Advanced Synthesis of 2-(2,2,2-Trifluoroethoxy)phenol for Scalable Silodosin Production
The pharmaceutical landscape for benign prostatic hyperplasia (BPH) treatment has been significantly shaped by the introduction of Silodosin, a highly selective alpha-1A adrenoceptor antagonist. As detailed in patent CN100534973C, the efficient production of its key building block, 2-(2,2,2-trifluoroethoxy)phenol, represents a critical technological breakthrough for the global supply chain. This specific intermediate dictates the purity profile and cost structure of the final Active Pharmaceutical Ingredient (API). Traditional synthetic pathways have long been plagued by reliance on exotic, unstable reagents that introduce significant volatility into procurement strategies. The novel methodology disclosed in this patent fundamentally重构 s the synthetic logic, shifting from a fragile, high-cost demethylation strategy to a robust, commodity-driven nucleophilic substitution approach. For R&D directors and supply chain heads alike, understanding this transition is paramount, as it offers a pathway to drastically stabilize the manufacturing of this high-value urological agent while mitigating the risks associated with hazardous reagent handling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-(2,2,2-trifluoroethoxy)phenol relied on a route that began with guaiacol derivatives, necessitating a challenging etherification followed by a harsh demethylation step. As illustrated in the legacy pathway below, the initial etherification required 2,2,2-trifluoroiodoethane, a reagent that is not only prohibitively expensive due to import dependencies but also chemically unstable, prone to decomposition, and difficult to store under standard warehouse conditions. Furthermore, the subsequent demethylation step typically employed boron tribromide (BBr3), a highly corrosive and moisture-sensitive Lewis acid that demands cryogenic conditions and generates substantial amounts of boron-containing waste. These factors create a perfect storm of operational inefficiency: high raw material costs, stringent safety protocols for hazardous waste disposal, and a fragile supply chain susceptible to disruptions in the availability of specialized fluorinated iodides. Such a process is inherently unsuitable for the rigorous demands of modern Good Manufacturing Practice (GMP) environments where consistency and safety are non-negotiable.
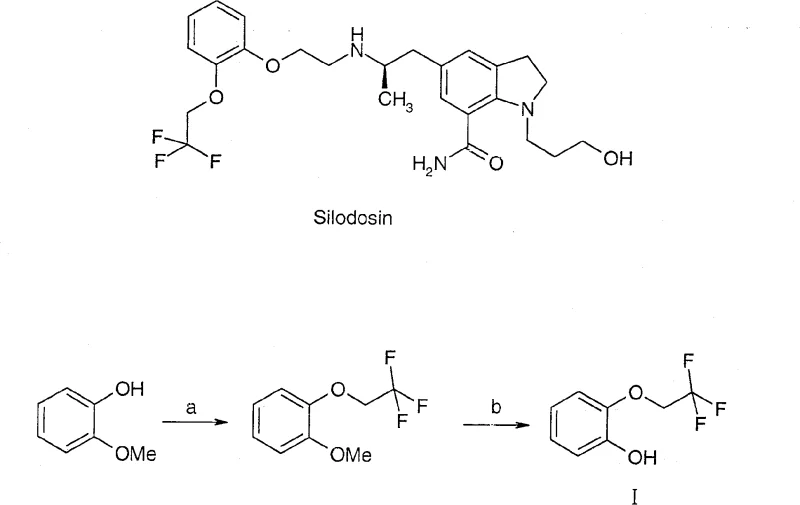
The Novel Approach
In stark contrast, the innovative route presented in patent CN100534973C leverages the activating power of the nitro group to facilitate a direct nucleophilic aromatic substitution. By starting with o-nitrochlorobenzene, a widely available and inexpensive commodity chemical, the process bypasses the need for unstable alkyl halides entirely. The synthesis proceeds through a smooth etherification with 2,2,2-trifluoroethanol, followed by a standard catalytic reduction and a final diazotization-hydrolysis sequence. This strategic pivot eliminates the most dangerous and costly steps of the legacy route, replacing them with unit operations that are well-understood and easily scalable in standard multipurpose reactors. The result is a streamlined workflow that not only enhances the safety profile of the manufacturing site but also significantly lowers the barrier to entry for commercial-scale production, making it an ideal candidate for reliable pharmaceutical intermediates supplier networks aiming for long-term sustainability.
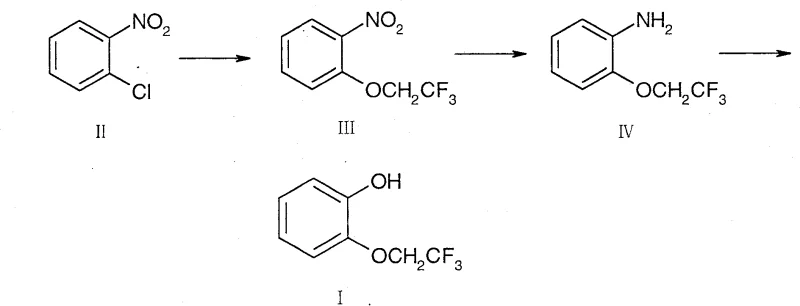
Mechanistic Insights into Phase Transfer Catalyzed Etherification
The cornerstone of this improved synthesis lies in the first step: the nucleophilic aromatic substitution (SNAr) of o-nitrochlorobenzene. The presence of the strong electron-withdrawing nitro group at the ortho position significantly activates the aromatic ring towards nucleophilic attack by destabilizing the chloride leaving group. In the presence of a base such as sodium hydroxide or potassium hydroxide, 2,2,2-trifluoroethanol is deprotonated to form the corresponding alkoxide, a potent nucleophile. However, the reaction efficiency is dramatically enhanced by the inclusion of a phase transfer catalyst, such as tetrabutylammonium bromide (TBAB). This catalyst facilitates the transport of the alkoxide anion from the aqueous phase into the organic phase where the substrate resides, effectively overcoming solubility barriers and accelerating the reaction kinetics. This mechanistic nuance allows the reaction to proceed at moderate temperatures ranging from 0°C to 100°C, ensuring high conversion rates without the thermal degradation issues often seen in more aggressive alkylation protocols.
Following the formation of the nitro-ether intermediate, the process employs a versatile reduction strategy to generate the aniline derivative. This step can be achieved via catalytic hydrogenation using palladium on carbon (Pd/C) or Raney nickel, or alternatively through chemical reduction using iron or zinc powder in acidic media. The resulting aniline is then subjected to diazotization using sodium nitrite and a mineral acid like sulfuric acid at low temperatures (-20°C to 50°C) to form the diazonium salt. Crucially, this unstable intermediate is not isolated; instead, it undergoes immediate hydrolysis in the presence of acid and heat to yield the final phenolic product. This telescoped approach minimizes the handling of energetic diazonium species, thereby enhancing process safety while maintaining a tight control over the impurity profile, which is essential for meeting the stringent purity specifications required for API precursors.
How to Synthesize 2-(2,2,2-Trifluoroethoxy)phenol Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize byproduct formation. The process begins with the careful addition of the phase transfer catalyst and base to the reaction vessel containing the nitro-chloride substrate, followed by the controlled dosing of the trifluoroethanol to manage exotherms. Detailed standard operating procedures (SOPs) must be established for the hydrogenation step to ensure complete reduction of the nitro group without over-reduction or dehalogenation side reactions. Finally, the diazotization and hydrolysis steps demand rigorous temperature monitoring to prevent the accumulation of unreacted diazonium salts, which could pose safety risks. For a comprehensive breakdown of the specific molar ratios, solvent choices, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide provided below.
- Perform nucleophilic aromatic substitution of o-nitrochlorobenzene with 2,2,2-trifluoroethanol using a phase transfer catalyst and base.
- Execute catalytic hydrogenation or chemical reduction of the nitro group to form the corresponding aniline derivative.
- Conduct diazotization of the aniline followed by acid-catalyzed hydrolysis to yield the final phenol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers profound advantages for procurement managers and supply chain directors tasked with optimizing the cost of goods sold (COGS) for Silodosin. The primary driver of value creation is the substitution of high-cost, imported specialty reagents with low-cost, domestically sourced commodity chemicals. By eliminating the dependency on 2,2,2-trifluoroiodoethane and boron tribromide, manufacturers can insulate their production costs from the volatility of the fine fluorine chemical market. Furthermore, the removal of corrosive and hazardous reagents simplifies the engineering requirements for the production facility, reducing the need for specialized corrosion-resistant equipment and lowering capital expenditure (CAPEX) for plant maintenance. This shift translates directly into a more resilient supply chain capable of sustaining continuous production runs without the frequent interruptions caused by reagent shortages or safety incidents.
- Cost Reduction in Manufacturing: The economic impact of this route is driven by the drastic reduction in raw material expenses. 2,2,2-Trifluoroethanol is significantly cheaper and more stable than its iodo-analog, and the avoidance of boron tribromide removes a major cost center associated with both reagent purchase and hazardous waste disposal. Additionally, the use of phase transfer catalysis improves atom economy and reaction throughput, allowing for higher batch yields and reduced solvent consumption per kilogram of product. These cumulative efficiencies result in substantial cost savings that can be passed down the value chain, enhancing the competitiveness of the final API in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of o-nitrochlorobenzene, a bulk chemical produced by numerous suppliers worldwide, ensuring that sourcing is never a single point of failure. Unlike the legacy route, which relies on niche fluorinated building blocks with limited production capacity, this new method leverages a robust infrastructure of basic organic chemical manufacturing. This diversification of the supplier base mitigates the risk of geopolitical disruptions or logistics bottlenecks, guaranteeing a steady flow of high-purity intermediates to downstream API manufacturers. Consequently, lead times for high-purity pharmaceutical intermediates are significantly reduced, enabling faster time-to-market for generic formulations.
- Scalability and Environmental Compliance: The environmental footprint of the process is markedly lower, aligning with increasingly strict global regulations on industrial emissions and waste management. The elimination of boron waste streams simplifies effluent treatment, while the use of catalytic hydrogenation offers a cleaner alternative to stoichiometric metal reductions. This green chemistry profile not only reduces regulatory compliance costs but also enhances the corporate social responsibility (CSR) standing of the manufacturing entity. Moreover, the simplicity of the unit operations—etherification, filtration, hydrogenation, and hydrolysis—makes the process highly amenable to commercial scale-up of complex fluorinated compounds, allowing for seamless transition from pilot plant to multi-ton production scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements. The answers below clarify the specific advantages of the new method over existing art, focusing on practical operational parameters and quality outcomes.
Q: Why is the new synthetic route for 2-(2,2,2-trifluoroethoxy)phenol superior to the conventional method?
A: The new route utilizes inexpensive and stable o-nitrochlorobenzene instead of costly 2,2,2-trifluoroiodoethane. It also eliminates the need for hazardous boron tribromide (BBr3) for demethylation, significantly improving safety and reducing waste treatment costs.
Q: What are the critical reaction conditions for the etherification step?
A: The etherification requires alkaline conditions (e.g., NaOH or KOH) and a phase transfer catalyst like tetrabutylammonium bromide. The reaction temperature is maintained between 0°C and 100°C to ensure high conversion without decomposition.
Q: How does this process impact the supply chain for Silodosin manufacturing?
A: By relying on commodity chemicals like o-nitrochlorobenzene and avoiding imported, unstable fluorinated iodides, the process ensures consistent raw material availability and reduces lead times for large-scale API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2,2,2-Trifluoroethoxy)phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of Silodosin depends on a secure and high-quality supply of its critical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped to handle fluorinated chemistry safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards. We understand the complexities of bringing urological APIs to market and are committed to being a strategic partner in your supply chain.
We invite you to engage with our technical team to explore how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic potential of switching to this superior manufacturing method. We encourage you to contact our technical procurement team today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of this advanced intermediate into your production schedule.
