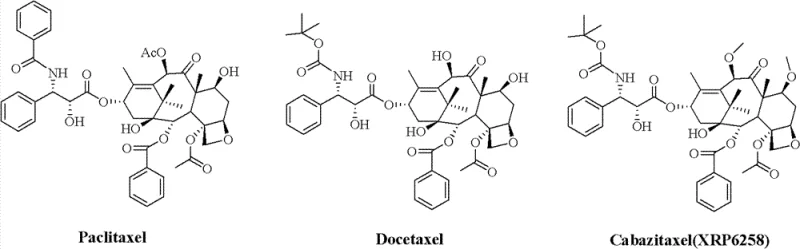
Revolutionizing Cabazitaxel Production: Advanced Synthetic Routes for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for second-generation taxanes, particularly Cabazitaxel (XRP6258), to address multidrug resistance in oncology treatments. Patent CN103012328A introduces a transformative methodology that fundamentally alters the production landscape of this critical anticancer agent. By leveraging a novel combination of Pummerer Rearrangement and Swern Oxidation, this technology bypasses the historical bottlenecks associated with C-7 and C-10 hydroxyl modifications. For R&D Directors and Supply Chain Heads, this represents a pivotal shift from low-yield, multi-step alkylation processes to a streamlined, high-efficiency protocol. The strategic implementation of methylthio-methylene (MTM) groups as temporary protecting and activating functionalities allows for unprecedented regioselectivity. This technical breakthrough ensures that the production of high-purity Cabazitaxel intermediates is no longer constrained by the inefficiencies of prior art, paving the way for reliable pharmaceutical intermediates supplier networks to deliver consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semisynthesis of Cabazitaxel has been plagued by significant chemical inefficiencies that hinder cost reduction in API manufacturing. Traditional routes typically rely on direct alkylation of 10-deacetylbaccatin III (10-DAB) using methyl iodide or methyl sulfate under strongly basic conditions. This approach suffers from severe drawbacks, primarily the susceptibility of the C-7 hydroxyl group to epimerization via anti-aldol condensation. Consequently, the alkylation at the C-7 position becomes exceedingly difficult to control, resulting in abysmal yields often ranging between 10% to 20%. Furthermore, these legacy methods necessitate complex reaction systems and tedious liquid-phase separations that are ill-suited for commercial scale-up of complex taxanes. The reliance on hazardous alkylating agents and the generation of substantial chemical waste further exacerbate the environmental and safety burdens on production facilities. For procurement managers, these inefficiencies translate into volatile pricing and unpredictable availability of critical starting materials.
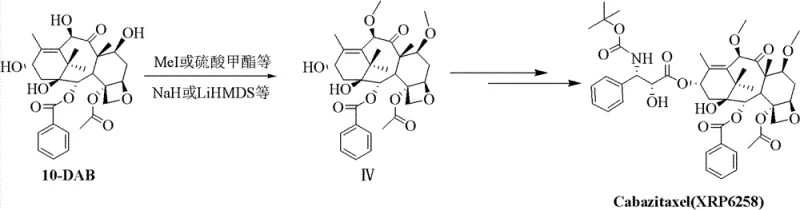
The Novel Approach
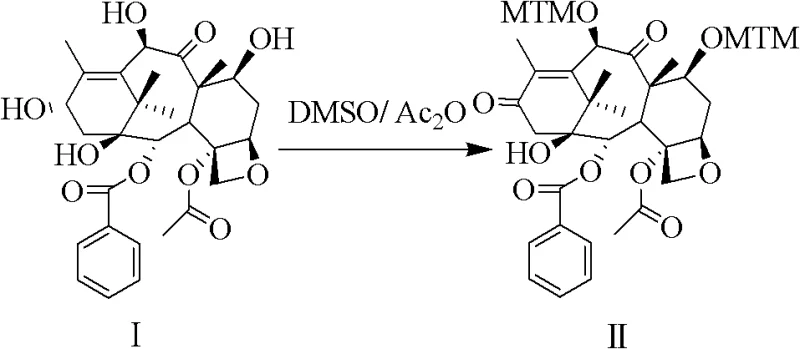
In stark contrast, the patented methodology introduces a sophisticated strategy that circumvents these inherent chemical limitations through a clever manipulation of sulfur chemistry. Instead of direct alkylation, the process employs a simultaneous Pummerer Rearrangement and Swern Oxidation using dimethyl sulfoxide (DMSO) and acetic anhydride. This innovative sequence converts the C-7 and C-10 hydroxyls into methylthio-methylene (MTM) ethers while oxidizing the C-13 hydroxyl, achieving high regioselectivity without the risk of epimerization. The subsequent one-pot desulfurization using Raney Nickel and hydrogen cleanly removes the sulfur groups to reveal the desired methyl ethers. This approach not only drastically simplifies the operational workflow but also significantly enhances the overall yield and purity profile. By eliminating the need for harsh bases and unstable intermediates, this novel approach offers a safer, more sustainable pathway for the commercial production of next-generation taxane therapeutics.
Mechanistic Insights into Pummerer-Swern Tandem Reaction
The core of this technological advancement lies in the precise execution of the tandem Pummerer-Swern reaction sequence, which serves as the foundation for constructing the Cabazitaxel nucleus. The mechanism initiates with the activation of DMSO by acetic anhydride, generating a reactive sulfonium species that selectively targets the C-7 and C-10 hydroxyl groups of the baccatin III core. This transformation installs the methylthio-methylene (MTM) protecting groups with exceptional fidelity, effectively masking the sensitive hydroxyls while preventing unwanted side reactions at the C-13 position. The concurrent oxidation of the C-13 hydroxyl to a ketone further stabilizes the intermediate, setting the stage for the subsequent reduction steps. For technical teams, understanding this mechanistic nuance is crucial, as it highlights the importance of reagent stoichiometry and temperature control in maintaining the integrity of the taxane skeleton. The ability to perform these transformations in a coordinated manner reduces the number of isolation steps, thereby minimizing material loss and maximizing throughput efficiency.
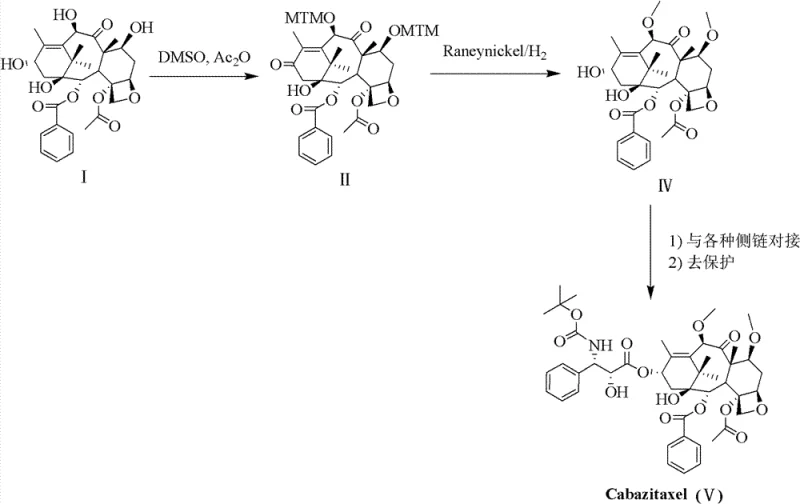
Following the formation of the MTM-protected intermediate, the process employs a catalytic hydrogenolysis step that is critical for impurity control and final product quality. The use of Raney Nickel under hydrogen pressure facilitates the cleavage of the carbon-sulfur bonds, converting the MTM groups into the desired methyl ethers while simultaneously reducing the C-13 ketone back to the hydroxyl configuration. This one-pot operation is particularly advantageous because it avoids the exposure of the sensitive taxane core to acidic or basic conditions that could trigger degradation or rearrangement. The result is a nucleus intermediate with a remarkably clean impurity profile, requiring minimal purification before side-chain coupling. This level of control over the impurity spectrum is vital for meeting the stringent regulatory standards required for oncology drugs. It ensures that the final active pharmaceutical ingredient maintains the necessary safety and efficacy profiles demanded by global health authorities.
How to Synthesize Cabazitaxel Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction conditions outlined in the patent to ensure reproducibility and safety. The process begins with the dissolution of 10-deacetylbaccatin III in dry DMSO, followed by the controlled addition of acetic anhydride and a catalytic amount of acid. Maintaining the reaction temperature between 25°C and 60°C is essential to drive the Pummerer rearrangement to completion without degrading the substrate. Once the MTM intermediate is formed, it proceeds directly to the desulfurization step without intermediate purification, showcasing the telescoped nature of this efficient route. The detailed standardized synthesis steps see the guide below for specific molar ratios and workup procedures.
- Perform simultaneous Pummerer Rearrangement and Swern Oxidation on 10-deacetylbaccatin III using DMSO and acetic anhydride.
- Execute one-pot desulfurization of the intermediate using Raney Nickel and hydrogen to obtain the protected nucleus.
- Couple the nucleus with protected beta-lactam side chains and remove protecting groups to finalize Cabazitaxel.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented synthesis offers profound benefits that directly address the pain points of modern pharmaceutical supply chains. By replacing low-yielding alkylation steps with a high-efficiency rearrangement protocol, manufacturers can achieve substantial cost savings in raw material consumption and waste disposal. The elimination of complex purification sequences, such as multiple column chromatography runs, significantly reduces processing time and solvent usage. This streamlining of the manufacturing process enhances supply chain reliability by shortening production cycles and increasing the overall capacity of existing facilities. For procurement managers, this translates into a more stable supply of high-purity Cabazitaxel intermediates, mitigating the risks associated with production bottlenecks and market shortages.
- Cost Reduction in Manufacturing: The transition from traditional alkylation to the Pummerer-based route eliminates the need for expensive and hazardous alkylating agents like methyl iodide. Furthermore, the high yield of the key steps reduces the amount of starting 10-DAB required per kilogram of final product, driving down the cost of goods sold. The simplified workup procedures also lower labor and utility costs associated with extended reaction times and complex separations. These cumulative efficiencies create a robust economic model that supports competitive pricing strategies in the generic and branded drug markets.
- Enhanced Supply Chain Reliability: The use of commercially available and stable reagents such as DMSO, acetic anhydride, and Raney Nickel ensures that the supply chain is not vulnerable to the fluctuations of specialized chemical markets. The robustness of the reaction conditions allows for consistent production outcomes, reducing the rate of batch failures and reworks. This reliability is critical for maintaining continuous supply to downstream formulation partners, ensuring that patient access to life-saving medications is never compromised by manufacturing delays.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor equipment and avoiding extreme conditions that pose safety risks at large volumes. The reduction in solvent intensity and the avoidance of heavy metal catalysts in the key steps align with green chemistry principles, facilitating easier regulatory approval and environmental compliance. This sustainability aspect is increasingly important for pharmaceutical companies aiming to reduce their carbon footprint and meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. They are derived from the specific advantages and operational details described in the patent documentation. Understanding these aspects helps stakeholders evaluate the feasibility of adopting this route for their own production needs.
Q: How does this new method improve yield compared to traditional alkylation?
A: Traditional methods using methyl iodide often suffer from low yields (10-20%) due to epimerization. This patent utilizes a Pummerer rearrangement strategy that achieves significantly higher regioselectivity and overall yield.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method eliminates complex liquid-phase separation steps and uses common reagents like Raney Nickel, making it highly scalable and cost-effective for commercial manufacturing.
Q: What are the purity implications of the one-pot desulfurization step?
A: The one-pot desulfurization simplifies purification, reducing the need for extensive column chromatography and resulting in a cleaner intermediate profile suitable for GMP production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cabazitaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for complex oncology intermediates like Cabazitaxel. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. We operate with stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the highest international standards. Our commitment to technological excellence allows us to offer partners a secure and high-quality source for their taxane requirements.
We invite you to collaborate with us to leverage this advanced synthesis for your product pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain resilience and profitability.
