Advanced Manufacturing of Benzodiazepine Compounds via Novel Intermediate K
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic structures, particularly those acting as modulators for neurotransmitter receptors. Patent CN111620834A introduces a significant breakthrough in the preparation of a specific benzodiazepine compound, identified as a partial agonist allosteric modulator of the GABAA receptor. This compound exhibits selective affinity for the α1 subtype, offering therapeutic potential with a reduced side effect profile compared to traditional benzodiazepines. The core innovation lies in the discovery and utilization of a novel intermediate, designated as Compound K, which streamlines the final construction of the molecular architecture. By leveraging this key intermediate, manufacturers can achieve high yields while minimizing the operational complexity typically associated with building fused ring systems. This technical advancement represents a pivotal shift from labor-intensive legacy methods to a more efficient, scalable protocol suitable for modern fine chemical production standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes, such as the one disclosed in CN1350538A, present substantial challenges for industrial scale-up due to their inherent inefficiencies and rigorous operational demands. The conventional pathway typically involves converting a precursor Compound D into Compound G through a sequence that suffers from disappointingly low reaction yields. Furthermore, critical steps in this legacy process require maintenance of extremely low temperatures, specifically around -35°C, which imposes severe constraints on reactor equipment and energy consumption. Such cryogenic conditions not only increase the capital expenditure for specialized cooling infrastructure but also introduce significant safety risks and operational bottlenecks in a plant setting. Additionally, the overall synthetic route is protracted, necessitating up to five distinct reaction steps just to reach the intermediate stage, each requiring separate isolation and purification procedures that erode overall mass efficiency.
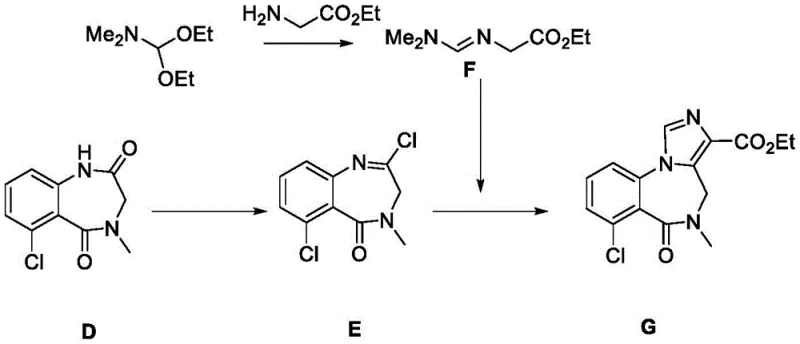
The Novel Approach
In stark contrast, the methodology outlined in CN111620834A circumvents these historical bottlenecks by introducing a convergent strategy centered on Intermediate K. This new approach enables the formation of the target Formula I compound in a single, high-yielding step from Compound K, drastically reducing the number of unit operations required. The reaction conditions are markedly milder, operating effectively within a temperature range of -10°C to 25°C, which eliminates the need for extreme cryogenic cooling and allows for the use of standard industrial reactors. The simplicity extends to the work-up phase, where standard techniques like concentration, extraction, and solvent pulping are sufficient to achieve high purity without resorting to complex chromatographic separations. This transition from a linear, low-temperature process to a convergent, ambient-temperature protocol fundamentally alters the economic and technical feasibility of manufacturing this valuable pharmacological scaffold.
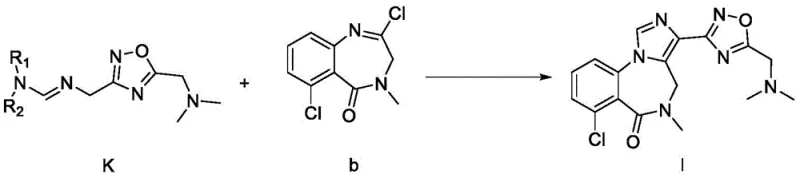
Mechanistic Insights into the Formation of Intermediate K and Final Cyclization
The chemical elegance of this patent lies in the construction of the 1,2,4-oxadiazole ring system and its subsequent integration into the benzodiazepine core. The synthesis of Intermediate K begins with the condensation of an aminomethyl-oxadiazole derivative (Compound F) with an appropriate acetal (Compound L). This aldehyde-amine condensation reaction proceeds smoothly under mild conditions to form the imine linkage characteristic of Compound K. The versatility of this step is highlighted by the tolerance for various R groups, allowing for the tuning of steric and electronic properties without compromising reaction efficiency. Following the formation of K, the final cyclization involves the reaction with a chloro-substituted benzodiazepine precursor (Compound b) in the presence of a strong non-nucleophilic base. This step facilitates the nucleophilic attack and subsequent ring closure that establishes the final fused ring system, locking the molecule into its bioactive conformation with high stereochemical integrity.
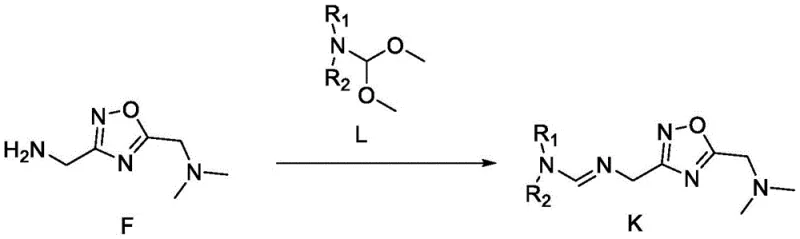
Impurity control is inherently managed through the robustness of the reaction mechanism and the simplicity of the purification strategy. The use of strong bases like lithium bis(trimethylsilyl)amide ensures complete deprotonation and minimizes side reactions that could lead to oligomerization or incomplete conversion. Moreover, the final product precipitation via pulping with cold ethanol serves as an effective crystallization-induced purity enhancement step. This physical purification method leverages solubility differences to exclude structurally related impurities and residual starting materials, ensuring that the final API intermediate meets stringent quality specifications. The mechanistic pathway avoids the generation of difficult-to-remove byproducts common in older methods, thereby simplifying the analytical burden on quality control laboratories and ensuring batch-to-batch consistency essential for regulatory compliance in pharmaceutical manufacturing.
How to Synthesize Benzodiazepine Compound Efficiently
The standardized synthesis protocol derived from this patent offers a clear roadmap for process chemists aiming to implement this technology. The procedure begins with the preparation of the aminomethyl-oxadiazole precursor, followed by the critical condensation step to generate Intermediate K. Once K is isolated, it is coupled with the chloro-benzodiazepine fragment under controlled basic conditions to yield the final product. The detailed standardized synthesis steps see the guide below, which outlines the specific reagents, stoichiometry, and thermal profiles required to replicate the high yields reported in the intellectual property documentation. Adhering to these parameters ensures optimal conversion rates and minimizes the formation of degradation products.
- Protect aminoacetonitrile hydrochloride to form compound B, then react with hydroxylamine to generate amidoxime compound C.
- Perform cyclization with chloroacetyl chloride or dimethylglycine to form the oxadiazole ring intermediate E, followed by deprotection to yield compound F.
- Condense compound F with an acetal to form key intermediate K, then couple with compound b under basic conditions to finalize the benzodiazepine structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route translates directly into tangible operational improvements and risk mitigation. The elimination of extreme low-temperature requirements significantly reduces the energy load on manufacturing facilities, leading to lower utility costs and a smaller carbon footprint per kilogram of product. Furthermore, the reduction in the number of synthetic steps decreases the total volume of solvents and reagents consumed, which simplifies waste management protocols and lowers the cost of raw material procurement. The robustness of the chemistry ensures higher throughput and reliability, allowing suppliers to maintain consistent inventory levels even during periods of high market demand. These factors collectively contribute to a more resilient supply chain capable of supporting long-term commercial agreements without the volatility associated with complex, low-yield manufacturing processes.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive cryogenic equipment and reduces the consumption of energy-intensive cooling resources, resulting in substantial operational cost savings. By consolidating multiple reaction steps into fewer, higher-yielding transformations, the overall material cost per unit is significantly decreased, enhancing the margin potential for downstream drug developers. The simplified purification process also reduces the reliance on costly chromatographic resins and large volumes of high-grade solvents, further driving down the cost of goods sold.
- Enhanced Supply Chain Reliability: The use of commercially available starting materials and standard reaction conditions mitigates the risk of supply disruptions caused by specialized reagent shortages. The high yield and reproducibility of the synthesis ensure that production schedules can be met consistently, reducing the likelihood of delays in API delivery. This reliability is crucial for pharmaceutical companies managing tight development timelines and regulatory submission deadlines, as it guarantees a steady flow of high-quality intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing common organic solvents and straightforward work-up procedures that are easily adapted to large-scale reactors. The reduction in hazardous waste generation and energy consumption aligns with modern green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific technical disclosures and beneficial effects outlined in the patent documentation, providing clarity on yield expectations, reaction conditions, and scalability. Understanding these details helps stakeholders assess the feasibility of integrating this route into their existing supply chains and development pipelines.
Q: What is the primary advantage of using Intermediate K in this synthesis?
A: Intermediate K allows for the preparation of the target benzodiazepine compound in a single step with high yield (93-98%), significantly simplifying the post-treatment and purification processes compared to conventional multi-step routes.
Q: How does the new method improve upon the reaction conditions described in CN1350538A?
A: Unlike the prior art which requires strict low-temperature conditions around -35°C and suffers from low yields, the new method operates at milder temperatures ranging from -10°C to 25°C, reducing energy consumption and equipment stress.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process is designed for scalability with simple work-up operations such as concentration, extraction, and pulping, making it environment-friendly and beneficial for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzodiazepine Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial plant is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and quality of every batch produced. Our commitment to technical excellence ensures that the benzodiazepine intermediates we supply meet the exacting standards required for global pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall project costs.
