Advanced Low-Pressure Ammonolysis Technology for Commercial Phenylenediamine Production
Introduction to Next-Generation Phenylenediamine Manufacturing
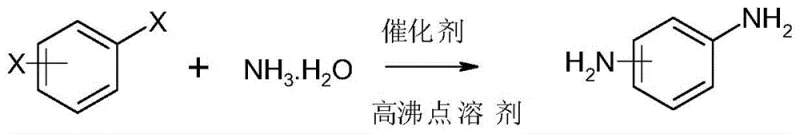
The global demand for high-purity phenylenediamine derivatives continues to surge, driven by their critical applications as key intermediates in the synthesis of advanced polymers, agrochemicals, and active pharmaceutical ingredients (APIs). Traditionally, the industrial production of these vital compounds has relied on hazardous nitration pathways followed by high-pressure hydrogenation, processes fraught with safety risks and environmental liabilities. However, a groundbreaking technological shift is emerging, exemplified by the innovations detailed in Chinese Patent CN111909038A. This patent discloses a novel preparation method that utilizes dihalobenzenes as starting materials and employs specialized Schiff base metal complex catalysts to facilitate direct ammonolysis. By operating under mild low-pressure conditions not exceeding 0.2 MPa, this technology effectively bypasses the dangerous nitration and high-pressure hydrogenolysis steps inherent in legacy manufacturing. For R&D directors and supply chain leaders, this represents a paradigm shift towards safer, greener, and more economically viable production of essential chemical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylenediamine has been dominated by a two-stage process involving the nitration of benzene with mixed acids to form dinitrobenzene, followed by catalytic reduction. This conventional route presents severe operational challenges, particularly regarding thermal management and safety. The nitration stage is highly exothermic; temperatures must be carefully controlled around 40°C for mononitration and raised to 90°C for dinitration, creating a significant risk of thermal runaway due to the explosive nature of dinitrobenzene intermediates. Furthermore, the reliance on mixed acid systems generates substantial volumes of hazardous waste acid, complicating post-treatment and disposal while inflating environmental compliance costs. Alternative routes, such as those involving Hoffmann degradation or multi-step functional group transformations, often suffer from excessive synthetic length and cumbersome purification requirements, rendering them unsuitable for cost-effective large-scale manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the technology described in CN111909038A introduces a streamlined direct ammonolysis strategy that fundamentally alters the reaction landscape. By utilizing dihalobenzenes (where the halogen is preferably chlorine or bromine) and ammonia water in the presence of a robust Schiff base metal complex catalyst, the process achieves high conversion rates under remarkably mild conditions. The reaction is conducted at temperatures between 80°C and 160°C and pressures below 0.2 MPa, eliminating the need for high-pressure autoclaves typically required for hydrogenation. This approach not only drastically reduces the capital expenditure associated with pressure-rated equipment but also minimizes the formation of hazardous byproducts. The simplicity of the workflow, combined with the ability to recycle the catalyst, offers a compelling value proposition for manufacturers seeking to optimize their production lines for both safety and efficiency.
Mechanistic Insights into Schiff Base Metal Complex Catalysis
The core innovation driving this process efficiency lies in the sophisticated design of the Schiff base metal complex catalysts, designated as ML1 and ML2 within the patent literature. These catalysts are formed through the coordination of transition metal ions—such as Pd²⁺, Ni²⁺, Co²⁺, Fe²⁺, or Cu²⁺—with organic ligands synthesized from salicylaldehyde and diamines like ethylenediamine or p-phenylenediamine. The unique electronic environment created by the Schiff base ligand stabilizes the metal center, enhancing its ability to activate the carbon-halogen bond in the dihalobenzene substrate. This activation lowers the energy barrier for nucleophilic attack by ammonia, facilitating the substitution reaction at significantly lower temperatures and pressures than would be possible with uncatalyzed systems. The tunability of the ligand structure allows for precise optimization of the catalyst's activity and selectivity, ensuring that the reaction proceeds cleanly towards the desired diamine product with minimal formation of mono-substituted intermediates or other side products.
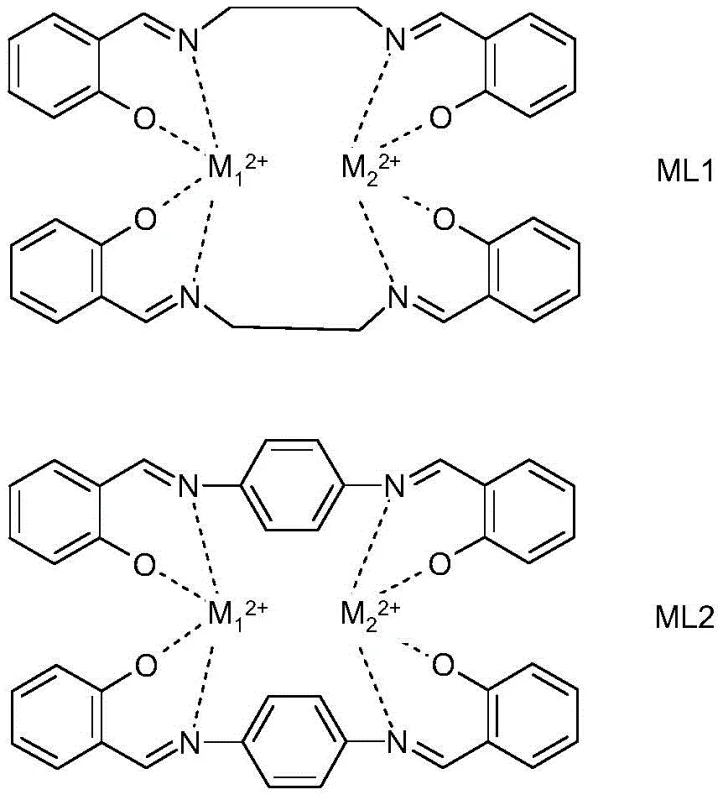
Beyond mere activity, the mechanistic robustness of these catalysts plays a pivotal role in impurity control and product quality. In traditional nitration-reduction sequences, the presence of nitro groups often leads to complex impurity profiles that require extensive purification to meet pharmaceutical standards. The direct ammonolysis pathway, however, avoids the introduction of nitro functionalities entirely, thereby simplifying the downstream purification process. The catalyst's stability under the reaction conditions ensures consistent performance over extended periods, and crucially, the patent highlights the potential for catalyst recovery and reuse. This recyclability is not merely an economic benefit but also a quality assurance measure, as it prevents the leaching of heavy metals into the product stream—a critical consideration for API intermediates. The ability to maintain high selectivity while operating in aqueous ammonia environments demonstrates the superior chemical resilience of these Schiff base complexes compared to simpler metal salt catalysts.
How to Synthesize Phenylenediamine Efficiently
Implementing this advanced synthesis route requires a systematic approach to reaction engineering and process control, leveraging the specific parameters outlined in the patent to maximize yield and purity. The process begins with the careful preparation of the catalyst, followed by the precise charging of reactants into a low-pressure reactor system. Operational excellence depends on maintaining the delicate balance between temperature, pressure, and reaction time to ensure complete conversion of the dihalobenzene starting material. The following guide summarizes the critical operational phases derived from the patented methodology, providing a foundational framework for scaling this technology from laboratory benchtop to commercial production vessels.
- Prepare the Schiff base metal complex catalyst (ML1 or ML2) by reacting salicylaldehyde with ethylenediamine or p-phenylenediamine, followed by complexation with metal salts like Pd, Ni, or Co.
- Charge a low-pressure reactor with dihalobenzene, ammonia water, organic solvent, and the catalyst, maintaining pressure below 0.2 MPa while heating to 130-160°C.
- After reaction completion, recover solvent via vacuum distillation, crystallize the crude product with water, and purify via alcohol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this low-pressure ammonolysis technology translates into tangible strategic advantages that extend far beyond simple unit cost metrics. The elimination of high-pressure hydrogenation steps removes the need for specialized, expensive high-pressure reactors and the associated safety infrastructure, leading to a significant reduction in capital expenditure (CAPEX) for new production facilities. Furthermore, the avoidance of mixed acid nitration eradicates the costly and logistically challenging burden of treating large volumes of spent acid waste, directly lowering operational expenditure (OPEX) related to environmental compliance and waste disposal. The use of readily available starting materials like dihalobenzenes and ammonia water ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized nitration reagents. Additionally, the ability to recycle the catalyst contributes to long-term cost stability, insulating the manufacturing process from fluctuations in precious metal prices.
- Cost Reduction in Manufacturing: The streamlined synthetic route significantly reduces processing time and energy consumption by eliminating the multi-step nitration and high-pressure reduction sequences. By operating at atmospheric or near-atmospheric pressures, the process lowers energy demands for compression and heating, while the recyclability of the catalyst minimizes raw material consumption. This holistic reduction in process complexity drives down the overall cost of goods sold (COGS), allowing for more competitive pricing in the global market for fine chemical intermediates without compromising on margin.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as dihalobenzenes and ammonia water ensures a robust and diversified supply base, reducing vulnerability to single-source supplier disruptions. The simplified process flow shortens the overall production cycle time, enabling faster response to market demand fluctuations and reducing lead times for customers. Moreover, the inherent safety of the low-pressure operation minimizes the risk of unplanned plant shutdowns due to safety incidents, ensuring consistent product availability and strengthening trust with downstream pharmaceutical and agrochemical partners.
- Scalability and Environmental Compliance: The technology is inherently scalable, as demonstrated by successful trials in 100L reactors, making it suitable for rapid expansion to multi-ton production scales. From an environmental perspective, the process aligns perfectly with green chemistry principles by avoiding the generation of toxic nitro-compounds and acidic waste streams. This eco-friendly profile simplifies regulatory approvals and enhances the sustainability credentials of the final product, a factor that is increasingly critical for multinational corporations aiming to meet stringent ESG (Environmental, Social, and Governance) targets in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel phenylenediamine synthesis technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing clarity on catalyst performance, reaction conditions, and product quality. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios or for procurement specialists assessing the long-term viability of suppliers utilizing this method.
Q: How does the Schiff base catalyst improve safety compared to traditional nitration?
A: The Schiff base catalyzed ammonolysis operates at low pressure (≤ 0.2 MPa) and avoids the use of explosive dinitrobenzene intermediates and corrosive mixed acids, significantly reducing thermal runaway risks and hazardous waste generation.
Q: Can the metal complex catalyst be recycled for industrial scale-up?
A: Yes, the patent explicitly highlights that the Schiff base metal complex catalysts can be recovered and reused, which is a critical factor for reducing long-term production costs and minimizing heavy metal contamination in the final API intermediate.
Q: What represent the key impurities in this low-pressure ammonolysis process?
A: Unlike nitration routes which generate nitro-impurities requiring rigorous reduction, this direct substitution method primarily yields mono-substituted byproducts which are easier to separate via crystallization, resulting in a cleaner impurity profile suitable for pharmaceutical grades.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylenediamine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies like the Schiff base catalyzed ammonolysis process is key to securing a competitive edge in the fine chemical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. We understand that for R&D directors, consistency is paramount, and our advanced process control systems guarantee that the high yields and selectivity reported in patent literature are consistently achieved at commercial scale.
We invite global partners to collaborate with us to leverage these technological advancements for their specific supply chain needs. Whether you require custom synthesis of specific phenylenediamine isomers or a comprehensive Customized Cost-Saving Analysis for your current procurement strategy, our technical team is ready to assist. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your project requirements. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only cost-effective and reliable but also aligned with the future of sustainable chemical manufacturing.
