Scalable Organocatalytic Synthesis of High-Purity Chiral Diarylindolemethane Intermediates
Scalable Organocatalytic Synthesis of High-Purity Chiral Diarylindolemethane Intermediates
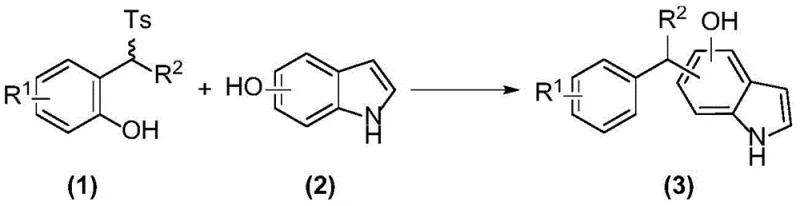
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign methodologies for constructing complex chiral scaffolds essential for next-generation therapeutics. Patent CN110950793B introduces a groundbreaking preparation method for chiral diarylindolemethane compounds, leveraging a sophisticated chiral bifunctional catalyst system within a water-oil two-phase environment. This technology represents a significant leap forward in asymmetric organocatalysis, specifically addressing the critical need for high-purity indole-based building blocks that are ubiquitous in bioactive natural products and small molecule drugs. By utilizing a chiral tertiary amine-nitrogen squaric acid catalyst, the process achieves exceptional stereocontrol under remarkably mild conditions, typically between 20°C and 30°C, thereby minimizing thermal degradation risks often associated with sensitive pharmaceutical intermediates. The strategic implementation of a biphasic reaction system not only enhances the interaction between organic substrates and ionic reagents but also streamlines the workup procedure, offering a compelling value proposition for manufacturers aiming to optimize their production workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing chiral diarylindolemethane frameworks have historically relied heavily on transition metal catalysis or harsh acidic conditions, which present substantial drawbacks for large-scale manufacturing. Metal-catalyzed processes often necessitate the use of expensive noble metals such as palladium or rhodium, introducing significant cost burdens and supply chain vulnerabilities related to metal availability. Furthermore, the presence of residual heavy metals in the final product is a critical regulatory concern for pharmaceutical applications, requiring rigorous and costly purification steps like scavenging or recrystallization to meet stringent ppm limits. Additionally, many conventional methods operate under anhydrous conditions with sensitive reagents, demanding specialized equipment and increasing the risk of operational failures due to moisture sensitivity. These factors collectively contribute to prolonged lead times, elevated production costs, and a larger environmental footprint, making traditional approaches less attractive for the sustainable manufacturing of high-volume active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology disclosed in CN110950793B employs a metal-free organocatalytic strategy that fundamentally reshapes the economic and operational landscape of indole functionalization. The core innovation lies in the use of a chiral bifunctional catalyst containing both a tertiary amine and a nitrogen squaric acid functional group, which acts as a highly efficient hydrogen bond donor to activate the electrophile while the amine moiety activates the nucleophile. This dual activation mechanism enables the Friedel-Crafts diarylmethylation reaction to proceed with high enantioselectivity in a water-oil two-phase system, effectively bypassing the need for toxic metals. The reaction tolerates a broad range of substrates, including various substituted phenols and hydroxyindoles, demonstrating remarkable versatility. By operating at ambient temperatures and utilizing common inorganic bases like potassium carbonate, this novel approach drastically simplifies the reaction setup and reduces energy consumption, positioning it as a superior alternative for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Chiral Bifunctional Organocatalysis
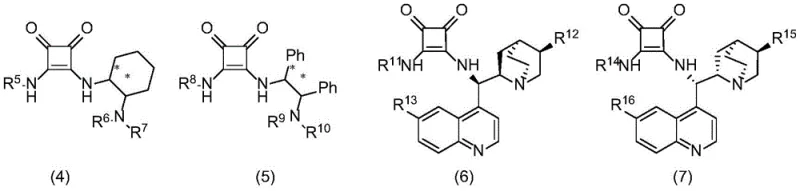
The success of this synthetic protocol is rooted in the precise molecular recognition and activation capabilities of the chiral bifunctional catalyst, specifically those belonging to the squaramide or azoic acid derivative classes depicted in the patent structures. The catalyst functions through a cooperative hydrogen-bonding network where the acidic protons of the squaramide or nitrogen squaric acid moiety form strong hydrogen bonds with the leaving group or the electrophilic center of the 2-(aryl(p-toluenesulfonyl)methyl)phenol substrate. Simultaneously, the basic tertiary amine site interacts with the hydroxyindole nucleophile, likely deprotonating it or organizing it via hydrogen bonding to enhance its nucleophilicity. This spatial arrangement creates a rigid chiral pocket that directs the attack of the indole C3-position onto the benzylic carbon of the phenol derivative with high facial selectivity. The result is the formation of a new carbon-carbon bond with the generation of a chiral center, yielding the diarylindolemethane product with enantiomeric excess (ee) values reaching up to 97% in optimized conditions, as demonstrated by the specific catalyst (7)-h.
Furthermore, the impurity profile of the reaction is tightly controlled by the specificity of the organocatalyst, which minimizes side reactions such as polymerization or non-selective alkylation that are common in acid-catalyzed variants. The water-oil two-phase system plays a crucial mechanistic role by sequestering the inorganic base and the sulfonate leaving group in the aqueous phase, driving the equilibrium forward and preventing the accumulation of acidic byproducts that could racemize the product. This phase separation also protects the catalyst from potential deactivation by ionic species, ensuring sustained catalytic activity throughout the 1 to 48-hour reaction window. The structural diversity of the catalysts, ranging from cyclohexyl-derived amines to quinine-based scaffolds as shown in the patent figures, allows for fine-tuning the steric and electronic properties to match specific substrate pairs, ensuring consistent high performance across a wide substrate scope.
How to Synthesize Chiral Diarylindolemethane Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory optimization and pilot plant scaling. The process begins with the sequential addition of the chiral catalyst, such as the highly effective (7)-h variant, the 2-(aryl(p-toluenesulfonyl)methyl)phenol substrate, the hydroxyindole coupling partner, and an inorganic base like potassium carbonate into a reaction vessel purged with nitrogen. A mixture of an organic solvent, preferably 1,2-dichloroethane (DCE), and water is then introduced to establish the biphasic medium. The reaction proceeds under magnetic stirring at room temperature, monitored by TLC until the starting phenol is fully consumed. Following the reaction, the mixture undergoes a straightforward workup involving liquid-liquid extraction, where the organic phase is separated, dried, and concentrated. The crude product is finally purified using standard silica gel column chromatography with a petroleum ether and ethyl acetate gradient to isolate the target chiral compound in high purity. For detailed standardized synthesis steps and specific parameter adjustments, please refer to the guide below.
- Prepare the reaction mixture by combining 2-(aryl(p-toluenesulfonyl)methyl)phenol, hydroxyindole, inorganic base, and chiral bifunctional catalyst in a dry reaction vessel under nitrogen atmosphere.
- Add the water-oil two-phase solvent system (e.g., DCE and water) and stir the mixture at room temperature (20-30°C) for 1 to 48 hours until the starting material is consumed.
- Perform post-treatment by separating the organic phase, concentrating under reduced pressure, and purifying the crude product via silica gel column chromatography to obtain the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this organocatalytic technology offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of transition metals removes a major cost center associated with catalyst procurement and, more importantly, the downstream purification required to meet regulatory limits for heavy metals in drug substances. This simplification of the purification train translates to significant cost reduction in pharmaceutical intermediate manufacturing by reducing solvent usage, filtration media, and processing time. Moreover, the use of readily available and inexpensive raw materials, such as substituted phenols and hydroxyindoles, ensures a stable supply base that is less susceptible to the geopolitical volatility often seen with precious metal catalysts. The robustness of the reaction conditions, which tolerate ambient temperatures and do not require strictly anhydrous environments, further lowers the barrier for entry for contract manufacturing organizations, enhancing overall supply chain reliability.
- Cost Reduction in Manufacturing: The metal-free nature of this process eliminates the need for expensive metal scavengers and complex waste treatment protocols associated with heavy metal disposal, leading to substantial operational savings. The high atom economy and excellent yields observed across various substrates minimize raw material waste, while the mild reaction conditions reduce energy consumption for heating or cooling, collectively driving down the cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: By relying on stable organic small molecule catalysts rather than air-sensitive metal complexes, the process ensures consistent batch-to-batch quality and reduces the risk of production delays caused by catalyst degradation or supply shortages. The broad substrate scope allows for the flexible sourcing of starting materials, enabling procurement teams to negotiate better terms with multiple suppliers and mitigate the risk of single-source dependency for critical precursors.
- Scalability and Environmental Compliance: The water-oil two-phase system is inherently scalable, as the heat transfer and mixing characteristics are favorable for large reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates. Additionally, the absence of toxic metals and the use of greener reaction conditions align with increasingly stringent environmental regulations, reducing the compliance burden and enhancing the sustainability profile of the manufacturing process, which is a key differentiator for modern supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production pipelines.
Q: What are the key advantages of this organocatalytic method over traditional metal-catalyzed routes?
A: This method utilizes chiral organic small molecules instead of toxic transition metals, eliminating the need for expensive and complex heavy metal removal steps, which significantly simplifies downstream processing and ensures higher purity suitable for pharmaceutical applications.
Q: How does the water-oil two-phase system benefit the reaction efficiency?
A: The biphasic system facilitates the efficient separation of organic substrates and ionic reactants, enhances reaction rates through phase transfer effects, and allows for milder reaction conditions (20-30°C) while maintaining excellent stereoselectivity up to 97% ee.
Q: Is this process scalable for commercial production of API intermediates?
A: Yes, the process uses readily available raw materials, operates under mild conditions without stringent anhydrous requirements, and employs robust organocatalysts, making it highly operable and suitable for commercial scale-up from kilogram to ton levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Diarylindolemethane Supplier
At NINGBO INNO PHARMCHEM, we recognize the pivotal role that high-quality chiral intermediates play in the development of life-saving medications. Our team of expert chemists has extensively evaluated the technology described in CN110950793B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative organocatalytic process to fruition. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to ensure that every batch of chiral diarylindolemethane complies with the highest industry standards for enantiomeric excess and chemical purity. Our state-of-the-art facilities are equipped to handle the specific requirements of biphasic reactions and organocatalysis, ensuring a seamless transition from laboratory discovery to industrial manufacturing.
We invite pharmaceutical companies and research institutions to collaborate with us to unlock the full potential of this efficient synthetic route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs, demonstrating how this metal-free approach can optimize your budget. We encourage you to contact us today to obtain specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets, ensuring a reliable and cost-effective supply of these critical building blocks for your drug development programs.
