Advanced Ethanol-Based Synthesis of 2,6-Dihydroxybenzoic Acid for Commercial Scale-Up
Introduction to Patent CN102211995A Technology
The chemical industry is constantly seeking more efficient pathways for synthesizing critical intermediates, and the technology disclosed in patent CN102211995A represents a significant leap forward in the production of 2,6-dihydroxybenzoic acid. This compound serves as a vital building block for high-potency herbicides and pharmaceutical agents, yet its traditional manufacturing routes have long been plagued by excessive energy consumption and suboptimal selectivity. The patented methodology introduces a refined carboxylation process utilizing resorcinol and carbon dioxide in the presence of an alkali metal salt, specifically leveraging absolute ethanol as a reaction medium to overcome historical limitations. By optimizing reaction parameters to a temperature range of 130 to 150°C and a pressure of 1.35 to 1.45 MPa, this innovation achieves a remarkable balance between operational safety and chemical efficiency. For global procurement teams and R&D directors, understanding this shift from harsh solvent-free conditions to a controlled liquid-phase system is essential for securing a reliable agrochemical intermediate supplier capable of meeting stringent quality and volume demands.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,6-dihydroxybenzoic acid has relied heavily on two distinct approaches, both of which present substantial drawbacks for large-scale commercial operations. The first approach, a solvent-free technique described in earlier patents such as CN:02111061.1, requires extreme reaction conditions including temperatures up to 160°C and pressures reaching 2.0 MPa for prolonged periods of up to 10 hours. These aggressive parameters not only impose severe stress on reactor equipment, necessitating expensive high-pressure vessels, but also result in a disappointingly low molar yield of the desired 2,6-isomer, often hovering around merely 30%. Furthermore, the majority of the output in these solvent-free processes consists of the less valuable 2,4-dihydroxybenzoic acid byproduct, creating significant waste management challenges and reducing overall atom economy. The second conventional method employs solvents like dimethylformamide (DMF) or water; however, water yields are abysmal at roughly 10%, while DMF introduces complex and energy-intensive separation steps that erode profit margins and complicate downstream processing.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN102211995A utilizes absolute ethanol as a strategic solvent to fundamentally alter the reaction landscape. This ethanol-based system allows the carboxylation of resorcinol to proceed efficiently at a reduced pressure of 1.45 MPa and a shortened reaction time of just 3 to 4 hours, dramatically improving equipment utilization rates and throughput. The use of ethanol facilitates a much cleaner separation of the target product from the reaction matrix, avoiding the difficult distillation hurdles associated with high-boiling solvents like DMF. Moreover, this method incorporates a sophisticated recycling loop where the unwanted 2,4-dihydroxybenzoic acid byproduct is recovered and reconverted into resorcinol, thereby closing the material loop and enhancing sustainability. This holistic improvement in process design translates directly into cost reduction in agrochemical intermediates manufacturing by lowering energy inputs and maximizing the value extracted from every kilogram of raw material fed into the reactor.
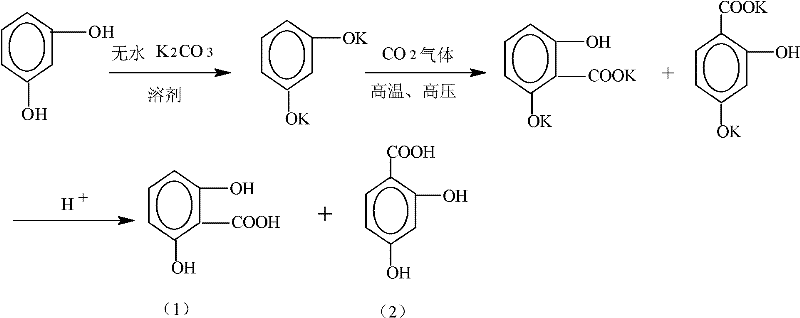
Mechanistic Insights into Ethanol-Mediated Carboxylation
The core of this technological advancement lies in the precise manipulation of the carboxylation mechanism, which is a variation of the classic Kolbe-Schmitt reaction tailored for dihydroxybenzenes. In this system, the alkali metal salt, preferably anhydrous potassium carbonate, acts as a base to deprotonate the hydroxyl groups of resorcinol, generating a highly nucleophilic phenoxide species in situ. The presence of absolute ethanol stabilizes these ionic intermediates and facilitates the attack of carbon dioxide on the aromatic ring, specifically directing the electrophilic substitution to the ortho-position relative to the hydroxyl groups to form the 2,6-isomer. The reaction mechanism involves the initial formation of a potassium salt intermediate which subsequently undergoes acidification to release the free acid. Understanding this mechanistic pathway is crucial for R&D teams aiming to replicate or scale this process, as the choice of base and the dryness of the ethanol solvent are critical factors in preventing side reactions and ensuring high selectivity for the target molecule over its 2,4-isomer counterpart.
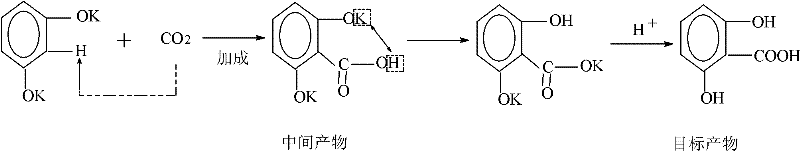
Furthermore, the control of impurities is intrinsically linked to the reaction conditions and the subsequent workup procedure defined in the patent. The formation of the 2,4-dihydroxybenzoic acid byproduct is inevitable in electrophilic aromatic substitution, but the patented process mitigates its impact through a multi-stage pH adjustment strategy during the post-treatment phase. By carefully controlling the acidity of the reaction mixture at different temperature stages, the process selectively precipitates inorganic salts and the unwanted 2,4-isomer before finally isolating the 2,6-dihydroxybenzoic acid at a lower pH. This fractional precipitation technique ensures that the final crude product is already enriched with the target compound before the final recrystallization step, which pushes the purity to 99%. Such rigorous impurity control mechanisms are vital for pharmaceutical applications where trace contaminants can compromise the safety and efficacy of the final drug product, making this synthesis route highly attractive for the production of high-purity pharmaceutical intermediates.
How to Synthesize 2,6-Dihydroxybenzoic Acid Efficiently
Implementing this synthesis route requires strict adherence to the optimized parameters regarding molar ratios and thermal profiles to ensure reproducibility and safety. The process begins with the grinding and mixing of resorcinol and anhydrous potassium carbonate in a 1:1 molar ratio, followed by dissolution in absolute ethanol at a mass ratio of 1:3 to create a homogeneous reaction mixture. Once charged into an autoclave, the system is pressurized with CO2 and heated to 130°C, maintaining a pressure of 1.45 MPa for 4 hours to drive the carboxylation to completion. Following the reaction, the mixture undergoes a complex but highly effective workup involving rapid cooling, water addition, and sequential acidification with sulfuric acid to separate salts and byproducts before final purification. For detailed operational protocols and safety guidelines, please refer to the standardized synthesis steps outlined below.
- Charge resorcinol and anhydrous potassium carbonate (1: 1 molar ratio) into an autoclave with absolute ethanol (1:3 mass ratio) and dissolve under stirring.
- Pressurize with CO2 gas to 1.35-1.45 MPa and maintain temperature at 130-150°C for 3 to 4 hours to facilitate carboxylation.
- Cool the reaction mixture, adjust pH sequentially to isolate crude product, and purify via recrystallization to obtain 99% pure 2,6-dihydroxybenzoic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this ethanol-based synthesis method offers profound advantages that extend well beyond simple chemical yield improvements, addressing key pain points for supply chain managers and procurement officers. The shift to milder reaction conditions significantly reduces the operational risks associated with high-pressure manufacturing, leading to enhanced supply chain reliability by minimizing unplanned downtime caused by equipment stress or failure. Additionally, the ability to recycle the byproduct 2,4-dihydroxybenzoic acid back into the starting material resorcinol creates a circular economy within the production facility, drastically reducing raw material costs and waste disposal fees. This closed-loop system not only improves the overall economics of the process but also aligns with increasingly stringent environmental regulations regarding phenolic wastewater discharge, ensuring long-term regulatory compliance and operational continuity for the manufacturer.
- Cost Reduction in Manufacturing: The elimination of high-boiling, difficult-to-remove solvents like DMF in favor of ethanol results in substantially lower energy consumption during the solvent recovery phase. Since ethanol forms an azeotrope with water that is easily managed and the solvent itself is cheaper and more readily available than specialized polar aprotic solvents, the overall cost of goods sold is significantly optimized. Furthermore, the shortened reaction time from 10 hours to roughly 4 hours increases the batch turnover rate, allowing the same capital equipment to produce significantly more product over a fiscal year without requiring additional investment in new reactors.
- Enhanced Supply Chain Reliability: By operating at lower pressures (1.45 MPa versus 2.0 MPa) and temperatures, the process places less mechanical demand on the production infrastructure, thereby reducing the frequency of maintenance interventions and extending the lifespan of critical assets. The robustness of the ethanol solvent system also means that the process is less sensitive to minor fluctuations in feedstock quality compared to solvent-free methods, ensuring consistent output quality and dependable delivery schedules for downstream customers who rely on just-in-time inventory models for their own production lines.
- Scalability and Environmental Compliance: The simplified workup procedure, which relies on standard pH adjustments and filtration rather than complex extraction or chromatography, makes this process inherently easier to scale from pilot plant to multi-ton commercial production. The reduction in phenolic wastewater discharge due to the recycling of byproducts and the use of a more benign solvent system greatly simplifies effluent treatment requirements, allowing manufacturers to meet rigorous environmental standards with lower capital expenditure on waste treatment facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their supply chains. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is critical for assessing the feasibility of adopting this method for the commercial scale-up of complex agrochemical intermediates.
Q: What are the primary advantages of using absolute ethanol over DMF in this synthesis?
A: Using absolute ethanol significantly simplifies the separation process compared to dimethylformamide (DMF), reducing energy consumption during solvent recovery and minimizing environmental impact associated with phenolic wastewater discharge.
Q: How does this method improve upon traditional solvent-free carboxylation techniques?
A: This ethanol-based method operates at lower pressures (1.45 MPa vs 2.0 MPa) and shorter reaction times (4 hours vs 10 hours), while simultaneously increasing the molar yield of the target 2,6-isomer from approximately 30% to over 55%.
Q: Can the byproduct 2,4-dihydroxybenzoic acid be utilized?
A: Yes, the process includes a specific recycling step where the byproduct 2,4-dihydroxybenzoic acid is recovered and converted back into resorcinol, thereby enhancing overall raw material utilization efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dihydroxybenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes for key intermediates like 2,6-dihydroxybenzoic acid in the global agrochemical and pharmaceutical markets. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102211995A are fully realized in practical, industrial settings. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced process control systems designed to meet stringent purity specifications, guaranteeing that every batch delivered meets the exacting standards required for the synthesis of high-value herbicides and active pharmaceutical ingredients.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs, whether you require custom synthesis or large-volume supply agreements. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-performance chemical solutions.
