Advanced Synthesis of Kinase Inhibitor Intermediates: Scalable Routes for Commercial Production
Introduction to Advanced Intermediate Synthesis
The pharmaceutical industry constantly seeks robust, scalable, and safe pathways for constructing complex heterocyclic scaffolds essential for modern oncology therapeutics. Patent CN102180836B discloses a groundbreaking methodology for the synthesis of 3-(4-methyl-imidazol-1-yl)-5-trifluoromethyl-phenyl amine, designated as Compound (I). This molecule serves as a critical building block for substituted pyrimidylaminobenzamides, a class of compounds known to inhibit tyrosine protein kinases such as c-Abl, Bcr-Abl, and VEGF-R, which are pivotal targets in the treatment of neoplastic diseases including leukemia. As a reliable pharmaceutical intermediate supplier, understanding the nuances of this patent is vital for securing the supply chain of next-generation kinase inhibitors. The disclosed technology moves away from legacy processes that rely on hazardous reagents and extreme thermal conditions, offering a streamlined approach that aligns with modern green chemistry principles and commercial viability.
The strategic importance of Compound (I) cannot be overstated, as it forms the core structural motif for potent anticancer agents. Traditional synthetic routes often suffer from low atom economy and significant safety liabilities, creating bottlenecks in the manufacturing of active pharmaceutical ingredients (APIs). By leveraging the innovative pathways detailed in this intellectual property, manufacturers can achieve substantial cost reduction in API manufacturing while ensuring a consistent supply of high-quality material. The patent outlines multiple embodiments, ranging from nitro-reduction strategies to novel palladium-catalyzed couplings, providing flexibility for process chemists to optimize based on available infrastructure and raw material costs. This report delves deep into the technical merits and commercial implications of these synthetic advancements.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
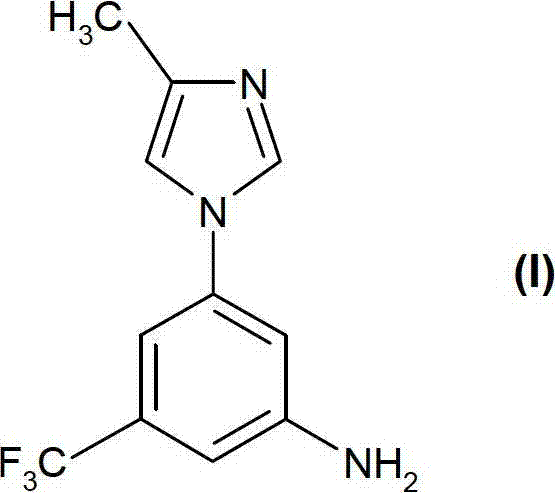
Prior art methodologies for synthesizing Compound (I) were fraught with significant operational challenges that hindered efficient commercial scale-up. As illustrated in the background of the invention, the conventional four-step synthesis initiated with an aromatic substitution reaction between 4-methylimidazole and a fluorinated benzonitrile derivative. This initial step notoriously required high energy input, specifically heating to 150°C in dimethylacetamide, which poses risks of thermal runaway and decomposition. Furthermore, subsequent transformations involved the conversion of a carboxylic acid intermediate using diphenylphosphoryl azide via a Curtius rearrangement. This reagent is inherently unstable and potentially explosive, necessitating specialized handling equipment and rigorous safety protocols that drive up operational expenditures.
Beyond safety concerns, the legacy process suffered from poor product quality and difficult purification profiles. The Curtius rearrangement often yielded inconsistent results, generating diphenylphosphoric acid by-products that were notoriously difficult to remove from the reaction mixture. Consequently, the resulting carbamate intermediate required purification via column chromatography, a technique that is economically prohibitive and logistically cumbersome on a multi-kilogram or tonne scale. The reliance on chromatography not only increases solvent consumption and waste generation but also extends cycle times, negatively impacting the overall throughput and cost efficiency of the manufacturing campaign. These cumulative inefficiencies underscored the urgent need for a safer, more direct synthetic strategy.
The Novel Approach
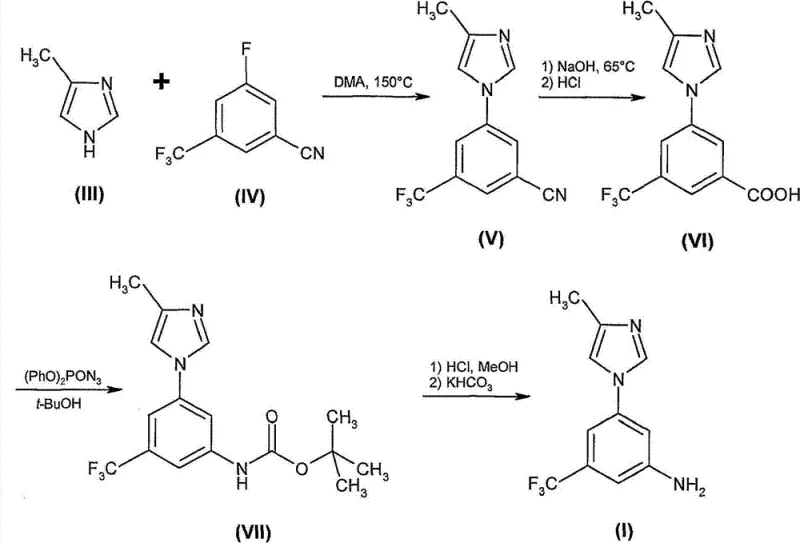
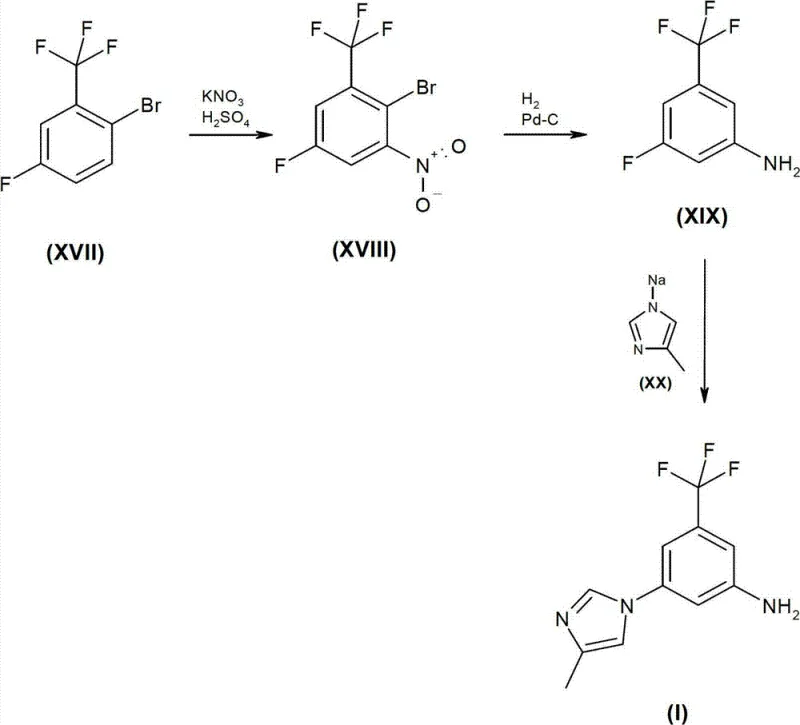
The inventive methods presented in CN102180836B offer a paradigm shift by introducing safer reagents and more convergent synthetic designs. One of the most compelling embodiments involves the utilization of a novel intermediate, 2-bromo-5-fluoro-1-nitro-3-trifluoromethylbenzene (Compound XVIII). This intermediate is accessed via the nitration of commercially available 2-bromo-5-fluoro-benzotrifluoride, a cost-effective starting material. The subsequent catalytic hydrogenation of the nitro group allows for the selective formation of the aniline derivative without the need for hazardous azide chemistry. This route effectively bypasses the high-temperature substitution and the dangerous Curtius rearrangement entirely.
Additionally, the patent describes alternative pathways utilizing transition metal catalysis, such as copper or palladium-mediated coupling reactions. For instance, the direct coupling of 3-bromo-5-trifluoromethyl-phenyl amine with 4-methylimidazole under basic conditions provides a concise entry to the target scaffold. Another embodiment employs a palladium-catalyzed arylation of benzophenone imine followed by hydrolysis, which ensures high regioselectivity and purity. These novel approaches prioritize the use of stable, commercially accessible reagents and facilitate purification through simple crystallization techniques rather than chromatography. By eliminating the most hazardous and inefficient steps of the prior art, the new methodology significantly enhances the safety profile and economic feasibility of producing this valuable kinase inhibitor intermediate.
Mechanistic Insights into Transition Metal Catalyzed Coupling and Reduction
The core of the novel synthetic strategy relies heavily on precise control over aromatic substitution and reduction mechanisms. In the embodiments utilizing transition metal catalysts, such as copper(I) iodide or palladium acetate, the reaction proceeds through a coordinated cycle of oxidative addition, ligand exchange, and reductive elimination. For example, in the coupling of aryl halides with 4-methylimidazole, the presence of a bidentate ligand like 1,2-diaminocyclohexane or Xantphos stabilizes the metal center, facilitating the activation of the carbon-halogen bond even in the presence of electron-withdrawing trifluoromethyl groups. This mechanistic understanding is crucial for R&D directors aiming to optimize reaction parameters, as the choice of base (e.g., cesium carbonate vs. potassium carbonate) and solvent (e.g., diglyme vs. NMP) directly influences the rate of oxidative addition and the suppression of side reactions.
Furthermore, the selective reduction of nitro groups in the presence of other sensitive functionalities is a key mechanistic feature of this patent. The use of catalytic hydrogenation over palladium on carbon allows for the clean conversion of nitro-aromatics to anilines. However, care must be taken to prevent hydrodehalogenation, particularly when bromine or fluorine substituents are present on the ring. The patent details specific conditions, such as controlled temperature and pressure, to ensure that the nitro group is reduced while retaining the halogen handle for subsequent coupling steps if necessary. In routes where debromination is desired, the conditions are adjusted to promote hydrogenolysis of the carbon-bromine bond. This level of mechanistic control ensures that the final product, Compound (I), is obtained with minimal impurities, thereby simplifying downstream processing and ensuring compliance with stringent regulatory standards for pharmaceutical intermediates.
Impurity control is another critical aspect addressed by the mechanistic design of these routes. In the palladium-catalyzed pathways involving benzophenone imine, the formation of regioisomers is a potential risk. The patent demonstrates that through careful selection of ligands and reaction temperatures, the formation of unwanted constitutional isomers can be minimized. Moreover, the final hydrolysis step converts the imine protection group back to the free amine, and subsequent salt formation with hydrochloric acid allows for the precipitation of the product in high purity. The ability to purge residual palladium to levels as low as 0.5 ppm through activated carbon treatment highlights the robustness of the purification protocol. This attention to detail in the reaction mechanism translates directly to a cleaner impurity profile, reducing the burden on quality control laboratories and ensuring the material is fit for purpose in GMP environments.
How to Synthesize 3-(4-Methyl-Imidazol-1-Yl)-5-Trifluoromethyl-Phenyl Amine Efficiently
The synthesis of this high-value intermediate can be executed through several validated pathways, each offering distinct advantages depending on the starting materials available. The general procedure involves the preparation of a functionalized aniline precursor, followed by N-arylation with 4-methylimidazole. For the most cost-effective route, one may start with 2-bromo-5-fluoro-benzotrifluoride, subjecting it to nitration to install the nitro group ortho to the bromine. Subsequent catalytic hydrogenation reduces the nitro group to an amine, yielding 3-fluoro-5-trifluoromethyl-phenyl amine. This aniline is then reacted with 4-methylimidazole in the presence of a strong base like sodium hydride in a polar aprotic solvent such as NMP at elevated temperatures. The detailed standardized synthesis steps for optimizing yield and purity are provided in the guide below.
- Preparation of Novel Intermediate: Nitration of 2-bromo-5-fluoro-benzotrifluoride to form 2-bromo-5-fluoro-1-nitro-3-trifluoromethylbenzene (XVIII).
- Selective Reduction: Catalytic hydrogenation of the nitro group to an amine while managing halogen substituents.
- Final Coupling: N-arylation of 4-methylimidazole with the fluorinated aniline derivative using base and heat.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic routes described in CN102180836B offers transformative benefits regarding cost stability and operational continuity. The primary advantage lies in the elimination of hazardous reagents like diphenylphosphoryl azide. By removing the need for specialized safety infrastructure and expensive hazard mitigation measures associated with azide chemistry, the overall manufacturing overhead is significantly reduced. Furthermore, the shift away from chromatographic purification to crystallization-based isolation drastically cuts down on solvent usage and waste disposal costs. Crystallization is inherently more scalable and reproducible than column chromatography, allowing for larger batch sizes and shorter cycle times, which directly contributes to cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The new methodologies utilize commercially available and inexpensive starting materials, such as 2-bromo-5-fluoro-benzotrifluoride and 4-methylimidazole. By avoiding the multi-step sequence involving nitrile hydrolysis and Curtius rearrangement, the number of unit operations is reduced. Fewer steps mean less labor, lower energy consumption, and reduced equipment occupancy time. Additionally, the avoidance of precious metal catalysts in certain embodiments (using copper instead of palladium) further lowers the raw material cost basis. The ability to recycle solvents like toluene and NMP in a closed-loop system enhances the economic efficiency of the process, ensuring a competitive pricing structure for the final intermediate.
- Enhanced Supply Chain Reliability: Reliance on exotic or custom-synthesized reagents often introduces supply chain fragility. The novel routes prioritize commodity chemicals that are widely available from multiple global suppliers, mitigating the risk of raw material shortages. The robustness of the catalytic hydrogenation and nucleophilic substitution steps ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results. This reliability is crucial for maintaining the continuity of API production schedules, especially for oncology drugs where patient demand is critical. The simplified process flow also reduces the lead time for high-purity pharmaceutical intermediates, allowing for faster response to market fluctuations.
- Scalability and Environmental Compliance: The processes described are designed with scalability in mind, utilizing standard reactor configurations and avoiding extreme conditions that limit vessel size. The reduction in hazardous waste generation, particularly the elimination of phosphorus-containing by-products and azide residues, simplifies environmental compliance and wastewater treatment. This aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing site against regulatory changes. The high atom economy of the direct coupling routes minimizes waste at the source, supporting sustainability goals while maintaining high throughput capabilities suitable for commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this kinase inhibitor intermediate. These answers are derived directly from the technical disclosures within the patent documentation to provide accurate guidance for stakeholders evaluating this technology.
Q: How does this new synthesis method improve safety compared to conventional routes?
A: The conventional route utilizes diphenylphosphoryl azide, a hazardous reagent requiring strict safety protocols. The novel methods described in CN102180836B replace this with safer catalytic hydrogenation and nucleophilic aromatic substitution, significantly reducing operational risk.
Q: What are the purity specifications achievable with the new crystallization methods?
A: The patent demonstrates that through optimized recrystallization from solvents like toluene or heptane, HPLC purity exceeding 99% can be achieved, eliminating the need for expensive and time-consuming chromatographic purification.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the process avoids high-energy conditions (150°C) where possible and utilizes commercially available starting materials like 2-bromo-5-fluoro-benzotrifluoride, making it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(4-Methyl-Imidazol-1-Yl)-5-Trifluoromethyl-Phenyl Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our technical team has extensively analyzed the pathways disclosed in CN102180836B and possesses the expertise to implement these advanced synthetic routes effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of transition metal catalysis and catalytic hydrogenation, adhering to stringent purity specifications and rigorous QC labs to guarantee material that exceeds industry standards.
We invite you to collaborate with us to leverage these technological advancements for your drug development programs. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you secure a supply chain that is not only cost-effective and reliable but also aligned with the highest standards of safety and environmental stewardship, ensuring the successful commercialization of your therapeutic candidates.
