Optimized Synthesis of Glasdegib Key Intermediate for Commercial API Production
Optimized Synthesis of Glasdegib Key Intermediate for Commercial API Production
The pharmaceutical landscape for treating Acute Myeloid Leukemia (AML) has been significantly advanced by the development of Glasdegib, a hedgehog pathway inhibitor. Central to the efficient manufacturing of this life-saving medication is the reliable production of its key chiral intermediate, chemically defined as the compound of structural formula 3a. A recent technological breakthrough, documented in patent CN113443995B, introduces a refined preparation method that addresses critical bottlenecks in yield, safety, and impurity control. This novel synthetic route replaces hazardous gaseous reagents with a controlled liquid-phase ring-opening strategy, ensuring a more robust supply chain for this high-value pharmaceutical intermediate. By optimizing the Diels-Alder cycloaddition and subsequent esterification steps, manufacturers can achieve superior purity profiles essential for downstream API synthesis.
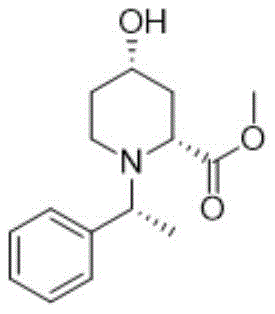
The strategic importance of this intermediate cannot be overstated, as its stereochemical integrity directly influences the efficacy of the final drug substance. The patent details a comprehensive protocol that navigates the complexities of bicyclic lactone formation and selective ring opening. For R&D directors and process chemists, understanding the nuances of this improved methodology offers a pathway to reduce cost of goods sold (COGS) while enhancing environmental compliance. The following analysis dissects the technical advantages of this approach, contrasting it with legacy methods to highlight its potential for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing the Glasdegib key intermediate have historically relied on the use of ammonia gas to facilitate the ring-opening of the bicyclic lactone precursor. This approach presents significant operational challenges, primarily stemming from the difficulty in precisely controlling the concentration of ammonia within the reaction matrix. If the ammonia concentration is too low, the ring-opening reaction fails to proceed to completion, leaving unreacted starting materials that complicate downstream purification. Conversely, if the ammonia concentration is too high, the reaction pathway diverges, leading to the formation of undesirable amide by-products which are structurally similar to the target ester and notoriously difficult to separate. Furthermore, the handling of anhydrous ammonia gas requires specialized pressure-rated equipment and rigorous safety protocols to mitigate the risks of toxicity and leakage, thereby inflating capital expenditure and operational overheads for manufacturing facilities.
The Novel Approach
The innovative process disclosed in patent CN113443995B circumvents these issues by employing a sodium methoxide-mediated ring-opening reaction in a methanol solvent system. This liquid-phase reagent allows for precise stoichiometric control, ensuring that the nucleophilic attack on the lactone carbonyl occurs efficiently without the volatility associated with gaseous ammonia. The method initiates with a Diels-Alder reaction between a specific diene precursor (Formula 1) and glyoxylic acid in the presence of 4A molecular sieves and acetonitrile, generating the bicyclic intermediate mixture (Formula 2a and 2b). Subsequent treatment with sodium methoxide at controlled low temperatures facilitates a clean conversion to the methyl ester target (Formula 3a). This shift not only eliminates the generation of amide impurities but also simplifies the workup procedure, as the reaction can be quenched and neutralized using standard aqueous acid solutions, drastically reducing the complexity of cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Diels-Alder Cycloaddition and Methanolic Ring-Opening
The core of this synthetic strategy lies in the stereoselective construction of the piperidine-fused lactone scaffold via a Diels-Alder cycloaddition. In the first step, the electron-deficient glyoxylic acid acts as the dienophile, reacting with the electron-rich diene component (Formula 1) to form the bicyclic structure. The use of 4A molecular sieves is critical here, as they sequester water generated during the condensation or present in the reagents, driving the equilibrium towards the formation of the iminium or activated carbonyl species necessary for the cycloaddition. This step yields a mixture of diastereomers (Formula 2a and 2b), which are carried forward without immediate separation, demonstrating the robustness of the subsequent ring-opening step to handle stereochemical mixtures effectively. The reaction kinetics are managed by maintaining the temperature below 40°C during the addition of the diene to prevent thermal degradation or polymerization of the reactive aldehyde species.
The subsequent ring-opening mechanism is a classic nucleophilic acyl substitution, where the methoxide ion attacks the carbonyl carbon of the lactone ring. The patent emphasizes a critical temperature window of -5°C to 10°C for this transformation. Mechanistically, this low-temperature regime is vital to suppress competing hydrolysis reactions. At temperatures exceeding 10°C, the kinetic energy of the system allows water molecules (either residual or from the quench) to compete with methoxide, leading to the hydrolysis of the lactone into the corresponding carboxylic acid rather than the desired methyl ester. Data from the patent examples indicates that at temperatures above 20°C, nearly half of the intermediate is lost to hydrolysis, severely impacting the overall mass balance. By strictly adhering to the sub-ambient temperature protocol, the process ensures high chemoselectivity for the ester product, thereby maximizing the yield of the high-purity pharmaceutical intermediate required for GMP production.
How to Synthesize Glasdegib Key Intermediate Efficiently
The synthesis of this critical building block requires meticulous attention to reaction parameters, particularly regarding moisture control and thermal management during the ring-opening phase. The process is designed to be scalable, utilizing common organic solvents like acetonitrile and methanol which are easily recovered and recycled in an industrial setting. Operators must ensure that the addition of sodium methoxide is performed slowly to manage the exotherm and maintain the critical temperature range. Following the reaction, the pH adjustment to 6-7 using dilute hydrochloric acid is essential to neutralize excess base without inducing acid-catalyzed decomposition of the sensitive ester functionality. For a detailed, step-by-step breakdown of the standardized operating procedures, including exact reagent quantities and workup protocols, please refer to the technical guide below.
- Perform a Diels-Alder reaction by adding 4A molecular sieves, acetonitrile, and the diene precursor (Formula 1) to glyoxylic acid to generate the bicyclic lactone intermediate mixture.
- Execute a controlled ring-opening reaction by dissolving the intermediate in methanol and slowly adding sodium methoxide while maintaining a strict temperature range of -5 to 10°C to prevent hydrolysis.
- Purify the resulting crude mixture via silica gel column chromatography using a gradient elution of petroleum ether and ethyl acetate to isolate the high-purity target compound (Formula 3a).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers substantial strategic benefits that extend beyond mere chemical yield. The elimination of ammonia gas from the process flow removes a significant safety hazard and regulatory burden, allowing manufacturing sites to operate with greater flexibility and reduced insurance liabilities. This transition to a liquid-phase reagent system simplifies the supply chain logistics, as sodium methoxide solutions are widely available and easier to transport and store than pressurized ammonia cylinders. Consequently, this leads to a more resilient supply chain capable of meeting the rigorous demands of global API production schedules without the risk of shutdowns due to hazardous material handling incidents.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the significant improvement in yield resulting from the suppression of side reactions. By avoiding the formation of amide by-products and minimizing hydrolysis through precise temperature control, the consumption of expensive starting materials is drastically reduced. Furthermore, the simplified purification process, which avoids complex separations of amide impurities, lowers the demand for chromatography media and solvents. The ability to carry forward diastereomeric mixtures until the final purification step also reduces the number of unit operations, translating to lower labor and utility costs per kilogram of produced intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as methanol, acetonitrile, and sodium methoxide ensures that the raw material supply is stable and less prone to market volatility compared to specialized gaseous reagents. The robustness of the reaction conditions, specifically the tolerance for carrying mixed diastereomers through the synthesis, enhances the reliability of batch-to-batch consistency. This stability is crucial for long-term supply agreements, as it minimizes the risk of batch failures and ensures a continuous flow of material to downstream API manufacturers, thereby reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently greener, as it avoids the release of volatile ammonia into the scrubber systems, reducing the load on wastewater treatment facilities. The use of standard organic solvents facilitates efficient recovery and recycling loops, aligning with modern sustainability goals and environmental regulations. The scalability is further evidenced by the successful demonstration of the process in larger reactor volumes (e.g., 100L scale examples in the patent), proving that the heat transfer and mixing requirements can be met in standard stainless steel reactors without the need for exotic high-pressure equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on critical process parameters and quality control measures. Understanding these factors is essential for technology transfer teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the new ring-opening method superior to the conventional ammonia gas method?
A: The conventional method relies on ammonia gas, where concentration control is difficult; low concentrations fail to react while high concentrations generate unwanted amide by-products. The new method utilizes sodium methoxide in methanol, which ensures smooth ring-opening without amide formation, significantly improving safety and product purity.
Q: What is the critical temperature control parameter during the ring-opening step?
A: The ring-opening reaction must be strictly maintained between -5°C and 10°C. Exceeding 10°C leads to partial hydrolysis of the intermediate, and temperatures above 20°C can result in the loss of up to half the product yield due to hydrolysis.
Q: How does this process impact the scalability of Glasdegib production?
A: By eliminating the need for hazardous ammonia gas handling and utilizing standard solvent systems like acetonitrile and methanol, the process simplifies equipment requirements and waste treatment, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glasdegib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the timely delivery of life-saving oncology therapies. Our technical team has extensively analyzed the process described in CN113443995B and possesses the expertise to implement this optimized route with precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Glasdegib intermediate meets the exacting standards required for global regulatory filings.
We invite you to collaborate with us to leverage this advanced synthetic technology for your API projects. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this improved process can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming production campaigns.
