Revolutionizing Para-Chloroaniline Production: A Green Catalytic Approach for Global Supply Chains
Revolutionizing Para-Chloroaniline Production: A Green Catalytic Approach for Global Supply Chains
The chemical industry is constantly seeking more efficient and environmentally benign pathways for synthesizing critical building blocks, particularly substituted anilines which serve as foundational scaffolds in medicinal chemistry and crop protection. Patent CN102993023A introduces a transformative methodology for the preparation of mono-substituted para-chloroanilines, addressing long-standing challenges in regioselectivity and process safety. This innovation leverages a copper-catalyzed oxidative chlorination system that utilizes molecular oxygen and hydrogen chloride gas in the presence of copper chloride, effectively bypassing the cumbersome protection-deprotection sequences traditionally required. By integrating this advanced catalytic cycle, manufacturers can achieve superior para-selectivity while minimizing hazardous waste generation, marking a significant leap forward in sustainable fine chemical synthesis.
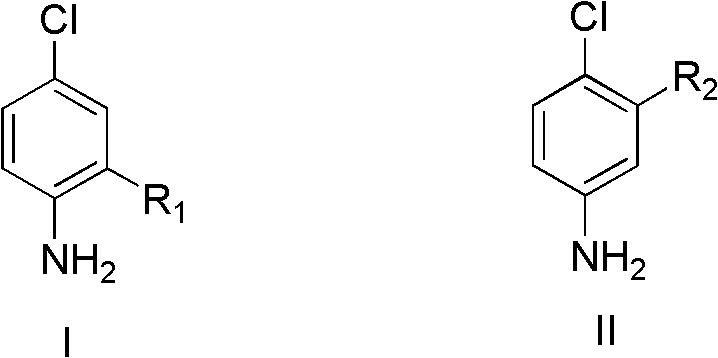
For R&D directors and process chemists, the implications of this technology are profound, offering a robust route to high-purity intermediates such as 4-chloro-2-methylaniline and 4-chloro-2-fluoroaniline. The ability to directly functionalize the aromatic ring at the para-position relative to the amino group without prior derivatization streamlines the synthetic timeline considerably. Furthermore, the versatility of the substrate scope, accommodating various electron-donating and electron-withdrawing groups, ensures broad applicability across diverse therapeutic and agrochemical portfolios. As we delve deeper into the technical specifics, it becomes evident that this patent represents not just a laboratory curiosity, but a scalable industrial solution capable of meeting the rigorous demands of modern supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of substituted para-chloroanilines has been plagued by inefficiencies and safety concerns inherent to traditional electrophilic aromatic substitution strategies. Conventional routes typically necessitate a multi-step sequence involving the initial protection of the sensitive amino group, often as an acetamide or similar derivative, to prevent over-chlorination and oxidation side reactions. Following protection, harsh chlorinating agents such as sulfuryl chloride or molecular chlorine are employed under vigorous conditions, which frequently lead to mixtures of ortho- and para-isomers that are difficult and costly to separate. The final deprotection step adds further complexity, requiring additional reagents, energy input, and generating stoichiometric amounts of acidic or basic waste streams. This linear, resource-intensive approach results in low overall atom economy and poses significant environmental compliance challenges for large-scale facilities.
The Novel Approach
In stark contrast, the methodology disclosed in CN102993023A offers a direct, one-pot chlorination strategy that elegantly circumvents the need for amino protection. By utilizing copper(II) chloride as a dual-function reagent acting as both the oxidant and the chlorine source, the process achieves remarkable para-selectivity through a distinct mechanistic pathway. The reaction proceeds under mild thermal conditions, typically between 30°C and 70°C, in a variety of solvents ranging from aqueous hydrochloric acid to organic media like tetrahydrofuran or even ionic liquids. This flexibility allows process engineers to optimize solubility and reaction kinetics without compromising safety. Moreover, the continuous introduction of oxygen facilitates the in-situ regeneration of the active copper species, creating a semi-catalytic cycle that drastically reduces copper consumption compared to stoichiometric methods.
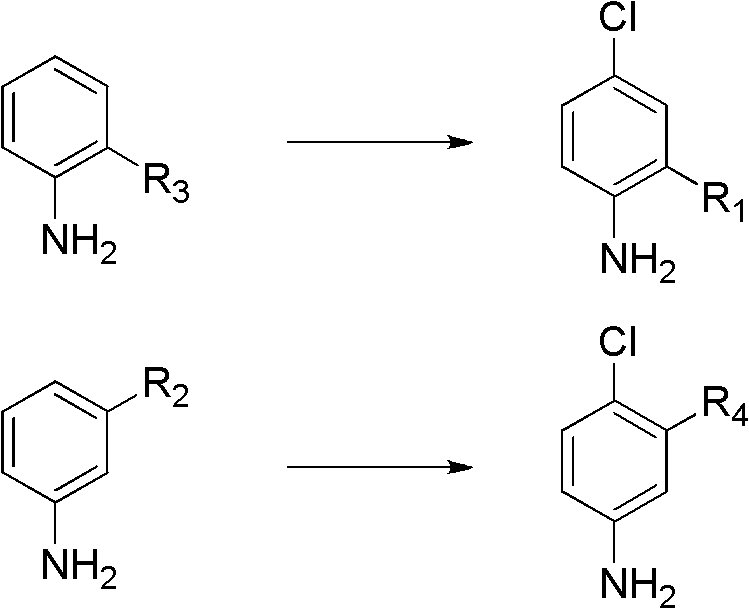
The scope of this novel approach is exceptionally broad, successfully converting a wide array of ortho- and meta-substituted anilines into their corresponding para-chloro derivatives with high fidelity. As illustrated in the patent examples, substrates bearing methyl, methoxy, fluoro, trifluoromethyl, and even nitro groups are tolerated, yielding products like 4-chloro-2-trifluoromethylaniline and 4-chloro-3-methoxyaniline in excellent yields. This universality makes the process highly attractive for the production of diverse pharmaceutical intermediates and agrochemical intermediates, where structural diversity is key. The elimination of protection groups not only shortens the synthetic route but also significantly reduces the E-factor of the process, aligning perfectly with green chemistry principles and modern regulatory expectations for sustainable manufacturing.
Mechanistic Insights into Copper-Catalyzed Oxidative Chlorination
Understanding the underlying mechanism is crucial for R&D teams aiming to implement this technology at scale, as it explains the observed high regioselectivity and efficiency. The inventors propose that the reaction initiates with a single-electron transfer (SET) from the electron-rich benzene ring of the aniline substrate to the copper(II) ion. This oxidation event generates a transient radical cation intermediate and reduces the copper to the +1 oxidation state. Computational studies cited in the patent indicate that the resulting radical cation adopts a specific conformation where the para-position is electronically activated and sterically accessible for nucleophilic attack. Subsequently, a chloride ion, abundant in the reaction medium from the copper salt or added HCl, rapidly quenches this high-energy intermediate to form the C-Cl bond. This pathway inherently favors para-substitution due to the stability of the transition state leading to the para-isomer compared to the ortho-isomer.
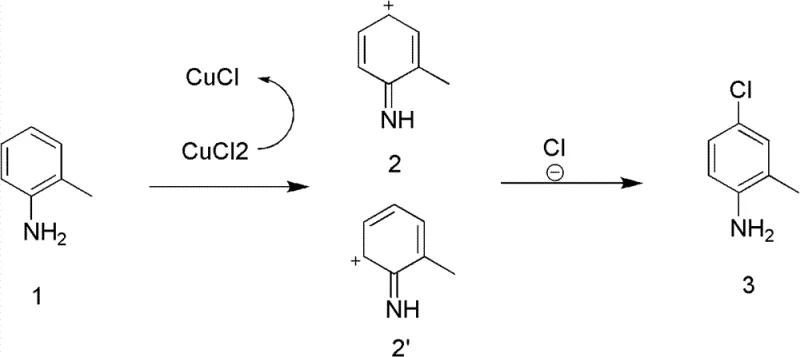
A critical feature of this mechanism is the regeneration loop that sustains the catalytic activity. As the Cu(II) is reduced to Cu(I) during the oxidation of the substrate, the continuous sparging of oxygen gas serves to re-oxidize the Cu(I) species back to the active Cu(II) state. This redox cycling ensures that the copper chloride functions effectively as a catalyst rather than a stoichiometric reagent, although an initial excess may be used to drive kinetics. Additionally, the co-feed of hydrogen chloride gas plays a dual role: it maintains the acidity required to keep the copper species soluble and prevents the formation of insoluble copper oxides or hydroxides, while also suppressing potential polymerization of the reactive aniline intermediates. This intricate balance of oxidation potential and acid-base chemistry allows for the clean conversion of sensitive substrates without degradation, ensuring the production of high-purity OLED material precursors or drug intermediates with minimal impurity profiles.
How to Synthesize 4-Chloro-2-methylaniline Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and selectivity, particularly regarding gas flow rates and temperature control. The patent outlines a robust protocol where the substrate is dissolved in a solvent such as concentrated hydrochloric acid or THF, followed by the addition of copper chloride. The mixture is then heated, and a steady stream of oxygen and HCl gas is introduced to maintain the oxidative environment. Reaction progress is typically monitored via TLC or GC-MS until the starting material is fully consumed, usually within 1 to 8 hours depending on the substrate electronics. Workup involves neutralization and extraction, leveraging the fact that the copper salts remain in the aqueous phase or can be filtered off if ionic liquids are used, facilitating easy product isolation. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Dissolve the substituted aniline substrate (e.g., 2-methylaniline) in a suitable solvent such as hydrochloric acid, THF, or ionic liquids.
- Add copper chloride (CuCl2) to the reaction mixture and heat to a temperature between 30°C and 70°C while introducing oxygen and hydrogen chloride gas.
- Monitor the reaction until the starting material is consumed, then extract the product with organic solvents like ethyl acetate and purify via crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this copper-catalyzed chlorination technology offers substantial strategic benefits that extend beyond mere technical feasibility. The most immediate impact is seen in the simplification of the supply chain for raw materials; by eliminating the need for protecting group reagents and harsh chlorinating agents like thionyl chloride, purchasing managers can source fewer, more commoditized chemicals. This consolidation of the bill of materials reduces administrative overhead and mitigates the risk of supply disruptions associated with specialized reagents. Furthermore, the ability to recycle the copper catalyst through oxygen regeneration means that the effective consumption of copper salts is drastically lowered, leading to significant cost reduction in pharmaceutical intermediates manufacturing. The process transforms what was once a waste liability into a reusable asset, enhancing the overall economic viability of the production line.
- Cost Reduction in Manufacturing: The elimination of multi-step protection and deprotection sequences fundamentally alters the cost structure of producing para-chloroanilines. By condensing the synthesis into a single pot, facilities save on labor, energy, and solvent usage, which are major cost drivers in batch processing. The removal of expensive protecting groups and the associated reagents for their installation and removal directly lowers the variable cost per kilogram. Additionally, the high regioselectivity minimizes the formation of ortho-isomers, reducing the need for costly and yield-loss-inducing purification steps like preparative chromatography or repeated recrystallizations. This streamlined workflow translates to a leaner manufacturing process with a smaller physical footprint and lower utility consumption.
- Enhanced Supply Chain Reliability: Relying on abundant and stable reagents like copper chloride and oxygen gas enhances the resilience of the supply chain against market volatility. Unlike molecular chlorine, which requires specialized handling and transport infrastructure due to its toxicity and corrosivity, the reagents used in this process are safer and easier to manage logistically. The robustness of the reaction conditions, which tolerate a range of solvents including water and ionic liquids, provides flexibility in sourcing. If a specific organic solvent faces a shortage, the process can potentially be adapted to use alternative media without compromising the core chemistry. This adaptability ensures consistent production schedules and reliable delivery of commercial scale-up of complex polymer additives or drug precursors to downstream customers.
- Scalability and Environmental Compliance: The environmental profile of this method is a key enabler for long-term scalability in regions with strict emission regulations. The process avoids the generation of large volumes of acidic wastewater associated with traditional chlorination and deprotection steps. The regeneration of the copper catalyst reduces the load of heavy metals in the effluent, simplifying waste treatment protocols. Moreover, the use of oxygen as the terminal oxidant produces water as the only byproduct of the redox cycle, aligning with green chemistry metrics. This cleaner process reduces the burden on environmental health and safety (EHS) departments, lowering compliance costs and facilitating faster regulatory approvals for new facilities. It positions the manufacturer as a sustainable partner capable of meeting the rigorous ESG criteria of global multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chlorination technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational advantages and scope limitations for potential partners evaluating this route for their own production needs. Understanding these nuances is essential for making informed decisions about process adoption and technology transfer.
Q: What is the primary advantage of using CuCl2 in this chlorination process?
A: CuCl2 serves a dual role as both the oxidant and the chlorine source, eliminating the need for hazardous molecular chlorine gas and simplifying the workup procedure significantly.
Q: How does this method improve regioselectivity compared to traditional methods?
A: The mechanism involves a single-electron transfer forming a specific radical cation intermediate that favors para-substitution, resulting in high selectivity without needing amino protection groups.
Q: Can the copper catalyst be recycled in this process?
A: Yes, the reduced CuCl formed during the reaction can be regenerated back to CuCl2 by introducing oxygen, allowing for catalyst recycling and reducing heavy metal waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Chloro-2-methylaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in the lab are faithfully reproduced on an industrial scale. We understand that consistency is paramount; therefore, our stringent purity specifications and rigorous QC labs guarantee that every batch of 4-chloro-2-methylaniline or related intermediates meets the exacting standards required for API synthesis. Our commitment to quality assurance means that we do not just supply chemicals; we deliver reliability and peace of mind to your production lines.
We invite you to collaborate with us to leverage this advanced chlorination technology for your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this greener route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us be your partner in driving efficiency and innovation in your supply chain.
