Advanced Synthesis of Procaterol Hydrochloride Impurity for Pharmaceutical Quality Control
Advanced Synthesis of Procaterol Hydrochloride Impurity for Pharmaceutical Quality Control
The pharmaceutical industry faces constant challenges in maintaining the highest standards of drug safety, particularly regarding the identification and quantification of impurities in Active Pharmaceutical Ingredients (APIs). A recent technological breakthrough, documented in patent CN112645875A, introduces a highly efficient preparation method for Procaterol Hydrochloride Impurity I, a critical reference standard for quality control laboratories. This innovation addresses the historical difficulty in acquiring specific degradation products or synthetic byproducts, which are essential for validating analytical methods and ensuring batch consistency. By leveraging a streamlined three-step synthetic pathway, this method transforms the readily available API, Procaterol Hydrochloride, into its corresponding aldehyde impurity with remarkable efficiency. For R&D directors and quality assurance teams, this represents a significant advancement in impurity profiling capabilities, allowing for more rigorous stress testing and stability studies of bronchodilator formulations.
Furthermore, the strategic implications of this synthesis route extend beyond mere laboratory convenience; it offers a robust framework for supply chain resilience in the production of high-purity pharmaceutical intermediates. Traditional methods for generating such specific impurities often involve complex, multi-step sequences starting from basic commodity chemicals, leading to high costs and extended lead times. In contrast, the approach outlined in CN112645875A utilizes a semi-synthetic strategy that capitalizes on the existing molecular scaffold of the drug substance. This not only ensures structural fidelity but also drastically simplifies the logistical burden on procurement departments. As a reliable pharmaceutical intermediate supplier, understanding and adopting such optimized pathways is crucial for maintaining competitive advantage in the global market for reference standards and custom synthesis services.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of specific drug-related impurities like Procaterol Impurity I has been fraught with significant technical and economic hurdles. Conventional approaches often necessitate the construction of the quinolinone core from scratch, involving hazardous reagents, harsh reaction conditions, and multiple purification stages that erode overall yield. These traditional routes frequently suffer from poor regioselectivity, requiring expensive chromatographic separations to isolate the desired isomer from a complex mixture of byproducts. For procurement managers, this translates into exorbitant costs per gram and unpredictable availability, as few manufacturers are willing to undertake such labor-intensive processes for small-volume specialty chemicals. Moreover, the reliance on obscure starting materials creates supply chain vulnerabilities, where a disruption in the availability of a single precursor can halt the entire production of the reference standard, delaying critical quality control operations for the parent drug.
The Novel Approach
The methodology disclosed in patent CN112645875A fundamentally disrupts this paradigm by employing a "top-down" synthetic strategy that begins with the commercially abundant Procaterol Hydrochloride. This novel approach bypasses the need for complex ring construction, focusing instead on precise functional group manipulations: protection, oxidation, and deprotection. By selecting Procaterol Hydrochloride as the starting raw material, the synthesis cost is inherently reduced, and the preparation steps are significantly simplified. The process demonstrates exceptional selectivity, particularly in the oxidative cleavage step, which cleanly converts the secondary alcohol to an aldehyde without over-oxidation to the carboxylic acid. This efficiency saves substantial preparation time and improves overall preparation efficiency, offering a total yield of 50-70%, which is exceptionally high for a three-step sequence involving sensitive functional groups. This shift represents a major leap forward in cost reduction in API manufacturing support services.
Mechanistic Insights into the Three-Step Synthetic Route
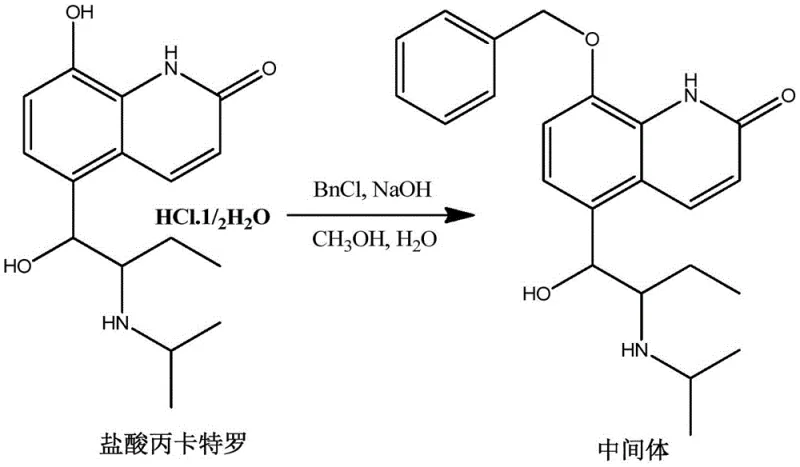
The core of this innovation lies in its elegant chemical logic, which balances reactivity with selectivity to ensure high product purity. The first step involves the protection of the phenolic hydroxyl group at the C8 position of the quinolinone ring. Under alkaline conditions, typically using sodium hydroxide in a methanol-water mixture, the phenol is deprotonated to form a nucleophilic phenoxide ion. This species then undergoes an SN2 reaction with benzyl chloride, installing a benzyl ether protecting group. This protection is critical because it prevents the phenol from interfering with subsequent oxidation steps and shields it from the harsh Lewis acidic conditions used later. The reaction is driven to completion by heating to reflux at approximately 80°C, yielding Intermediate 1 with high conversion rates. This robust protection strategy ensures that the sensitive aromatic system remains intact throughout the synthesis.
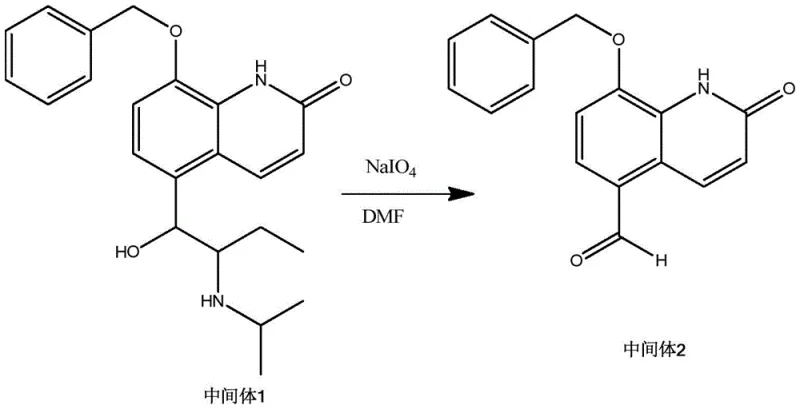
Following protection, the synthesis proceeds to the pivotal oxidative transformation. Intermediate 1 is subjected to oxidative cleavage using sodium periodate (NaIO4) in a polar aprotic solvent like DMF. This step targets the benzylic alcohol side chain, selectively oxidizing it to the corresponding aldehyde functionality found in Impurity I. The choice of sodium periodate is strategic; it is a mild yet effective oxidant that avoids the over-oxidation pitfalls associated with stronger agents like chromic acid. The reaction is conducted at a moderate temperature of 60°C, ensuring rapid kinetics while maintaining thermal stability of the substrate. The resulting Intermediate 2 precipitates upon pouring into ice water, facilitating a simple isolation procedure that avoids the need for column chromatography. This mechanistic precision is key to achieving the high purity levels required for analytical reference standards.
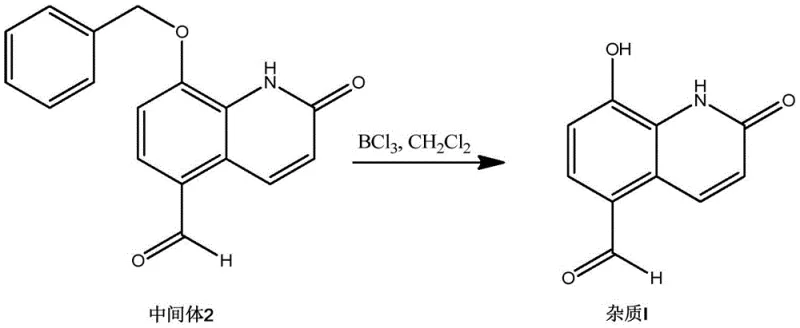
The final stage of the synthesis involves the removal of the benzyl protecting group to reveal the free phenol, thereby regenerating the core structure of the impurity. This deprotection is achieved using boron trichloride (BCl3) in dichloromethane at low temperatures, typically between -5°C and 0°C. Boron trichloride acts as a strong Lewis acid, coordinating with the ether oxygen and facilitating the cleavage of the C-O bond to release the phenol and benzyl chloride. The low-temperature condition is vital to prevent potential side reactions, such as the polymerization of the aldehyde or degradation of the quinolinone ring under acidic conditions. Upon quenching with ice water, the final product, Procaterol Impurity I, precipitates as a white solid. This final step boasts an impressive yield of roughly 97.5%, underscoring the efficiency of the deprotection protocol and the stability of the intermediate aldehyde.
How to Synthesize Procaterol Hydrochloride Impurity Efficiently
Implementing this synthesis requires careful attention to reaction parameters, particularly temperature control and stoichiometry, to maximize yield and purity. The process is designed to be scalable, moving seamlessly from gram-scale laboratory optimization to kilogram-scale commercial production. The use of common solvents like methanol, DMF, and dichloromethane ensures that the process is compatible with standard pharmaceutical manufacturing equipment. Detailed operational guidelines emphasize the importance of rapid addition of reagents in the oxidation step to maintain consistent reaction kinetics. For technical teams looking to replicate this high-purity pharmaceutical intermediate synthesis, the following standardized protocol outlines the critical operational phases derived from the patent examples.
- Step 1: Protection of the phenolic hydroxyl group by reacting Procaterol Hydrochloride with benzyl chloride under alkaline conditions to form Intermediate 1.
- Step 2: Oxidative cleavage of the side-chain alcohol in Intermediate 1 using sodium periodate in DMF to yield the aldehyde Intermediate 2.
- Step 3: Deprotection of the benzyl ether in Intermediate 2 using boron trichloride at low temperature to afford the final Procaterol Impurity I.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented method offers profound advantages that directly impact the bottom line and operational reliability of pharmaceutical supply chains. By shifting the starting material to the API itself, the process eliminates the need for sourcing rare or expensive precursors, thereby stabilizing the supply base. This strategic alignment means that fluctuations in the market for basic chemical building blocks have minimal impact on the production of the impurity standard. Furthermore, the simplified workup procedures, which rely primarily on precipitation and filtration rather than resource-intensive chromatography, significantly reduce processing time and solvent consumption. These factors collectively contribute to a more agile and responsive supply chain, capable of meeting the urgent demands of quality control laboratories worldwide.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the elimination of complex synthetic steps and the use of cost-effective reagents. By avoiding transition metal catalysts and expensive oxidants, the direct material costs are substantially lowered. Additionally, the high yields at each step minimize waste generation, reducing the costs associated with waste disposal and raw material loss. The ability to produce the impurity in fewer steps translates to lower labor and utility costs, making the final product more price-competitive for bulk purchasers. This efficiency allows suppliers to offer better pricing structures without compromising on the stringent quality requirements of the pharmaceutical industry.
- Enhanced Supply Chain Reliability: Sourcing Procaterol Hydrochloride as a feedstock leverages an established and robust global supply network, ensuring consistent availability of raw materials. Unlike custom synthesis routes that depend on niche intermediates, this method relies on a commodity drug substance that is widely manufactured. This redundancy in supply sources mitigates the risk of production stoppages due to raw material shortages. Consequently, lead times for delivering high-purity reference standards can be significantly reduced, enabling pharmaceutical companies to accelerate their own drug development and release timelines. This reliability is a critical asset for supply chain heads managing just-in-time inventory systems.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as reflux, filtration, and drying that are easily adapted for large-scale reactors. The absence of heavy metals and the use of relatively benign solvents simplify the environmental compliance landscape, reducing the regulatory burden associated with waste management. The high atom economy of the protection-deprotection sequence ensures that the process aligns with green chemistry principles, appealing to environmentally conscious stakeholders. This scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved smoothly, supporting the growing demand for impurity standards as regulatory scrutiny intensifies.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its adoption for internal production or external sourcing. The following questions address common concerns regarding the practical application, purity profiles, and regulatory alignment of this method. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: What is the starting material for this impurity synthesis?
A: The process utilizes Procaterol Hydrochloride itself as the starting raw material, which simplifies sourcing and reduces costs compared to de novo synthesis from basic chemicals.
Q: What is the overall yield of the described method?
A: The patent reports a total reaction yield ranging from 50% to 70%, with individual step yields reaching as high as 97.5% in the final deprotection stage.
Q: How is the purity of the final impurity controlled?
A: High purity (e.g., 99.65% by HPLC) is achieved through selective reactions and simple workup procedures like filtration and recrystallization, avoiding complex chromatographic separations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Procaterol Hydrochloride Impurity Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality impurities play in ensuring drug safety and regulatory compliance. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex molecules like Procaterol Impurity I are available in the quantities you need. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets or exceeds the 99% purity threshold required for reference standards. Our commitment to technical excellence ensures that your quality control workflows remain uninterrupted and compliant with global pharmacopoeia standards.
We invite you to collaborate with us to optimize your supply chain for critical reference materials. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and delivery schedules. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our advanced synthesis capabilities can support your R&D and QA objectives. Let us be your partner in delivering precision and reliability in pharmaceutical intermediate supply.
