Advanced Synthesis of Racemic Camptothecin Intermediates for Commercial API Manufacturing
Advanced Synthesis of Racemic Camptothecin Intermediates for Commercial API Manufacturing
The pharmaceutical industry's relentless pursuit of potent anticancer agents has placed camptothecin derivatives at the forefront of oncology research, driving an urgent demand for efficient manufacturing pathways for their complex intermediates. Patent CN112300184B, published in late 2022, introduces a groundbreaking preparation method for a critical three-membered ring compound, specifically 4-ethyl-4-hydroxy-7,8-dihydro-1H-pyrano[3,4-f]indolizine-3,6,10(4H)-trione, which serves as a pivotal precursor for racemic camptothecin. This technical disclosure represents a significant leap forward in process chemistry, addressing long-standing bottlenecks associated with the construction of the CDE ring system found in topoisomerase I inhibitors. By leveraging a strategic combination of directed lithiation, oxidative cyclization, and transition metal catalysis, this novel route offers a robust alternative to legacy syntheses that have historically plagued manufacturers with low throughput and prohibitive costs. For R&D directors and supply chain leaders, understanding the nuances of this patent is essential for securing a reliable camptothecin intermediate supplier capable of meeting the rigorous purity standards required for clinical and commercial API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the camptothecin CDE ring has been fraught with significant chemical and economic challenges, as evidenced by earlier literature such as J. Med. Chem. 1980 and J. Org. Chem. 1993. These conventional pathways often rely on multi-step sequences that suffer from inherently low overall yields, primarily due to the instability of intermediates and the requirement for harsh reaction conditions that degrade product quality. For instance, traditional Friedlander condensation strategies frequently necessitate expensive, hard-to-source starting materials that create substantial supply chain vulnerabilities and inflate the cost of goods sold (COGS). Furthermore, the purification of intermediates in these older routes often involves complex chromatographic separations that are difficult to translate from the laboratory bench to kiloton-scale manufacturing, leading to inconsistent batch-to-batch quality. The reliance on such inefficient methodologies not only extends lead times for high-purity pharmaceutical intermediates but also generates excessive chemical waste, posing environmental compliance risks that modern green chemistry initiatives seek to eliminate.
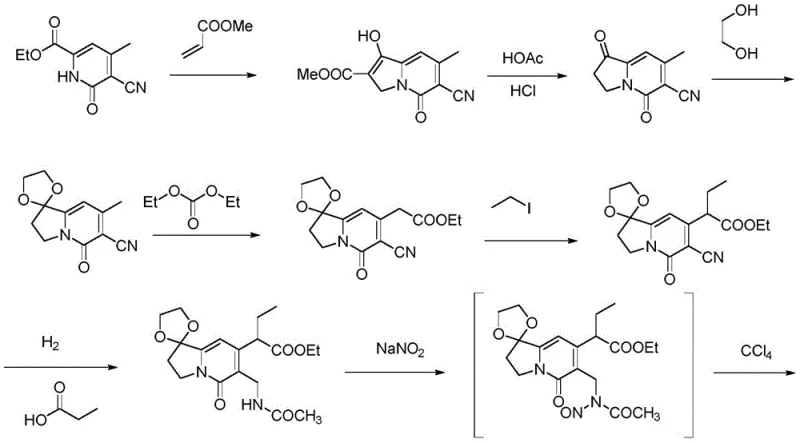
The Novel Approach
In stark contrast to these legacy methods, the methodology outlined in CN112300184B presents a streamlined, eight-step synthetic trajectory that prioritizes atom economy and operational simplicity without compromising on structural integrity. The innovation lies in the selection of 6-chloro-2-methoxynicotinic acid as a commercially abundant and inexpensive starting scaffold, which effectively bypasses the need for exotic reagents. This new approach integrates a highly efficient lactone formation followed by a precise oxidative sequence to construct the pyranone core, ultimately culminating in a palladium-catalyzed carbonylation that installs the necessary ester functionality with high fidelity. By optimizing reaction parameters such as temperature and stoichiometry, this route achieves superior yields at each stage, significantly reducing the accumulation of impurities that typically burden downstream processing. For procurement managers, this translates to a drastic simplification of the supply chain, as the availability of bulk commodity chemicals replaces the dependency on specialized, low-volume precursors, thereby enhancing supply continuity and reducing the risk of production stoppages.
![Novel patented synthesis route for 4-ethyl-4-hydroxy-7,8-dihydro-1H-pyrano[3,4-f]indolizine-3,6,10(4H)-trione](/insights/img/camptothecin-intermediate-synthesis-pharma-supplier-20260305130750-05.webp)
Mechanistic Insights into the Multi-Step Cascade Synthesis
The core of this synthetic strategy relies on a sophisticated orchestration of organometallic and oxidative transformations, beginning with a directed ortho-lithiation that sets the stereochemical and structural foundation for the entire molecule. In the initial step, 6-chloro-2-methoxynicotinic acid is treated with a sterically hindered base system comprising n-butyllithium and 2,2,6,6-tetramethylpiperidine at cryogenic temperatures ranging from -75°C to -60°C. This specific thermal window is critical for generating the reactive lithiated species while suppressing competing nucleophilic attacks, ensuring that the subsequent addition of 1-penten-3-one proceeds with high regioselectivity to form the desired lactone intermediate. Following this, the protocol employs a mild reduction using sodium borohydride or red aluminum to convert the lactone into a diol, which is then subjected to ozonolysis at -75°C to -65°C. This oxidative cleavage is meticulously controlled to generate a hemiacetal, which is immediately oxidized using an iodine-base system to yield a stable pyranone structure, demonstrating a masterful control over oxidation states that prevents over-oxidation or degradation of the sensitive heterocyclic core.
Further down the synthetic line, the introduction of the propyl ester moiety is achieved through a palladium-catalyzed carbonylation under carbon monoxide atmosphere, a step that exemplifies the route's industrial viability. The use of palladium acetate in conjunction with 1,3-bis(diphenylphosphine)propane (dppp) as a ligand facilitates the insertion of carbon monoxide into the carbon-halogen bond at elevated temperatures of 80°C to 100°C, a transformation that is both robust and scalable. Subsequent demethylation using trimethylchlorosilane and sodium iodide cleanly reveals the pyridone nitrogen, setting the stage for the final ring-closing event. The concluding cyclization with tert-butyl acrylate in the presence of cesium carbonate, followed by acid-mediated decarboxylation with trifluoroacetic acid, efficiently constructs the final indolizine trione framework. This mechanistic pathway is designed to minimize the formation of regioisomers and byproducts, ensuring that the final API intermediate meets the stringent purity specifications required for oncology drug development.
How to Synthesize 4-ethyl-4-hydroxy-7,8-dihydro-1H-pyrano[3,4-f]indolizine-3,6,10(4H)-trione Efficiently
Executing this synthesis requires precise adherence to the patented conditions to maximize yield and safety, particularly during the cryogenic lithiation and ozonolysis phases which demand specialized equipment and rigorous temperature monitoring. The process begins with the preparation of the lithiation reagent under an inert nitrogen atmosphere, followed by the controlled addition of the acid substrate and the enone electrophile, necessitating careful quenching and extraction protocols to isolate the lactone intermediate in high purity. Operators must maintain strict control over the stoichiometric ratios, specifically the 1:2 to 1:4 molar ratio of substrate to lithiation reagent, to prevent incomplete conversion or excessive side reactions. As the synthesis progresses to the carbonylation step, safety protocols regarding carbon monoxide handling become paramount, requiring closed-system reactors capable of maintaining positive pressure while ensuring effective gas-liquid mass transfer for optimal catalytic turnover. The detailed standardized synthesis steps, including specific workup procedures and purification parameters like column chromatography solvent systems, are outlined below to guide process engineers in replicating this high-efficiency route.
- Perform directed lithiation of 6-chloro-2-methoxynicotinic acid followed by addition of 1-penten-3-one to form the lactone precursor.
- Execute sequential reduction, ozonolysis, and iodine-mediated oxidation to construct the pyranone core structure.
- Complete the synthesis via palladium-catalyzed carbonylation, demethylation, and final cyclization with tert-butyl acrylate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers profound advantages for organizations seeking cost reduction in anticancer drug manufacturing, primarily driven by the substitution of expensive specialty chemicals with widely available commodity feedstocks. The reliance on 6-chloro-2-methoxynicotinic acid as the starting material fundamentally alters the cost structure of the synthesis, as this compound is produced on a massive scale for other industrial applications, ensuring a stable and competitive pricing landscape that shields manufacturers from volatile market fluctuations. Moreover, the high yields reported in the patent examples, such as the 98% yield in the reduction step and 97.8% in the ozonolysis step, imply a significant reduction in raw material consumption per kilogram of final product, directly translating to lower variable costs and improved margin potential for the final API. This efficiency is further compounded by the simplified purification requirements; the high selectivity of the reactions minimizes the generation of difficult-to-remove impurities, thereby reducing the consumption of silica gel and organic solvents associated with extensive chromatographic purification.
- Cost Reduction in Manufacturing: The elimination of costly chiral resolving agents and the use of catalytic amounts of palladium rather than stoichiometric heavy metals significantly lowers the direct material costs associated with the synthesis. By avoiding the need for complex chiral resolution steps early in the sequence, the process reduces both the time and the solvent volume required, leading to substantial operational expenditure savings. Additionally, the ability to filter and potentially recycle the palladium catalyst after the carbonylation step offers a secondary avenue for cost optimization, reducing the dependency on precious metal markets. The overall shorter synthetic route compared to prior art means fewer unit operations, less energy consumption for heating and cooling, and reduced labor hours, all of which contribute to a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: Utilizing starting materials that are common in the fine chemical industry mitigates the risk of supply disruptions that often plague niche pharmaceutical intermediates. The robustness of the reaction conditions, particularly the tolerance of the carbonylation step to standard industrial reactor setups, ensures that the process can be easily transferred between different manufacturing sites without significant re-validation burdens. This flexibility allows supply chain heads to diversify their vendor base, engaging multiple contract manufacturing organizations (CMOs) who possess standard hydrogenation and carbonylation capabilities, thus preventing single-source bottlenecks. Furthermore, the stability of the intermediates generated throughout the sequence allows for potential stockpiling of key precursors, providing a buffer against unexpected demand surges or logistical delays in the global distribution network.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that avoid extreme pressures or temperatures outside the range of standard glass-lined or stainless-steel reactors. The waste profile of this synthesis is notably cleaner than traditional routes, as the avoidance of harsh acidic or basic hydrolysis steps reduces the load on wastewater treatment facilities. The use of ozone, while requiring safety controls, generates oxygen as a byproduct, minimizing the formation of toxic heavy metal waste streams often associated with stoichiometric oxidants like chromium or manganese salts. This alignment with green chemistry principles not only simplifies regulatory compliance regarding effluent discharge but also enhances the corporate sustainability profile of the manufacturing entity, a factor increasingly weighted in vendor selection criteria by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this technology for large-scale production of camptothecin analogs. Understanding these details is crucial for technical teams evaluating the integration of this process into existing manufacturing portfolios.
Q: What are the critical temperature controls for the lithiation step in this camptothecin intermediate synthesis?
A: The lithiation reaction requires strict cryogenic conditions between -75°C and -60°C to ensure regioselectivity and prevent side reactions during the addition of 1-penten-3-one.
Q: How does this novel route improve upon traditional camptothecin CDE ring synthesis methods?
A: Unlike conventional methods suffering from low yields and expensive raw materials, this patent utilizes cheap starting materials like 6-chloro-2-methoxynicotinic acid and achieves significantly higher overall yields through a streamlined 8-step sequence.
Q: Is the palladium catalyst used in the carbonylation step recoverable for cost efficiency?
A: The process involves filtering off the palladium reagent using celite after the reaction, allowing for potential recovery and recycling strategies to further reduce heavy metal costs in large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-ethyl-4-hydroxy-7,8-dihydro-1H-pyrano[3,4-f]indolizine-3,6,10(4H)-trione Supplier
As the global demand for next-generation topoisomerase inhibitors continues to rise, partnering with a technically proficient CDMO is essential for navigating the complexities of bringing these potent therapeutics to market. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from pilot plant to full-scale manufacturing without compromising on quality or timeline. Our state-of-the-art facilities are equipped to handle the specific requirements of this synthesis, including cryogenic lithiation capabilities and high-pressure carbonylation reactors, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the pharmaceutical industry. We understand that consistency is key in API manufacturing, and our dedicated process chemistry team is committed to continuous optimization to further enhance yields and reduce costs for our partners.
We invite you to engage with our technical procurement team to discuss how this patented route can be tailored to your specific supply chain needs and volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this more efficient synthesis method compared to your current supply sources. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven technical capability. Let us collaborate to secure your supply of high-purity camptothecin intermediates and accelerate the development of life-saving anticancer medications.
