Advanced Synthesis of 4-Nitro-3-Hydroxy-2-Picolinic Acid for Commercial Scale-Up
The pharmaceutical and agrochemical industries continuously demand high-purity heterocyclic intermediates to ensure the efficacy and safety of final active ingredients. A critical breakthrough in this domain is detailed in patent CN103880740A, which discloses a highly selective synthesis method for 4-nitro-3-hydroxy-2-picolinic acid. This compound serves as a vital building block for various functional materials and medicinal agents, yet its production has historically been plagued by regioselectivity issues. The patented technology utilizes a strategic three-step sequence involving sulfonation, nitration, and desulfonation to overcome these barriers. By employing 3-hydroxy-2-picolinic acid as the starting material, the process achieves exceptional control over the substitution pattern on the pyridine ring. This technical advancement is not merely a laboratory curiosity but represents a robust pathway for industrial manufacturing, addressing the persistent challenges of isomer separation and yield optimization that have long hindered the reliable supply of this key intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrated picolinic acid derivatives has relied on direct nitration strategies, which suffer from severe inherent limitations regarding regioselectivity. As illustrated in the reaction scheme below, attempting to nitrate 3-hydroxy-2-picolinic acid directly using standard nitrating agents like concentrated nitric acid in methylene chloride results in a chaotic mixture of products. The hydroxyl group acts as an ortho-para directing group, leading to competition between the 4-position and the 6-position on the pyridine ring. Experimental data indicates that this direct approach yields a roughly 6:4 ratio of the desired 4-nitro isomer to the unwanted 6-nitro isomer. Since these two isomers possess very similar polarities and physical properties, separating them via standard crystallization or chromatography is extremely difficult and economically prohibitive on a large scale. This lack of selectivity drastically reduces the effective yield of the desired product and generates significant chemical waste, making conventional direct nitration unsuitable for cost-effective commercial manufacturing.
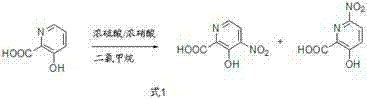
The Novel Approach
To circumvent the selectivity issues of direct nitration, the patented methodology introduces a sophisticated "blocking group" strategy that fundamentally alters the reaction landscape. Instead of attacking the substrate directly with nitric acid, the process first employs a sulfonation reaction to install a sulfonic acid group specifically at the 6-position of the pyridine ring. This bulky sulfonic acid group effectively blocks the 6-position, preventing the nitronium ion from attacking that site during the subsequent nitration step. Consequently, the nitration is forced to occur exclusively at the 4-position, ensuring high regioselectivity. Following nitration, the temporary sulfonic acid blocking group is removed via a desulfonation reaction under acidic hydrolysis conditions. This three-step sequence—sulfonation, nitration, and desulfonation—allows for the highly selective production of 4-nitro-3-hydroxy-2-picolinic acid with minimal isomer contamination, transforming a previously low-yielding process into a viable industrial operation.
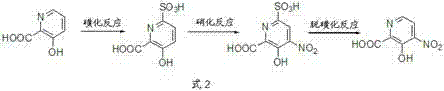
Mechanistic Insights into Regioselective Sulfonation and Nitration
The success of this synthesis hinges on the precise control of thermodynamic and kinetic factors during the initial sulfonation step. When 3-hydroxy-2-picolinic acid reacts with sulfuric acid, the sulfonation can theoretically occur at either the 4-position or the 6-position. Initially, the reaction may produce a mixture containing the kinetic product, which is the 4-sulfonic acid derivative. However, the patent details a critical temperature programming protocol where the reaction mixture is heated from 90-130°C up to 140°C. This elevated temperature facilitates the conversion of the kinetically formed 4-sulfonic acid isomer into the thermodynamically more stable 6-sulfonic acid isomer. By ensuring that the sulfonic acid group resides almost exclusively at the 6-position before nitration begins, the process guarantees that the subsequent nitration step will be directed solely to the 4-position. This mechanistic understanding allows for the suppression of the 6-nitro impurity to levels below 0.1%, a purity profile that is unattainable through direct methods.
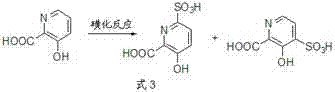
Furthermore, the catalytic system employed in this process plays a pivotal role in driving the reaction efficiency and selectivity. The sulfonation step utilizes cuprous catalysts, such as cuprous chloride or cuprous bromide, which enhance the rate of sulfonation and assist in the isomerization to the thermodynamic product. In the nitration step, the addition of specific nitrate catalysts, such as cerous nitrate or zirconium nitrate, works in synergy with the residual cuprous species to promote efficient nitration under milder conditions. This dual-catalyst approach minimizes side reactions and decomposition, which are common risks when dealing with electron-deficient pyridine rings under strong acidic conditions. The careful orchestration of these catalytic cycles ensures that the intermediate 6-sulfonic-4-nitro derivative is formed in high yield before the final desulfonation step releases the target molecule. This deep mechanistic control is what enables the process to achieve total yields ranging from 60% to 87%, providing a robust foundation for scalable production.
How to Synthesize 4-Nitro-3-Hydroxy-2-Picolinic Acid Efficiently
The implementation of this synthesis route requires strict adherence to the temperature profiles and catalyst loadings described in the patent to maximize yield and purity. The process is designed to be operationally simple, with the first two steps potentially being performed in a telescoped manner without isolating the intermediate, which significantly reduces processing time and solvent usage. For R&D teams looking to replicate or scale this chemistry, the key lies in monitoring the conversion of the sulfonic acid isomers via liquid chromatography to ensure the thermodynamic product is fully formed before proceeding to nitration. The detailed standardized synthesis steps, including specific reagent quantities and safety precautions for handling concentrated acids at elevated temperatures, are outlined in the guide below.
- Perform sulfonation of 3-hydroxy-2-picolinic acid using sulfuric acid and a cuprous catalyst at 90-140°C to form the 6-sulfonic acid derivative.
- Conduct nitration on the sulfonated intermediate using concentrated nitric acid and a nitrate catalyst at 40-55°C.
- Execute desulfonation by adding water and heating to 130-160°C, followed by crystallization to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic advantages over traditional sourcing methods. The primary benefit lies in the drastic simplification of the purification process. By eliminating the formation of the difficult-to-separate 6-nitro isomer, manufacturers avoid the need for complex and costly chromatographic separations or multiple recrystallization cycles that typically erode profit margins. This streamlined downstream processing translates directly into a more predictable and stable cost structure for the final intermediate. Furthermore, the high overall yield of the process means that less raw material is required to produce the same amount of finished goods, optimizing the utilization of starting materials like 3-hydroxy-2-picolinic acid and reducing the overall cost of goods sold (COGS) for the supply chain.
- Cost Reduction in Manufacturing: The elimination of isomer separation steps removes a significant bottleneck in production, leading to lower operational expenditures and reduced solvent consumption. The ability to telescope the sulfonation and nitration steps without intermediate isolation further decreases labor and equipment usage time. Additionally, the use of relatively inexpensive and commercially available catalysts like cuprous chloride and cerous nitrate ensures that the catalytic costs remain low compared to processes requiring precious metals. These factors combine to create a highly cost-efficient manufacturing protocol that enhances the competitiveness of the final product in the global market.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream pharmaceutical customers. Because the process relies on widely available commodity chemicals such as sulfuric acid and nitric acid, it is less susceptible to supply disruptions associated with exotic or specialized reagents. The high selectivity of the reaction minimizes the generation of hazardous waste streams associated with failed batches or off-spec material, simplifying environmental compliance and reducing the risk of production stoppages due to regulatory issues. This reliability makes the supplier a more dependable partner for long-term contracts.
- Scalability and Environmental Compliance: The reaction conditions, while requiring elevated temperatures, utilize standard glass-lined or stainless steel reactors commonly found in fine chemical plants, facilitating easy scale-up from pilot to commercial tonnage. The process avoids the use of chlorinated solvents in the main reaction steps, relying instead on aqueous acid media, which simplifies waste treatment and aligns with modern green chemistry principles. The high atom economy achieved through the blocking group strategy ensures that the majority of the starting material ends up in the final product rather than in waste byproducts. This environmental efficiency not only reduces disposal costs but also future-proofs the supply chain against increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-nitro-3-hydroxy-2-picolinic acid. These answers are derived directly from the technical specifications and experimental data provided in the underlying patent documentation. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific drug development or agrochemical formulation pipelines. We encourage technical teams to review these points to gain a clearer picture of the material's quality attributes and production capabilities.
Q: Why is direct nitration of 3-hydroxy-2-picolinic acid inefficient?
A: Direct nitration results in a difficult-to-separate mixture of 4-nitro and 6-nitro isomers with a ratio of approximately 6:4, significantly lowering yield and increasing purification costs.
Q: How does the sulfonation blocking strategy improve selectivity?
A: By introducing a sulfonic acid group at the 6-position first, the reactive site is blocked, forcing the subsequent nitration to occur exclusively at the 4-position, thereby eliminating isomer formation.
Q: What represents the key quality advantage of this patented process?
A: The process achieves a total yield of 60-87% with a final product purity exceeding 95%, and critically, reduces the content of the 6-nitro isomer impurity to below 0.1%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Nitro-3-Hydroxy-2-Picolinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a reliable commercial supply requires deep technical expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high selectivity and purity demonstrated in the patent are maintained at an industrial level. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch of 4-nitro-3-hydroxy-2-picolinic acid meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to technical excellence ensures that you receive a material that is not only chemically pure but also consistent in its physical properties, facilitating smooth downstream processing in your own facilities.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that demonstrates how switching to our optimized supply chain can reduce your overall manufacturing expenses. Please contact us today to request specific COA data for our current inventory or to discuss route feasibility assessments for your custom synthesis requirements. By partnering with us, you gain access to a secure, high-quality supply of this critical intermediate, backed by our proven track record in fine chemical manufacturing.
