Advanced Synthetic Strategy for LND1088 Linker Enabling Scalable ADC Production
The rapidly evolving landscape of oncology therapeutics has placed Antibody-Drug Conjugates (ADCs) at the forefront of targeted cancer treatment, driving an urgent demand for high-quality, stable linkers that ensure precise drug delivery. Patent CN113264983A discloses a groundbreaking synthetic method for LND1088, a sophisticated linker payload conjugate comprising a DBCO moiety, a PEG4 spacer, a GGFG peptide sequence, and the potent topoisomerase I inhibitor Exatecan derivative (Dxd). This innovation addresses critical bottlenecks in ADC manufacturing by streamlining the convergence of the cytotoxic payload and the click-chemistry handle. The structural complexity of LND1088 requires meticulous control over stereochemistry and functional group compatibility, which this new protocol achieves through a refined sequence of peptide couplings and deprotections. 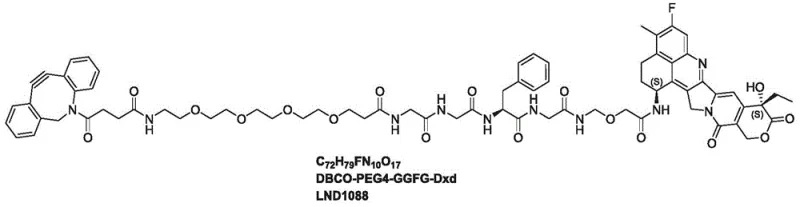
For R&D directors evaluating supply chain partners, the ability to source such complex intermediates with consistent purity is paramount. The disclosed method not only enhances the chemical integrity of the final product but also simplifies the downstream processing required to meet stringent regulatory standards. By integrating a DBCO group, the linker facilitates strain-promoted azide-alkyne cycloaddition (SPAAC), a bioorthogonal reaction essential for next-generation site-specific antibody conjugation. This technical advancement underscores the importance of adopting modern synthetic strategies that align with the rigorous demands of clinical-grade ADC production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of similar GGFG-based linkers has been plagued by inefficient protection group strategies that compromise overall yield and scalability. As illustrated in the prior art pathways, conventional methods often rely on Boc-protected amino acid sequences which necessitate harsh acidic conditions for deprotection, potentially risking the stability of the sensitive exatecan derivative. 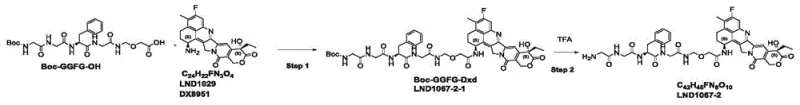
Furthermore, the traditional route involves the synthesis of an intermediate designated as LND1067-2-1, which presents significant purification challenges due to its physicochemical properties. Comparative data within the patent reveals that these legacy processes suffer from suboptimal conversion rates, with the final coupling step yielding as low as 35.7% in some embodiments. The accumulation of impurities at each stage necessitates extensive chromatographic purification, leading to excessive solvent consumption and prolonged cycle times. For procurement managers, these inefficiencies translate directly into higher cost of goods sold (COGS) and unreliable supply timelines, making the conventional approach economically unsustainable for commercial-scale ADC programs.
The Novel Approach
The innovative strategy presented in the patent circumvents these obstacles by employing an Fmoc-based protection scheme and optimizing the order of assembly to avoid the problematic LND1067-2-1 intermediate entirely. This novel approach utilizes LND1067-L1 (Fmoc-GGFG-A) as the starting scaffold, which reacts efficiently with the DX8951 payload under mild conditions mediated by HATU and DIPEA. 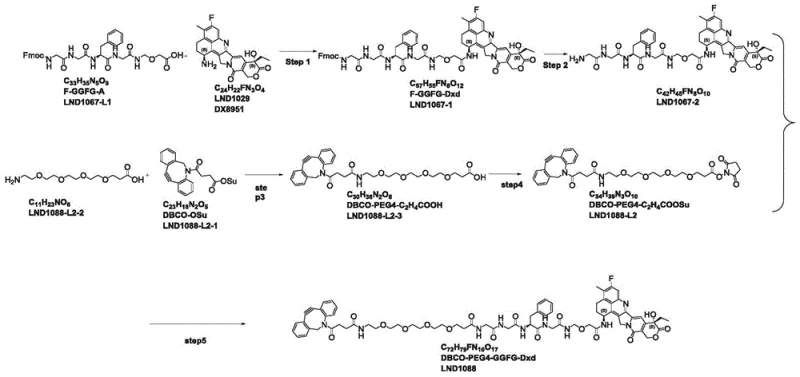
By shifting to an Fmoc strategy, the deprotection step can be performed using diethylamine (DEA) in DMF, a much milder process that preserves the integrity of the drug-linker complex. The patent data highlights a dramatic improvement in efficiency, with the optimized route achieving a final yield of 62.5% and an HPLC purity of 98.6%. This streamlined workflow not only reduces the number of unit operations but also enhances the robustness of the process, making it highly attractive for reliable antibody-drug conjugate linker supplier partnerships seeking to minimize production risks.
Mechanistic Insights into Peptide Coupling and Deprotection
The core of this synthetic breakthrough lies in the precise control of amide bond formation and orthogonal deprotection. In the initial step, the carboxylic acid of the Fmoc-GGFG-A precursor is activated by HATU (1-[Bis(dimethylamino)methylene]-1H-1,2,3-triazolo[4,5-b]pyridinium 3-oxid hexafluorophosphate), forming a highly reactive O-acylisourea intermediate. This species is immediately attacked by the amine group of the DX8951 payload in the presence of DIPEA, which acts as a base to scavenge the generated acid. The use of anhydrous DMF as the solvent is critical here, as it ensures complete solubility of the hydrophobic exatecan derivative, thereby maximizing collision frequency and reaction kinetics. The reaction is conducted at low temperatures (-5°C to 0°C) to suppress racemization and minimize side reactions such as epimerization of the chiral centers in the peptide backbone.
Following the coupling, the removal of the Fmoc group is executed using diethylamine, which proceeds via a beta-elimination mechanism to release the free amine necessary for the subsequent linker attachment. This step is particularly elegant because it avoids the use of strong acids like TFA in the early stages, which could prematurely cleave acid-sensitive moieties or degrade the payload. The resulting intermediate, LND1067-2, is then coupled with the pre-activated DBCO-PEG4-NHS ester (LND1088-L2). The NHS ester functionality ensures rapid and quantitative acylation of the terminal amine, forming the stable amide bond that connects the click-chemistry handle to the drug payload. This mechanistic precision is what allows for the high purity profiles observed, reducing the burden on downstream purification and ensuring a clean impurity profile for clinical applications.
How to Synthesize LND1088 Efficiently
The synthesis of LND1088 is a multi-step process that requires careful attention to stoichiometry, temperature control, and purification protocols to achieve the high yields reported in the patent literature. The procedure begins with the preparation of the drug-peptide intermediate, followed by the independent synthesis of the DBCO-linker arm, and concludes with their convergent coupling. Each stage utilizes standard organic synthesis techniques but is optimized specifically for the solubility and reactivity constraints of the exatecan derivative. Operators must ensure strict moisture control during the activation steps to prevent hydrolysis of the active esters.
- React LND1067-L1 with DX8951 using HATU and DIPEA in DMF to form the protected intermediate LND1067-1.
- Perform Fmoc deprotection on LND1067-1 using diethylamine (DEA) in DMF to obtain the free amine LND1067-2.
- Synthesize the DBCO-PEG4 acid component (LND1088-L2) by coupling DBCO-OSu with the PEG amine followed by NHS activation.
- Couple the activated linker LND1088-L2 with the peptide-drug intermediate LND1067-2 using DIPEA to yield the final LND1088 product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the substantial reduction of raw material waste and processing time, driven by the elimination of the low-yielding LND1067-2-1 intermediate. By bypassing this bottleneck, manufacturers can significantly reduce the volume of solvents and chromatography media required per kilogram of final product. This efficiency gain translates directly into cost reduction in antibody-drug conjugate manufacturing, allowing for more competitive pricing models without compromising on quality. Furthermore, the higher overall yield means that less starting material is needed to produce the same amount of API intermediate, insulating the supply chain from fluctuations in the cost of precious precursors like the exatecan derivative.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for complex isolation and purification of unstable intermediates, which traditionally consume significant resources. By achieving a final yield nearly double that of comparative methods, the process drastically lowers the effective cost per gram of the linker. The use of common reagents like HATU and DIPEA, rather than exotic catalysts, further ensures that material costs remain predictable and manageable. Additionally, the reduced number of processing steps lowers labor and utility costs associated with reactor occupancy and solvent recovery.
- Enhanced Supply Chain Reliability: A robust synthetic route is less prone to batch failures, ensuring a consistent supply of critical ADC components. The method's tolerance to standard industrial conditions means that production can be scaled up with minimal risk of deviation. This reliability is crucial for maintaining the continuity of clinical trials and commercial launches, where delays in linker supply can halt the entire drug development timeline. The ability to source high-purity LND1088 from a qualified partner mitigates the risk of supply disruptions caused by complex, finicky chemistry.
- Scalability and Environmental Compliance: The patent explicitly notes that the method is easy to amplify, a key requirement for commercial scale-up of complex pharmaceutical intermediates. The process avoids the generation of hazardous waste streams associated with harsh acidic deprotections, aligning better with modern environmental, health, and safety (EHS) standards. Reduced solvent usage and simpler workup procedures contribute to a smaller environmental footprint, facilitating regulatory approval and supporting sustainability goals within the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of the LND1088 linker. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for stakeholders evaluating the feasibility of incorporating this linker into their ADC development pipelines.
Q: How does the new synthetic method improve the yield of LND1088 compared to conventional routes?
A: The novel method described in patent CN113264983A significantly improves the overall yield by avoiding the synthesis of the difficult-to-purify intermediate LND1067-2-1. Experimental data demonstrates a final yield increase from approximately 35.7% in comparative examples to 62.5% using the optimized Fmoc-based strategy.
Q: What are the critical purification techniques used to ensure high purity of the LND1088 linker?
A: The process relies heavily on medium-pressure reverse-phase chromatography using acetonitrile and water systems with acidic modifiers (TFA or AcOH). This technique effectively removes unreacted starting materials and side products, achieving HPLC purities exceeding 98.6% for the final API intermediate.
Q: Is this synthetic route suitable for large-scale commercial manufacturing of ADC linkers?
A: Yes, the patent explicitly states that the method is easy to amplify. By utilizing robust reagents like HATU and standard solvents such as DMF and DCM, and eliminating complex isolation steps for unstable intermediates, the process is designed for seamless transition from laboratory scale to multi-kilogram commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable LND1088 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of ADC therapies hinges on the availability of high-quality linkers produced via robust, scalable pathways. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of global pharmaceutical partners. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of LND1088 meets the highest standards of identity, strength, and purity. Our commitment to technical excellence ensures that your critical drug substance is supported by a supply chain you can trust.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation ADC programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how our optimized processes can enhance your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to clinical success with reliable, high-performance linker solutions.
