Advanced Synthesis of Taspinine Biphenyl Derivatives for Scalable Antitumor Drug Manufacturing
Advanced Synthesis of Taspinine Biphenyl Derivatives for Scalable Antitumor Drug Manufacturing
The pharmaceutical industry is constantly seeking robust synthetic pathways for novel antitumor agents that balance efficacy with manufacturability. Patent CN101735100A introduces a significant advancement in this domain by disclosing a series of taspinine biphenyl derivatives and their preparation methods. These compounds, characterized by a 5,5'-dimethoxy-6,6'-dihydroxybiphenyl-2,2'-dicarboxamide core, exhibit potent antitumor activity, making them highly valuable candidates for oncology drug development. The innovation lies not only in the biological potential of the new chemical entities but also in the practicality of the synthetic route, which avoids the pitfalls of natural product extraction. By utilizing a step-wise organic synthesis approach starting from isovanillin, the process ensures high purity and structural consistency, addressing the critical needs of R&D directors for reliable reference standards and clinical trial materials.
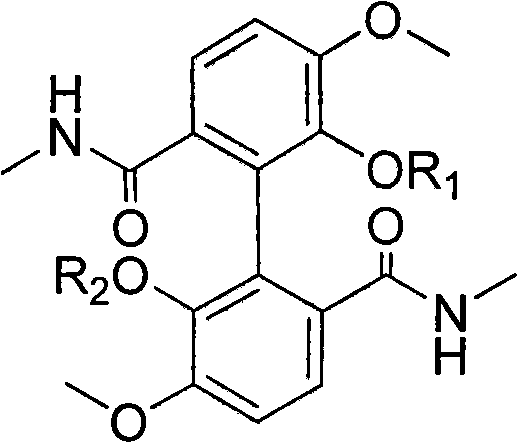
The core innovation described in the patent involves the strategic modification of the phenolic hydroxyl groups on the biphenyl scaffold. As illustrated in the structural overview, the R1 and R2 positions can be independently substituted with hydrogen, straight-chain alkyl groups terminated by secondary amines, or acetanilide moieties substituted with halogens. This modularity allows medicinal chemists to explore a wide chemical space to optimize pharmacokinetic properties and target affinity. The ability to systematically vary these substituents through controlled etherification reactions provides a versatile platform for structure-activity relationship (SAR) studies, which is essential for identifying lead compounds with superior therapeutic indices in the competitive landscape of cancer treatment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining aporphine alkaloids like taspinine and its derivatives has relied heavily on extraction from natural plant sources. This conventional approach is fraught with significant logistical and technical challenges that hinder large-scale pharmaceutical production. Natural extraction processes are inherently variable, dependent on seasonal harvests, geographical origin, and plant age, leading to inconsistent batch-to-batch quality. Furthermore, the concentration of active alkaloids in plant biomass is often extremely low, necessitating the processing of massive quantities of raw material to isolate minute amounts of the target compound. This results in exorbitant costs and a supply chain that is vulnerable to ecological and agricultural disruptions. Additionally, the purification of natural extracts is notoriously difficult due to the presence of numerous structurally similar impurities, often requiring extensive chromatographic separation that is not feasible for commercial scale-up.
The Novel Approach
In stark contrast, the synthetic methodology outlined in CN101735100A offers a deterministic and scalable alternative that bypasses the uncertainties of natural sourcing. The route begins with isovanillin, a commercially abundant and inexpensive starting material, ensuring a stable and cost-effective supply chain foundation. The synthesis employs well-established organic transformations such as bromination, oxidation, and amide formation, which are easily controlled and monitored in a standard chemical manufacturing facility. By constructing the biphenyl core through a copper-mediated Ullmann coupling, the process achieves high regioselectivity and yield without the need for exotic catalysts or extreme pressures. This shift from extraction to total synthesis empowers procurement managers to secure long-term supply agreements with predictable pricing, while providing R&D teams with the flexibility to produce gram-to-kilogram quantities of diverse derivatives for preclinical evaluation.
Mechanistic Insights into Cu-Mediated Ullmann Coupling and Etherification
The construction of the biaryl backbone is the pivotal step in this synthesis, achieved through a classic Ullmann coupling reaction. In this mechanism, two molecules of 4-methoxy-3-benzyloxy-2-bromobenzamide undergo reductive dimerization in the presence of activated copper powder in anhydrous N,N-dimethylformamide (DMF). The reaction is conducted under a nitrogen atmosphere at elevated temperatures ranging from 150°C to 160°C to overcome the activation energy barrier for the carbon-carbon bond formation. The copper surface facilitates the oxidative addition of the aryl bromide, followed by transmetallation and reductive elimination to forge the sterically hindered biphenyl linkage. This step is critical as it establishes the rigid spatial arrangement required for the molecule's biological activity. The use of stoichiometric copper powder, rather than expensive palladium catalysts, significantly reduces the metal burden in the final product, simplifying downstream purification and lowering the overall cost of goods sold (COGS).
Following the formation of the biphenyl core and subsequent deprotection of the benzyl groups via catalytic hydrogenation, the final diversification is achieved through nucleophilic substitution. The phenolic hydroxyl groups act as nucleophiles, attacking electrophilic alkyl halides or halogen-substituted acetanilides in the presence of a base like anhydrous potassium carbonate. This etherification proceeds smoothly in solvents such as anhydrous acetone or ethanol at moderate temperatures (56°C to 78°C). The choice of base and solvent is crucial for minimizing side reactions such as O-alkylation versus N-alkylation or hydrolysis of the amide bonds. The patent specifies precise molar ratios, such as 1:1 for mono-substitution or 1:2.2 for di-substitution, allowing for precise control over the substitution pattern. This level of control ensures that the impurity profile remains manageable, a key requirement for regulatory compliance in API manufacturing.
How to Synthesize Taspinine Biphenyl Derivatives Efficiently
The synthesis of these high-value intermediates requires a disciplined approach to reaction conditions and workup procedures to ensure optimal yield and purity. The process integrates seven distinct chemical transformations, each optimized to maximize efficiency while maintaining safety. From the initial bromination of isovanillin at controlled low temperatures to the final etherification steps, every stage contributes to the overall robustness of the manufacturing protocol. For detailed operational parameters, including specific reagent grades, stirring rates, and isolation techniques, operators should refer to the standardized synthesis guidelines derived from the patent data.
- Bromination of isovanillin using iron powder and liquid bromine in acetic acid at 23-25°C to obtain 2-bromoisovanillin.
- Benzyl protection of the hydroxyl group followed by oxidation of the aldehyde to carboxylic acid using sodium chlorite and hydrogen peroxide.
- Conversion to benzamide via thionyl chloride and methylamine, followed by Ullmann coupling with copper powder to form the biphenyl core.
- Catalytic hydrogenation to remove benzyl groups, followed by etherification with halogen-substituted acetanilide or secondary amines to finalize the derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic route represents a strategic opportunity to de-risk the supply of critical oncology intermediates. The reliance on commodity chemicals like isovanillin, iron powder, and copper eliminates the bottleneck associated with specialized or scarce reagents. This accessibility translates directly into enhanced supply chain reliability, as raw materials can be sourced from multiple global vendors, preventing single-source dependency. Furthermore, the synthetic pathway is designed with scalability in mind; the reaction conditions do not require specialized high-pressure equipment or cryogenic cooling, allowing for seamless technology transfer from laboratory benchtop to multi-ton production reactors. This inherent scalability ensures that as clinical demand grows, manufacturing capacity can be expanded rapidly without significant capital expenditure on new infrastructure.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the elimination of expensive transition metal catalysts and the use of high-yielding reaction steps. By utilizing copper powder instead of palladium for the coupling reaction, the material costs are drastically reduced, and the need for rigorous heavy metal scavenging steps is minimized. Additionally, the high yields reported in the patent examples, such as 95% for the protection step and 97% for the deprotection step, indicate a highly efficient atom economy. This efficiency reduces waste generation and solvent consumption, leading to substantial cost savings in both raw materials and waste disposal, ultimately lowering the unit cost of the final intermediate.
- Enhanced Supply Chain Reliability: The synthetic route relies on stable, shelf-stable reagents that are not subject to the volatility of agricultural markets. Unlike natural extracts which can be compromised by crop failures or trade restrictions, the starting materials for this synthesis are produced via established industrial chemical processes. This stability guarantees a continuous supply flow, essential for maintaining uninterrupted clinical trial timelines and commercial production schedules. The robustness of the chemistry also means that the process is less sensitive to minor variations in operating conditions, reducing the risk of batch failures and ensuring consistent delivery of high-quality material to downstream customers.
- Scalability and Environmental Compliance: From an environmental and operational perspective, the process is favorable for large-scale implementation. The reactions generally proceed under atmospheric pressure and moderate temperatures, reducing energy consumption compared to high-energy processes. The workup procedures involve standard extraction and crystallization techniques that are easily adapted to industrial centrifuges and dryers. Moreover, the avoidance of toxic heavy metals like palladium simplifies the environmental compliance landscape, reducing the burden on wastewater treatment facilities. The ability to recycle solvents like ethyl acetate and ethanol further enhances the sustainability profile of the manufacturing process, aligning with modern green chemistry initiatives and corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these taspinine derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating these intermediates into your drug development pipeline.
Q: What are the primary advantages of this synthetic route over natural extraction?
A: Unlike natural extraction which suffers from low yields and complex purification, this synthetic method utilizes cheap, readily available raw materials like isovanillin and offers mild reaction conditions with high controllability, ensuring consistent quality and supply stability for pharmaceutical applications.
Q: What is the key reaction mechanism used to construct the biphenyl scaffold?
A: The core biphenyl structure is constructed via a classic Ullmann coupling reaction using activated copper powder in anhydrous DMF at elevated temperatures (150-160°C), which effectively links two aromatic rings to form the necessary 5,5'-dimethoxy-6,6'-substituted backbone.
Q: Can the substituents on the phenolic hydroxyl groups be customized?
A: Yes, the method allows for significant structural diversity. The phenolic hydroxyl groups can be etherified with various compounds containing halogen-substituted acetanilide or secondary amines (such as morpholine or piperidine derivatives), enabling the tuning of physicochemical properties for specific drug development needs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Taspinine Biphenyl Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the race to develop new antitumor therapies. Our team of expert chemists has thoroughly analyzed the synthetic route disclosed in CN101735100A and is fully prepared to support your project from milligram-scale screening to commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of taspinine biphenyl derivative meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to optimize this synthesis for your specific requirements. Whether you need custom analogs with different R1 and R2 substituents or large volumes of the standard derivative, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis and discuss how we can support your supply chain. We encourage potential partners to reach out for specific COA data and route feasibility assessments to ensure the perfect fit for your development timeline and budget constraints.
