Advanced Manufacturing of Itopride Hydrochloride: A Cost-Effective Route for Global API Production
Introduction to Patent CN102993038A: A Paradigm Shift in Itopride Synthesis
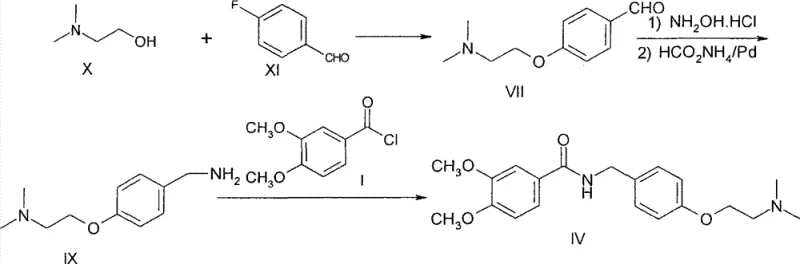
The global demand for gastrointestinal prokinetic agents continues to drive the need for efficient, cost-effective manufacturing processes for key active pharmaceutical ingredients (APIs). Patent CN102993038A introduces a groundbreaking preparation method for Itopride Hydrochloride that fundamentally alters the economic and technical landscape of its production. Unlike traditional routes that rely on costly precursors and hazardous high-pressure conditions, this novel methodology leverages inexpensive, readily available starting materials—specifically N,N-dimethylethanolamine and p-fluorobenzaldehyde—to construct the core molecular framework. The strategic innovation lies in a streamlined sequence involving an initial etherification followed by a sophisticated one-pot reductive amination under atmospheric pressure. This approach not only simplifies the operational workflow but also significantly mitigates the formation of difficult-to-remove ester by-products that plague earlier synthetic strategies. For R&D directors and process chemists, this patent represents a critical opportunity to optimize impurity profiles and enhance overall process robustness. By shifting the synthetic logic from complex amide couplings to a more direct ether-amine construction, the technology offers a compelling value proposition for reliable pharmaceutical intermediate suppliers aiming to secure long-term supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Itopride has been constrained by two primary methodologies, both of which suffer from significant economic and technical drawbacks that hinder cost reduction in API manufacturing. The first conventional route, often referred to as the Amide Method, initiates with the coupling of 3,4-dimethoxybenzoyl chloride and p-hydroxybenzylamine. While conceptually straightforward, this reaction is plagued by competitive side reactions; under the necessary alkaline conditions, the phenolic hydroxyl group frequently undergoes esterification with the acid chloride, generating substantial quantities of ester by-products. These impurities are structurally similar to the target amide, making their removal via standard crystallization or chromatography exceptionally difficult and yield-destructive. Furthermore, the second major bottleneck in this route is the reliance on p-hydroxybenzylamine and N,N-dimethylaminoethyl chloride hydrochloride, both of which command high market prices and exhibit limited availability, creating supply chain vulnerabilities.
The second conventional pathway, known as the Phenol Ether Method, attempts to address the ether linkage early but introduces severe safety and capital intensity issues. This route typically involves reacting p-hydroxybenzaldehyde with N,N-dimethylaminoethyl chloride hydrochloride to form an ether intermediate, followed by conversion to an oxime and subsequent reduction. The critical failure point here is the reduction step, which necessitates catalytic hydrogenation at high pressures, specifically around 50 atmospheres. Operating at such elevated pressures requires specialized, expensive autoclave equipment and rigorous safety protocols, drastically increasing both CAPEX and OPEX. Moreover, the multi-step nature of converting the oxime to the amine in this legacy process results in cumulative yield losses and increased solvent consumption, rendering it less attractive for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy limitations, the novel approach detailed in CN102993038A engineers a synthesis path that is both economically superior and operationally safer. By reversing the disconnection strategy, the process begins with the nucleophilic substitution of p-fluorobenzaldehyde by N,N-dimethylethanolamine. This etherification step is highly selective and utilizes cheap, bulk chemicals, effectively bypassing the expensive amine salts required in older methods. The true brilliance of this route, however, is manifested in the subsequent transformation of the aldehyde intermediate. Instead of isolating unstable intermediates or employing high-pressure gas, the patent describes a telescoped one-pot procedure where the aldehyde is converted to an oxime and immediately reduced to the benzylamine using ammonium formate and a palladium catalyst at atmospheric pressure. This elimination of high-pressure hydrogenation equipment removes a major barrier to entry for many manufacturers. The final acylation with 3,4-dimethoxybenzoyl chloride proceeds smoothly on this purified amine backbone, yielding the target molecule with exceptional purity. This holistic redesign of the synthetic tree ensures that high-purity pharmaceutical intermediates can be produced with a reported cost reduction of approximately 30% compared to prior art.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Transfer Hydrogenation
From a mechanistic perspective, the success of this novel route relies on the precise control of two distinct chemical transformations: the formation of the aryl alkyl ether and the subsequent reductive amination. The initial step involves a nucleophilic aromatic substitution (SNAr) where the alkoxide derived from N,N-dimethylethanolamine attacks the electron-deficient aromatic ring of p-fluorobenzaldehyde. The presence of the formyl group at the para-position activates the ring towards nucleophilic attack by withdrawing electron density, facilitating the displacement of the fluoride ion. The choice of base, such as sodium hydride or potassium tert-butoxide, is critical here; it ensures the quantitative generation of the alkoxide nucleophile without inducing side reactions like Cannizzaro disproportionation of the aldehyde. The use of polar aprotic solvents like DMF or DMA further enhances the reaction rate by stabilizing the transition state and solvating the cation, allowing the reaction to proceed efficiently at moderate temperatures of 60-80°C.
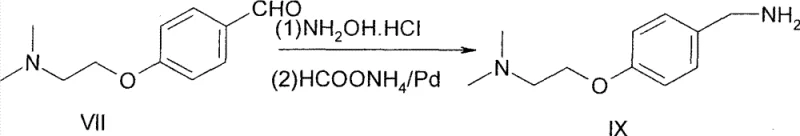
The second critical mechanistic feature is the one-pot reductive amination, which elegantly combines oxime formation and transfer hydrogenation. Initially, the aldehyde reacts with hydroxylamine hydrochloride to form the corresponding oxime intermediate. Rather than isolating this species, the protocol introduces ammonium formate and a Pd/C catalyst directly into the reaction mixture. In this transfer hydrogenation process, ammonium formate serves as the hydrogen donor, decomposing on the palladium surface to generate reactive atomic hydrogen in situ. This hydrogen species then reduces the C=N double bond of the oxime to a C-N single bond, yielding the primary amine. This "one-pot" strategy is mechanistically advantageous because it prevents the accumulation of potentially unstable intermediates and minimizes exposure to air or moisture that could degrade the product. By avoiding the need for external hydrogen gas cylinders and high-pressure reactors, the process inherently reduces the risk profile while maintaining high atom economy. This sophisticated orchestration of reaction conditions ensures that the final benzylamine intermediate is obtained with an HPLC purity of 96.7% directly from the crude mixture, demonstrating superior impurity control.
How to Synthesize Itopride Hydrochloride Efficiently
Implementing this synthesis requires strict adherence to the optimized conditions described in the patent to maximize yield and purity. The process is divided into four logical stages: etherification, one-pot reductive amination, amidation, and salt formation. Each stage has been fine-tuned to balance reaction kinetics with workup simplicity, ensuring that the process is robust enough for kilogram-to-ton scale manufacturing. Operators must pay particular attention to the pH adjustments during the workup phases, as the basic nature of the amine intermediates requires careful extraction protocols to separate organic products from inorganic salts. The following guide outlines the standardized operational parameters derived from the patent examples, serving as a foundational reference for process engineers aiming to replicate this high-efficiency route.
- Etherification: React N,N-dimethylethanolamine with p-fluorobenzaldehyde using NaH or t-BuOK in DMF/DMA at 60-80°C to form 4-(2-dimethylaminoethoxy)benzaldehyde.
- One-Pot Reductive Amination: Convert the aldehyde to an oxime using hydroxylamine hydrochloride, then reduce in situ with ammonium formate and Pd/C to obtain the benzylamine intermediate.
- Amidation and Salt Formation: Acylate the benzylamine with 3,4-dimethoxybenzoyl chloride, followed by treatment with HCl in alcohol to crystallize pure Itopride Hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple unit price reductions. The primary advantage lies in the radical simplification of the raw material portfolio. By substituting scarce and expensive specialty chemicals like p-hydroxybenzylamine with commodity chemicals like N,N-dimethylethanolamine and p-fluorobenzaldehyde, manufacturers can insulate themselves from volatile market fluctuations and supply disruptions. These bulk chemicals are produced on a massive global scale, ensuring consistent availability and stable pricing structures. Furthermore, the elimination of the high-pressure hydrogenation step removes a significant bottleneck in production scheduling. Facilities that lack specialized high-pressure autoclaves can now produce this intermediate, expanding the potential supplier base and enhancing competition, which naturally drives down costs for the end buyer.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, driven by both material and operational savings. The patent explicitly notes that the final production cost can be reduced by approximately 30% compared to previously reported methods. This saving is achieved not only through cheaper starting materials but also by eliminating the capital-intensive requirement for high-pressure reactors rated for 50 atmospheres. Additionally, the one-pot nature of the reduction step reduces solvent consumption and labor hours associated with intermediate isolation and purification. The avoidance of ester by-product formation in the initial stages also means higher overall yields and less waste disposal cost, contributing to a leaner, more profitable manufacturing model.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the robustness of the chemistry. Traditional routes relying on high-pressure hydrogenation are prone to delays due to equipment maintenance, safety inspections, and the logistical challenges of handling compressed hydrogen gas. The new atmospheric pressure transfer hydrogenation method utilizes solid ammonium formate and standard stirring reactors, which are ubiquitous in fine chemical plants. This universality means that production can be easily shifted between different manufacturing sites without requiring specialized infrastructure upgrades. Consequently, lead times for high-purity pharmaceutical intermediates can be shortened, and the risk of production stoppages due to equipment failure is markedly decreased.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this route aligns perfectly with modern green chemistry principles. The reaction conditions are mild (60-80°C), reducing energy consumption for heating and cooling. The use of transfer hydrogenation avoids the safety hazards associated with storing and transporting large volumes of hydrogen gas. Moreover, the high selectivity of the reactions minimizes the generation of hazardous waste streams, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The process has been demonstrated to scale effectively, with examples showing successful execution from gram to multi-hundred-gram scales, indicating a clear path to commercial scale-up of complex pharmaceutical intermediates without unforeseen engineering hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in CN102993038A, providing clarity for technical teams evaluating the feasibility of this method for their specific production needs. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: How does this new synthesis route reduce production costs compared to conventional methods?
A: The patented route replaces expensive starting materials like p-hydroxybenzylamine and N,N-dimethylaminoethyl chloride hydrochloride with significantly cheaper commodities such as N,N-dimethylethanolamine and p-fluorobenzaldehyde. Additionally, it eliminates the need for high-pressure hydrogenation equipment (50 atm), reducing both capital expenditure and operational energy costs by approximately 30%.
Q: What are the purity specifications achievable with this one-pot reductive amination method?
A: The process demonstrates excellent impurity control, achieving an HPLC purity of 96.7% for the crude benzylamine intermediate without purification. The final Itopride base reaches 98.6% purity after recrystallization, and the final hydrochloride salt achieves 99.5% purity after refinement with isopropanol.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is highly scalable. It utilizes mild reaction conditions (60-80°C) and atmospheric pressure for the reduction step, avoiding the safety risks and engineering complexities associated with high-pressure hydrogenation. The use of common industrial solvents like DMF, methanol, and dichloromethane further facilitates easy scale-up from pilot to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Itopride Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering quality commitment. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by patent CN102993038A are fully realized in your supply chain. Our facilities are equipped with state-of-the-art reactor systems capable of handling the specific solvent and temperature profiles required for this synthesis, including large-scale etherification and atmospheric transfer hydrogenation units. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Itopride Hydrochloride meets the highest international pharmacopeial standards, delivering the consistency your R&D and production teams depend on.
We invite you to leverage our technical expertise to optimize your sourcing strategy for this critical gastrointestinal drug intermediate. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this novel route can improve your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By collaborating with us, you gain access to a secure, cost-effective, and scalable supply of high-quality Itopride Hydrochloride, positioning your organization for success in the competitive global pharmaceutical market.
