Revolutionizing Halogenated Arene Production with Green Aqueous Copper Catalysis for Commercial Scale
Introduction to Advanced Halogenation Technology
The chemical industry is currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, particularly in the production of critical building blocks like halogenated aromatic compounds. A pivotal development in this arena is detailed in patent CN102295497A, which discloses a novel synthesis method utilizing water as the primary solvent. This technology represents a significant departure from traditional protocols that rely heavily on volatile organic compounds and harsh reaction conditions. By leveraging a copper-ammonia catalytic system under ambient oxygen pressure, this process achieves high-yield conversion of arylboronic acids into valuable halogenated arenes. For R&D directors and procurement specialists alike, this innovation offers a compelling value proposition: it drastically reduces the environmental footprint while maintaining rigorous standards for purity and functional group compatibility. The ability to operate at room temperature (15-25°C) not only enhances safety profiles but also lowers energy consumption, making it an ideal candidate for cost reduction in fine chemical manufacturing.
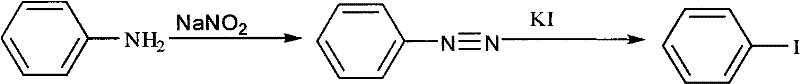
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of iodobenzene and related halogenated aromatics has relied predominantly on the diazotization of aniline followed by substitution. As illustrated in the conventional pathway, this method imposes severe operational constraints, requiring strict temperature control below 12°C to prevent the decomposition of unstable diazonium intermediates. Furthermore, the generation of nitrogen gas necessitates careful pressure management and extended reaction times to ensure complete evolution of the gas. From a supply chain perspective, the reliance on strong mineral acids like 30% hydrochloric acid and the subsequent need for neutralization with sodium hydroxide create significant waste streams that complicate disposal and increase operational costs. Moreover, this traditional route exhibits poor tolerance for sensitive functional groups; substituents such as methoxy or nitro groups often lead to side reactions or decomposition, limiting the scope of accessible derivatives and forcing manufacturers to seek more expensive, multi-step alternatives for complex intermediates.
The Novel Approach
In stark contrast, the methodology described in patent CN102295497A introduces a robust and versatile alternative that utilizes readily available arylboronic acids as starting materials. The core of this innovation lies in the use of water as the reaction medium, coupled with inexpensive cuprous oxide (Cu2O) or cuprous halides as catalysts and ammonia as a ligand. 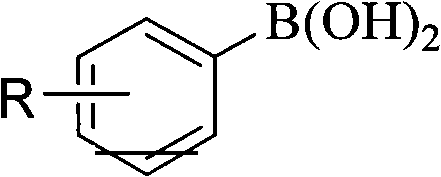 This aqueous system operates under mild conditions, typically between 15-25°C and atmospheric pressure, eliminating the need for cryogenic cooling or high-pressure equipment. The transition from organic solvents to water not only aligns with green chemistry principles but also simplifies the isolation process, as the products can often be extracted directly without complex workup procedures. This approach effectively bypasses the stability issues associated with diazonium salts, providing a safer and more reliable pathway for the commercial scale-up of complex pharmaceutical intermediates.
This aqueous system operates under mild conditions, typically between 15-25°C and atmospheric pressure, eliminating the need for cryogenic cooling or high-pressure equipment. The transition from organic solvents to water not only aligns with green chemistry principles but also simplifies the isolation process, as the products can often be extracted directly without complex workup procedures. This approach effectively bypasses the stability issues associated with diazonium salts, providing a safer and more reliable pathway for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Copper-Ammonia Catalyzed Substitution
The efficacy of this synthesis relies on the formation of an active copper-ammonia complex in situ, which facilitates the substitution of the boronic acid group with a halogen atom. The reaction mechanism involves the coordination of the copper catalyst with ammonia ligands, creating a soluble species capable of activating the arylboronic acid substrate. Crucially, the presence of molecular oxygen is required to maintain the catalytic cycle, likely by regulating the oxidation state of the copper species between Cu(I) and Cu(II). Experimental data indicates that inert atmospheres, such as nitrogen protection, severely inhibit the reaction, yielding only trace amounts of product. This dependence on air suggests an oxidative coupling mechanism that is both unique and highly efficient. The catalytic system demonstrates remarkable versatility, accommodating a wide range of halogen sources including iodides, bromides, and chlorides, thereby allowing for the precise installation of different halogen atoms based on downstream synthetic requirements.
Furthermore, the mechanistic robustness of this system is evidenced by its exceptional tolerance towards diverse functional groups on the aromatic ring. 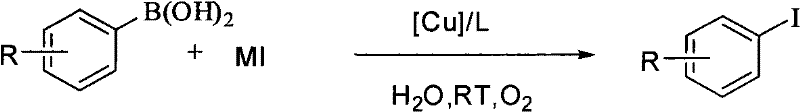 Whether the substrate contains electron-withdrawing groups like nitro or aldehyde moieties, or electron-donating groups such as methoxy and methyl, the reaction proceeds with high fidelity and yield. For instance, the synthesis of 3-iodonitrobenzene and 4-iodobenzaldehyde proceeds smoothly without the reduction of the nitro group or oxidation of the aldehyde, issues that frequently plague other metal-catalyzed protocols. This high level of chemoselectivity is paramount for R&D teams designing multi-step syntheses for active pharmaceutical ingredients (APIs), as it minimizes the formation of impurities and reduces the burden on purification processes. The ability to recycle the catalytic mother liquor further underscores the stability of the copper-ammonia complex, ensuring consistent performance over multiple batches.
Whether the substrate contains electron-withdrawing groups like nitro or aldehyde moieties, or electron-donating groups such as methoxy and methyl, the reaction proceeds with high fidelity and yield. For instance, the synthesis of 3-iodonitrobenzene and 4-iodobenzaldehyde proceeds smoothly without the reduction of the nitro group or oxidation of the aldehyde, issues that frequently plague other metal-catalyzed protocols. This high level of chemoselectivity is paramount for R&D teams designing multi-step syntheses for active pharmaceutical ingredients (APIs), as it minimizes the formation of impurities and reduces the burden on purification processes. The ability to recycle the catalytic mother liquor further underscores the stability of the copper-ammonia complex, ensuring consistent performance over multiple batches.
How to Synthesize Halogenated Aromatic Compounds Efficiently
Implementing this technology in a laboratory or pilot plant setting involves a straightforward protocol that prioritizes safety and efficiency. The process begins with the preparation of the catalytic system, where cuprous oxide is dissolved in aqueous ammonia under aerobic conditions to generate the active species. This pre-formation step is critical, as comparative examples show that adding reagents directly without pretreatment can result in significantly lower yields. Once the catalyst is ready, it is introduced to an aqueous suspension of the arylboronic acid and the chosen alkali metal halide salt. The reaction is then allowed to stir at room temperature for a period ranging from 24 to 48 hours, depending on the specific substrate reactivity.
- Prepare the catalytic system by mixing cuprous oxide (Cu2O) with aqueous ammonia (NH3-H2O) in the presence of oxygen to form the active copper-ammonia complex.
- Combine the arylboronic acid substrate and alkali metal halide salt (e.g., KI) in water within a reaction vessel equipped with stirring.
- Add the pre-formed catalyst system to the reaction mixture and stir at room temperature (15-25°C) for 24-48 hours under an air atmosphere.
- Upon completion, extract the product using dichloromethane, wash the organic phase with alkali solution, and concentrate to isolate the pure halogenated aromatic compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous copper-catalyzed method presents tangible economic and logistical benefits that extend beyond simple yield improvements. The most immediate impact is seen in raw material costs; replacing expensive organic solvents like DMF or acetonitrile with water results in substantial cost savings in solvent procurement and waste disposal. Additionally, the use of commodity chemicals such as cuprous oxide and ammonia, rather than precious metal catalysts like palladium or platinum, significantly lowers the bill of materials. This shift not only reduces the direct cost of goods sold but also mitigates the risk associated with the price volatility of noble metals. The simplified workup procedure, which avoids complex distillation or chromatography in many cases, further contributes to cost reduction in manufacturing by shortening production cycles and reducing labor intensity.
- Cost Reduction in Manufacturing: The elimination of volatile organic solvents and precious metal catalysts fundamentally alters the cost structure of halogenated arene production. By utilizing water as the sole solvent, facilities can avoid the capital expenditure associated with solvent recovery systems and explosion-proof infrastructure. The catalyst system itself is composed of earth-abundant copper, which is orders of magnitude cheaper than traditional cross-coupling catalysts. Moreover, the patent data confirms that the catalytic mother liquor can be recycled multiple times without significant loss of activity, effectively amortizing the catalyst cost over numerous batches. This recyclability translates directly into improved margins and a more predictable cost base for long-term supply contracts.
- Enhanced Supply Chain Reliability: Sourcing high-purity arylboronic acids is generally more stable and reliable than managing the supply chains for hazardous diazonium precursors or specialized organometallic reagents. The mild reaction conditions (15-25°C) mean that production is less susceptible to disruptions caused by utility failures, such as loss of cooling water or steam, which are critical in traditional low-temperature diazotization processes. This robustness ensures consistent delivery schedules and reduces the likelihood of batch failures due to thermal excursions. Furthermore, the high functional group tolerance allows for a more flexible sourcing strategy, as a wider variety of commercially available boronic acids can be utilized without extensive custom synthesis.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is facilitated by the absence of exothermic hazards and gas evolution issues common in diazotization. The aqueous nature of the reaction simplifies heat transfer and mixing, allowing for the use of standard stainless steel reactors without the need for specialized glass-lined or Hastelloy equipment. From an environmental compliance standpoint, the reduction in organic waste and the use of non-toxic reagents streamline the permitting process and reduce the regulatory burden on manufacturing sites. This alignment with increasingly stringent environmental regulations future-proofs the supply chain against potential legislative changes regarding solvent emissions and heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering clarity on operational parameters and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this method into existing production workflows.
Q: Can the copper catalyst system be recycled in this synthesis method?
A: Yes, the patent demonstrates that the catalytic system can be recycled without special purification. After extracting the product, adding fresh reactants to the mother liquor allows the reaction to proceed again with maintained efficiency.
Q: What is the functional group tolerance of this aqueous copper-catalyzed reaction?
A: The method exhibits high tolerance for various functional groups on the aromatic ring, including nitro, methoxy, chloro, and aldehyde groups, which are often problematic in traditional diazotization methods.
Q: Why is water preferred over organic solvents for this halogenation process?
A: Water serves as the cheapest and most environmentally benign solvent. Comparative data in the patent shows that organic solvents like DMF result in trace yields, while water facilitates high conversion rates and simplifies downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Aromatic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the water-based copper catalysis technology described in patent CN102295497A for the production of high-purity halogenated aromatic compounds. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative academic research into robust, commercial-scale manufacturing processes. Our facilities are equipped to handle diverse synthetic pathways, ranging from small-scale kilogram campaigns for clinical trials to large-scale production exceeding 100 MT annually. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch of pharmaceutical intermediate or fine chemical adheres to the highest quality standards required by global regulatory bodies.
We invite procurement leaders and R&D directors to collaborate with us to leverage this green chemistry advantage for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this aqueous protocol can optimize your bill of materials. We encourage you to contact our technical procurement team to request specific COA data for our halogenated arene portfolio and to discuss route feasibility assessments for your upcoming projects. Together, we can drive efficiency and sustainability in the manufacturing of critical chemical intermediates.
