Scalable Synthesis of Resveratrol Dimer Bisphosphate Derivatives for Osteoporosis Therapy
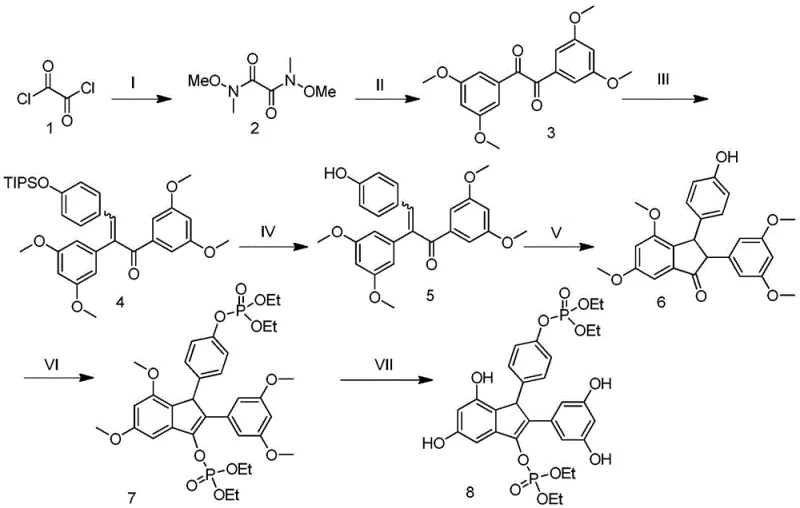
The pharmaceutical industry is constantly seeking robust synthetic routes for novel therapeutic agents, particularly in the realm of bone health. Patent CN113336788B, published in late 2022, introduces a significant advancement in the preparation of natural product resveratrol dimer bisphosphate derivatives. These compounds have demonstrated potent efficacy in promoting osteoblast differentiation and inhibiting osteoclast activity, positioning them as promising candidates for osteoporosis treatment. The disclosed methodology outlines a comprehensive seven-step synthetic pathway starting from readily available oxalyl chloride. This technical breakthrough addresses historical challenges associated with the synthesis of complex stilbene dimers, specifically targeting issues related to low yields, excessive consumable usage, and harsh reaction conditions. By optimizing catalyst selection and reaction parameters, this patent provides a viable framework for the commercial production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of resveratrol derivatives and their phosphorylated analogues has been plagued by inefficiencies that hinder large-scale manufacturing. Traditional approaches often suffer from poor regioselectivity during the construction of the carbon skeleton, leading to complex mixtures of isomers that are difficult and costly to separate. Furthermore, many existing protocols rely on multi-step sequences with cumulative yield losses that render the final product economically unviable for widespread clinical application. The use of unstable intermediates and sensitive protecting groups in older methods frequently necessitates cryogenic conditions and inert atmospheres throughout the entire process, drastically increasing energy consumption and operational complexity. Additionally, the removal of heavy metal catalysts or toxic reagents from the final API intermediate often requires extensive purification workflows, adding further time and cost to the supply chain. These factors collectively create a bottleneck for procurement teams looking to secure reliable sources of bone health therapeutics.
The Novel Approach
The methodology described in patent CN113336788B offers a streamlined alternative that mitigates these traditional pain points through strategic reaction design. By employing a Weinreb amide strategy in the early stages, the synthesis ensures precise control over carbonyl addition, preventing over-alkylation and ensuring high fidelity in the formation of the diketone precursor. The integration of a Wittig olefination followed by a Lewis acid-mediated Nazarov cyclization allows for the rapid assembly of the congested indanone core under relatively mild conditions. Crucially, the process utilizes common industrial solvents such as dichloromethane and tetrahydrofuran, alongside standard reagents like triethylamine and sodium hydride, which are easily sourced globally. The final demethylation step using boron tribromide is executed with high efficiency, ensuring the exposure of the necessary phenolic hydroxyl groups without degrading the phosphate ester moieties. This holistic optimization results in a process that is not only chemically elegant but also commercially pragmatic for fine chemical manufacturers.
Mechanistic Insights into Nazarov Cyclization and Phosphorylation
A pivotal moment in this synthetic route occurs during Step V, where the linear enone precursor undergoes a 4π-electrocyclic ring closure known as the Nazarov cyclization. In this specific embodiment, the reaction is catalyzed by boron trifluoride diethyl etherate (BF3·OEt2) in dichloromethane at room temperature. The Lewis acid coordinates with the carbonyl oxygen, increasing the electrophilicity of the system and facilitating the formation of a pentadienyl cation intermediate. This cationic species then undergoes conrotatory ring closure to form the cyclopentenyl cation, which is subsequently trapped or eliminated to yield the stable indanone scaffold found in Compound 6. The choice of BF3·OEt2 is particularly advantageous as it avoids the use of harsher protic acids that might compromise the acid-sensitive silyl or ether protecting groups present elsewhere in the molecule. This mechanistic precision ensures that the stereochemical integrity of the molecule is maintained while constructing the rigid core required for biological activity against osteoporosis targets.
Following the core construction, the introduction of the phosphate moiety in Step VI and the subsequent global deprotection in Step VII are critical for the compound's solubility and bioavailability. The phosphorylation is achieved via nucleophilic substitution using sodium hydride to deprotonate the phenolic hydroxyl group, followed by reaction with diethyl chlorophosphate. This generates the phosphotriester intermediate (Compound 7) with high selectivity. The final step involves the cleavage of methyl ethers using BBr3, a powerful demethylating agent. The mechanism here involves the coordination of boron to the ether oxygen, followed by nucleophilic attack by bromide ion to release methyl bromide and the free phenol. The patent specifies careful temperature control during this exothermic process to prevent degradation of the phosphate ester, demonstrating a nuanced understanding of functional group compatibility. This sequence ensures that the final product possesses the necessary polar groups for interaction with bone tissue while maintaining metabolic stability.
How to Synthesize Resveratrol Dimer Bisphosphate Derivative Efficiently
The synthesis of this complex intermediate requires strict adherence to the optimized conditions outlined in the patent to ensure reproducibility and safety. The process begins with the formation of the Weinreb amide, followed by lithiation and coupling, setting the stage for the carbon framework. Subsequent steps involve olefination, deprotection, cyclization, phosphorylation, and final deprotection. Each step has been refined to maximize yield and minimize impurity formation, making it suitable for transfer to pilot and production scales. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Condensation of oxalyl chloride with N,O-dimethylhydroxylamine to form the Weinreb amide intermediate.
- Lithiation of 1-bromo-3,5-dimethoxybenzene followed by addition to the Weinreb amide to generate the diketone.
- Wittig olefination followed by deprotection, Nazarov cyclization, phosphorylation, and final demethylation.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers substantial benefits regarding raw material security and cost structure. The starting materials, including oxalyl chloride and 1-bromo-3,5-dimethoxybenzene, are commodity chemicals produced in vast quantities by the global chemical industry, ensuring a stable and continuous supply without reliance on niche vendors. The avoidance of precious metal catalysts such as palladium or platinum eliminates the volatility associated with mining-dependent pricing and removes the need for expensive metal scavenging steps during purification. This simplification of the downstream processing directly translates to reduced manufacturing lead times and lower operational expenditures. Furthermore, the use of standard solvents and reagents means that existing multipurpose reactors in CDMO facilities can be utilized without requiring specialized equipment modifications, enhancing the overall agility of the production schedule.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of cost-effective Lewis acids like BF3·OEt2 significantly lowers the bill of materials. Additionally, the streamlined purification processes, primarily relying on crystallization and standard chromatography, reduce solvent consumption and waste disposal costs. The high selectivity of the reactions minimizes the formation of difficult-to-remove impurities, thereby increasing the overall throughput of the manufacturing line. By optimizing the molar ratios of reagents, particularly in the lithiation and phosphorylation steps, the process reduces excess reagent waste, contributing to a leaner and more economical production model.
- Enhanced Supply Chain Reliability: Because the synthesis relies on widely available organic building blocks rather than proprietary or scarce reagents, the risk of supply disruption is markedly decreased. The robustness of the reaction conditions, which tolerate ambient temperatures in several key steps, reduces the energy burden on manufacturing facilities and allows for more flexible scheduling. This reliability is crucial for long-term contracts with pharmaceutical partners who require guaranteed delivery of critical intermediates for clinical trials and commercial launches. The scalability of the process from gram to kilogram scale has been demonstrated, ensuring that supply can be ramped up quickly to meet market demand without compromising quality.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing the use of hazardous reagents and optimizing atom economy in the cyclization steps. The waste streams generated are primarily composed of standard organic solvents and inorganic salts, which can be treated using conventional wastewater management systems. The absence of heavy metals simplifies the environmental compliance profile, making it easier to obtain necessary regulatory approvals for manufacturing sites. This environmental friendliness aligns with the increasing corporate sustainability goals of major pharmaceutical buyers, adding value beyond mere cost and availability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this resveratrol derivative. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is essential for R&D and procurement teams evaluating this technology for integration into their pipelines.
Q: What is the key cyclization strategy used in this synthesis?
A: The synthesis utilizes a Lewis acid-catalyzed Nazarov cyclization (Step V) using BF3·OEt2 to construct the critical indanone core structure efficiently.
Q: Are expensive transition metal catalysts required for this process?
A: No, the process relies on standard organolithium reagents and Lewis acids rather than expensive precious metal catalysts, significantly reducing raw material costs.
Q: What is the overall yield of the target compound?
A: The patent reports a total yield of 3.48% over the seven-step sequence, which represents an optimization over previous low-yielding methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Resveratrol Dimer Bisphosphate Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing the technical expertise to translate complex patent methodologies like CN113336788B into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Resveratrol Dimer Bisphosphate Derivative meets the highest international standards. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, facilitating smoother regulatory filings for your downstream drug products.
We invite you to collaborate with us to leverage this innovative synthetic route for your osteoporosis drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your project timelines and optimize your budget.
