validate our material against your internal standards, we are ready to assist. Please contact us today to request route feasibility assessments and discover how our optimized manufacturing processes can drive value for your organization.
