Industrial Scale Synthesis of Upper-Edge Eliminated Calixarene Phosphate Derivatives for Enhanced Rare Earth Extraction
Industrial Scale Synthesis of Upper-Edge Eliminated Calixarene Phosphate Derivatives for Enhanced Rare Earth Extraction
The global demand for high-purity rare earth elements, particularly praseodymium and neodymium used in permanent magnets for electric vehicles and wind turbines, has necessitated the development of more efficient separation technologies. Patent CN109575070B introduces a groundbreaking chemical architecture: a calixarene phosphate derivative where the upper edge is completely eliminated of tert-butyl groups and the lower edge is fully substituted with phosphate esters. This structural modification represents a significant leap forward in supramolecular chemistry, specifically designed to overcome the steric limitations of traditional calixarene extractants. By systematically removing the bulky upper-rim substituents, the molecular cavity becomes more accessible, dramatically enhancing the host-guest interaction with light rare earth ions. This technical insight report analyzes the synthetic methodology and commercial viability of this novel compound, positioning it as a critical enabler for next-generation hydrometallurgical processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional calixarene phosphate derivatives typically retain the tert-butyl groups at the upper rim, inherited from the standard p-tert-butylcalixarene starting material. While these groups provide solubility, they create significant steric hindrance that physically blocks metal ions from entering the macrocyclic cavity efficiently. In industrial separation scenarios, this steric crowding results in lower distribution coefficients and poor selectivity between chemically similar lanthanides like Pr3+ and Nd3+. Furthermore, conventional synthesis routes often suffer from incomplete substitution at the lower hydroxyl rim, leading to heterogeneous product mixtures that require energy-intensive and solvent-heavy chromatographic purification. The toxicity of solvents used in older methods and the complexity of post-treatment procedures further exacerbate the environmental footprint and operational costs, making them less attractive for modern, green chemical manufacturing standards.
The Novel Approach
The patented methodology revolutionizes this landscape by implementing a strategic two-step synthesis that first strips the upper rim entirely before functionalizing the lower rim. The first stage involves a Lewis acid-catalyzed de-alkylation where anhydrous aluminum trichloride facilitates the removal of all eight tert-butyl groups in the presence of phenol and toluene. 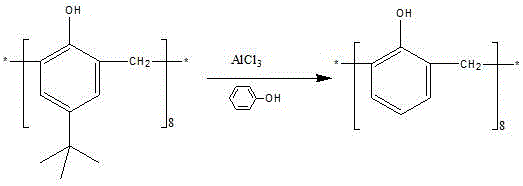 This creates a sterically unhindered calixarene scaffold. Subsequently, the second step employs a phase-transfer catalyzed phosphorylation using dialkyl chlorophosphate under strong alkaline conditions.
This creates a sterically unhindered calixarene scaffold. Subsequently, the second step employs a phase-transfer catalyzed phosphorylation using dialkyl chlorophosphate under strong alkaline conditions. 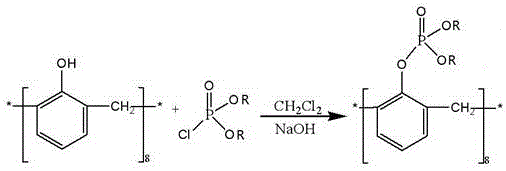 This sequence ensures complete substitution at all eight phenolic positions, yielding a homogeneous product with superior extraction capabilities. The process replaces complex purification with simple precipitation and washing, drastically simplifying the workflow for a reliable industrial specialty chemical supplier.
This sequence ensures complete substitution at all eight phenolic positions, yielding a homogeneous product with superior extraction capabilities. The process replaces complex purification with simple precipitation and washing, drastically simplifying the workflow for a reliable industrial specialty chemical supplier.
Mechanistic Insights into AlCl3-Catalyzed De-alkylation and Phosphorylation
The core of this innovation lies in the precise control of the de-tert-butylation mechanism. Anhydrous aluminum trichloride acts as a potent Lewis acid, coordinating with the aromatic pi-system and facilitating the cleavage of the C-C bond between the benzene ring and the tert-butyl group. The presence of phenol serves as a scavenger for the generated isobutylene carbocations, preventing re-alkylation and driving the equilibrium toward the fully de-alkylated product. This reaction transforms the lipophilic, bulky precursor into a more polar, open-cavity intermediate. The subsequent phosphorylation leverages tetrabutylammonium bromide (TBAB) as a phase transfer catalyst, which shuttles hydroxide ions into the organic dichloromethane phase. This generates the phenoxide anions in situ, which then nucleophilically attack the phosphorus center of the dialkyl chlorophosphate. The result is a robust P-O-C linkage that is stable under extraction conditions yet flexible enough to accommodate the coordination geometry of rare earth ions.
Impurity control is inherently built into this mechanistic pathway. The de-alkylation step produces a solid intermediate that can be easily isolated via precipitation in acidic media, washing away aluminum salts and phenolic byproducts. In the second step, the use of excess dialkyl chlorophosphate ensures that no free hydroxyl groups remain, which could otherwise act as competing binding sites or degradation points. The final product, a yellow gum, is purified by dissolution in methanol and precipitation in water, followed by a final wash in dichloromethane. 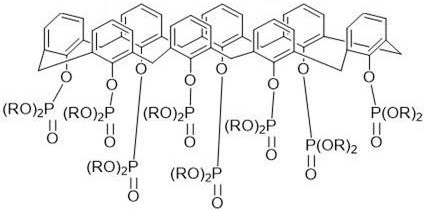 This rigorous purification protocol ensures that the final material meets stringent purity specifications required for high-performance separation applications, minimizing the risk of third-phase formation or emulsion issues during industrial solvent extraction.
This rigorous purification protocol ensures that the final material meets stringent purity specifications required for high-performance separation applications, minimizing the risk of third-phase formation or emulsion issues during industrial solvent extraction.
How to Synthesize Upper-Edge Eliminated Calixarene Phosphate Derivative Efficiently
The synthesis protocol outlined in the patent offers a reproducible pathway for manufacturing this high-value extractant. It begins with the careful handling of moisture-sensitive reagents like anhydrous aluminum trichloride under an inert nitrogen atmosphere to prevent catalyst deactivation. The reaction temperature is maintained at reflux in toluene to ensure complete conversion of the bulky starting material. Following the isolation of the intermediate, the phosphorylation step requires precise control of the base addition rate to manage exothermicity and maintain the integrity of the phase transfer catalyst system. For detailed operational parameters, stoichiometry, and safety precautions, please refer to the standardized synthesis guide below.
- De-alkylation: React p-tert-butylcalixarene with phenol and anhydrous aluminum trichloride in toluene under nitrogen protection to remove upper-edge tert-butyl groups.
- Purification of Intermediate: Separate the organic layer, wash with dilute hydrochloric acid, and precipitate to obtain off-white de-tert-butylcalixarene solid.
- Substitution Reaction: React the intermediate with dialkyl chlorophosphate and sodium hydroxide in dichloromethane using tetrabutylammonium bromide as a phase transfer catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial cost reduction in rare earth separation manufacturing by fundamentally simplifying the production of the extractant itself. The reliance on commodity chemicals such as phenol, toluene, and sodium hydroxide reduces raw material volatility risks compared to exotic reagents. Moreover, the elimination of chromatographic purification steps significantly lowers solvent consumption and waste disposal costs, directly impacting the bottom line. The high yields reported in the patent examples, consistently exceeding 85% and reaching up to 96% for the intermediate, indicate a highly efficient atom economy that translates to lower unit costs for the final active ingredient.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts or complex ligand systems often found in alternative separation technologies. By utilizing a straightforward Lewis acid de-alkylation followed by a robust nucleophilic substitution, the manufacturing overhead is drastically simplified. The ability to purify the product through simple precipitation and washing rather than column chromatography means that capital expenditure on purification equipment is minimized, and operational throughput is maximized. This streamlined approach ensures that the cost per kilogram of the extractant remains competitive, even when scaled to multi-ton production levels required by the growing rare earth industry.
- Enhanced Supply Chain Reliability: The raw materials for this synthesis are widely available in the global chemical market, reducing the risk of supply chain disruptions associated with niche precursors. The synthetic route is robust and tolerant to minor variations in reaction conditions, which ensures consistent batch-to-batch quality and reliable delivery schedules. For a reliable industrial specialty chemical supplier, this predictability is crucial for maintaining long-term contracts with hydrometallurgical plants that cannot afford downtime due to reagent shortages. The stability of the intermediate also allows for potential stockpiling strategies, further buffering the supply chain against market fluctuations.
- Scalability and Environmental Compliance: The reaction conditions are relatively mild, avoiding extreme pressures or temperatures that would require specialized high-pressure reactors. This makes the process highly scalable from pilot plant to commercial production using standard glass-lined or stainless steel reactors. Furthermore, the reduction in solvent usage and the avoidance of toxic heavy metal catalysts align with increasingly strict environmental regulations. The waste streams generated are primarily aqueous salts and recoverable organic solvents, which are easier to treat and recycle compared to the hazardous waste associated with traditional organophosphorus synthesis, supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel calixarene derivative in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy for engineering and procurement teams evaluating this technology for adoption.
Q: How does eliminating the upper-edge tert-butyl groups improve rare earth separation?
A: Removing the bulky tert-butyl groups eliminates steric hindrance around the macrocyclic cavity, allowing Pr3+ and Nd3+ ions to access the phosphoryl binding sites more effectively, thereby significantly increasing extraction efficiency and selectivity compared to traditional derivatives.
Q: What are the key advantages of this two-step synthesis method for large-scale production?
A: The process utilizes mild reaction conditions, inexpensive raw materials like phenol and toluene, and avoids complex chromatographic purification by relying on simple precipitation and washing steps, resulting in high yields exceeding 85% and reduced operational costs.
Q: Is the final calixarene phosphate derivative stable under industrial extraction conditions?
A: Yes, the derivative demonstrates robust stability in organic phases such as dichloroethane and maintains high extraction performance at neutral pH levels (around pH 6.5), making it suitable for continuous counter-current extraction processes in hydrometallurgy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Calixarene Phosphate Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for advanced functional materials, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of calixarene chemistry, including moisture-sensitive reactions and rigorous purification protocols. We understand that the purity of your extractant dictates the efficiency of your entire separation plant; therefore, we adhere to stringent purity specifications and utilize rigorous QC labs to verify every batch. Our team is ready to support your transition from laboratory validation to full-scale industrial deployment, ensuring a seamless supply of this high-performance material.
We invite you to engage with our technical procurement team to discuss how this innovative extractant can optimize your specific rare earth separation circuit. By requesting a Customized Cost-Saving Analysis, you can quantify the potential operational efficiencies this molecule offers for your facility. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production volume requirements, ensuring you secure a competitive advantage in the critical rare earth supply chain.
