Advanced Acid-Catalyzed Cyclization for Commercial-Scale Edaravone Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical neuroprotective agents, and the preparation method disclosed in patent CN102180834A represents a significant advancement in the manufacturing of Edaravone. This potent free-radical scavenger, chemically known as 3-methyl-1-phenyl-2-pyrazolin-5-one, is essential for treating acute cerebral infarction due to its ability to penetrate the blood-brain barrier and inhibit lipid peroxidation. The patented technology introduces a refined acid-catalyzed cyclization strategy that fundamentally alters the economic and operational landscape of producing this vital pharmaceutical intermediate. By shifting from stoichiometric acidic solvents to a catalytic system within an alcoholic medium, the process achieves exceptional yields exceeding 92% while maintaining rigorous quality control standards. This technical breakthrough not only addresses the longstanding issues of equipment corrosion and high energy consumption associated with legacy methods but also establishes a new benchmark for high-purity Edaravone production suitable for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for Edaravone have been plagued by significant operational inefficiencies and environmental drawbacks that hinder cost-effective commercial scale-up of complex pharmaceutical intermediates. For instance, early methodologies described in US4857542 relied on refluxing phenylhydrazine and methyl acetoacetate in dehydrated alcohol, which typically resulted in mediocre yields around 65%, necessitating extensive downstream processing to recover unreacted materials. Furthermore, alternative approaches such as those found in WO2006/71730 utilized glacial acetic acid as the primary reaction solvent at elevated temperatures, creating a hostile environment for standard stainless steel reactors due to severe corrosion. These traditional processes also suffered from high energy demands required to distill off large volumes of high-boiling acidic solvents during workup, generating substantial waste streams that complicate environmental compliance. The reliance on harsh acidic conditions often led to the formation of difficult-to-remove impurities, thereby compromising the overall purity profile and increasing the burden on purification teams to meet strict pharmacopeial specifications.
The Novel Approach
In stark contrast, the innovative methodology outlined in the present patent leverages a mild acid-catalyzed condensation in an alcoholic solvent system, effectively decoupling the reaction medium from the acidic promoter to optimize both kinetics and thermodynamics. By employing alcohols such as ethanol or isopropanol as the primary solvent and introducing acids like acetic acid or tartaric acid merely as catalysts, the reaction proceeds smoothly at moderate temperatures between 40°C and 90°C. This strategic modification eliminates the need for corrosive bulk acidic solvents, drastically reducing equipment maintenance costs and extending the lifecycle of reactor vessels in a production facility. The process incorporates a sophisticated crystallization technique where a non-alcoholic anti-solvent, such as n-hexane or ethyl acetate, is added post-reaction to precipitate the product directly from the mixture. This elegant workup strategy simplifies isolation, minimizes solvent usage, and inherently purifies the product during the crystallization phase, delivering a crude material with purity already surpassing 99%.
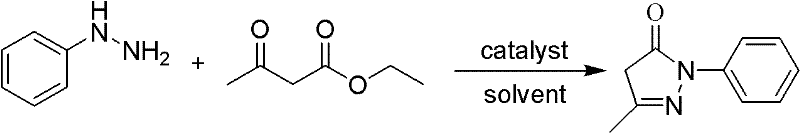
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this synthetic advancement lies in the precise mechanistic control of the condensation reaction between phenylhydrazine and ethyl acetoacetate, facilitated by proton donation from the acid catalyst. In this mechanism, the acid catalyst activates the carbonyl group of the ethyl acetoacetate, increasing its electrophilicity and promoting nucleophilic attack by the hydrazine nitrogen. This initial addition is followed by a cyclization step where the second nitrogen atom attacks the adjacent ketone carbonyl, forming the pyrazolone ring structure characteristic of Edaravone. The use of an alcoholic solvent stabilizes the transition states and intermediates through hydrogen bonding, ensuring that the reaction pathway favors the desired cyclic product over linear oligomers or hydrolysis byproducts. Furthermore, the mild thermal conditions prevent the degradation of the sensitive pyrazolone ring, which can occur under the harsher reflux conditions of older methods, thereby preserving the structural integrity of the molecule throughout the synthesis.
Impurity control is intrinsically built into this mechanism through the selective solubility properties of the reaction matrix. The choice of acid catalyst, particularly organic acids like tartaric acid, provides a buffered environment that minimizes side reactions such as over-alkylation or polymerization of the hydrazine species. As the reaction progresses, the formation of water as a byproduct is managed by the solvent system, preventing the hydrolysis of the ester group which could lead to carboxylic acid impurities. Upon completion, the introduction of the non-alcoholic anti-solvent shifts the solubility equilibrium dramatically, forcing the highly pure Edaravone to crystallize out while leaving soluble impurities and residual starting materials in the supernatant. This thermodynamic driving force ensures that the crystal lattice forms with high selectivity, effectively excluding structurally similar contaminants and resulting in a product with a superior impurity profile right from the crude stage.
How to Synthesize Edaravone Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and temperature gradients to maximize the benefits of the catalytic system. The process begins with the dissolution of phenylhydrazine in an alcoholic solvent, followed by the sequential addition of ethyl acetoacetate and the acid catalyst under an inert atmosphere to prevent oxidative degradation. The reaction mixture is then heated to a controlled range of 60°C to 85°C, allowing the cyclization to proceed to completion within a short timeframe of 1 to 3 hours. Following the reaction, the mixture is cooled, and a non-alcoholic solvent is introduced to induce precipitation, after which the solid is filtered, washed, and dried to yield the high-purity target compound. For a detailed breakdown of the specific operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- React phenylhydrazine with ethyl acetoacetate in an alcoholic solvent (e.g., ethanol) using a catalytic amount of acid (e.g., acetic acid) at 40-90°C under inert gas protection.
- Cool the reaction mixture to 50-65°C and add a non-alcoholic anti-solvent (e.g., n-hexane) to induce crystallization.
- Filter the crude product, wash with solvent mixtures, and dry under reduced pressure to obtain Edaravone with purity >99%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative advantages in terms of cost structure and operational reliability. By eliminating the need for bulk acetic acid as a solvent, the process significantly reduces raw material costs and removes the logistical burden associated with handling and disposing of large volumes of corrosive chemicals. The simplified workup procedure, which relies on direct crystallization rather than complex extraction and distillation sequences, translates into shorter batch cycle times and higher throughput capacity within existing manufacturing infrastructure. This efficiency gain allows suppliers to respond more rapidly to market demand fluctuations, ensuring a steady flow of high-purity pharmaceutical intermediates without the bottlenecks typical of older, labor-intensive technologies.
- Cost Reduction in Manufacturing: The shift to a catalytic acid system drastically lowers the consumption of expensive reagents and reduces the energy load required for solvent recovery. Since the acid is used in catalytic quantities rather than as a bulk solvent, the cost per kilogram of produced Edaravone is substantially decreased, offering a competitive pricing advantage in the global market. Additionally, the reduction in equipment corrosion extends the lifespan of reactor vessels and piping, leading to significant long-term savings on capital expenditure and maintenance budgets for manufacturing partners.
- Enhanced Supply Chain Reliability: The use of common, commercially available solvents like ethanol and n-hexane ensures that the supply chain is not vulnerable to shortages of specialized or hazardous reagents. The mild reaction conditions and robust nature of the process mean that production can be maintained consistently across different facilities without requiring highly specialized operator training or exotic equipment. This standardization facilitates easier technology transfer and multi-site manufacturing strategies, thereby mitigating the risk of supply disruptions and ensuring continuous availability for downstream drug formulation.
- Scalability and Environmental Compliance: The process is inherently green, generating minimal waste streams and avoiding the release of volatile acidic vapors that require complex scrubbing systems. The simplicity of the crystallization-based isolation makes it highly scalable from pilot plant to multi-ton commercial production without losing yield or purity performance. This environmental friendliness aligns with increasingly strict global regulations on industrial emissions, reducing the compliance burden and potential fines associated with waste management, thus securing the long-term viability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Edaravone using this advanced acid-catalyzed methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear understanding of the technology's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: How does the new acid-catalyzed method improve upon traditional acetic acid solvent methods?
A: Traditional methods often use acetic acid as both solvent and reactant, leading to high energy consumption due to high boiling points and severe equipment corrosion. The novel method uses acetic acid only as a catalyst in an alcohol solvent, significantly reducing energy costs and equipment maintenance while achieving yields over 92%.
Q: What purity levels can be achieved with this synthesis route?
A: The process allows for the production of crude Edaravone with purity exceeding 99%. Through a subsequent recrystallization step using alcoholic and non-alcoholic solvents, the final refined product can achieve purity levels greater than 99.9%, meeting stringent pharmaceutical standards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It operates under mild conditions (40-90°C), avoids the generation of significant three-waste problems, and utilizes simple workup procedures like cooling crystallization, making it highly efficient for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edaravone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and supply security for essential neuroprotective agents like Edaravone. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of international pharmaceutical clients. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Edaravone meets or exceeds the high standards set forth by global pharmacopeias, providing our partners with absolute confidence in the material they receive for their final drug formulations.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless partnership for your Edaravone sourcing strategy.
