Scalable Synthesis of Benzothiophene-3-Acetic Acid via Mild Chloromethylation and Cyanation
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for critical heterocyclic building blocks, and the recent disclosure in patent CN113896709B offers a transformative approach to synthesizing benzothiophene-3-acetic acid (CAS: 1131-09-5). This compound serves as a vital fragment in the construction of complex drug molecules, yet its historical production has been plagued by operational hazards and economic inefficiencies. The patented methodology introduces a streamlined three-step sequence starting from readily available benzothiophene, effectively bypassing the need for malodorous thiols or expensive transition metal catalysts. By leveraging mild electrophilic chloromethylation followed by nucleophilic cyanation and final hydrolysis, this process establishes a new benchmark for reliable pharma intermediate supplier capabilities. The technical breakthrough lies not just in the chemical transformation itself, but in the holistic optimization of safety, cost, and scalability, addressing the core pain points of modern API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
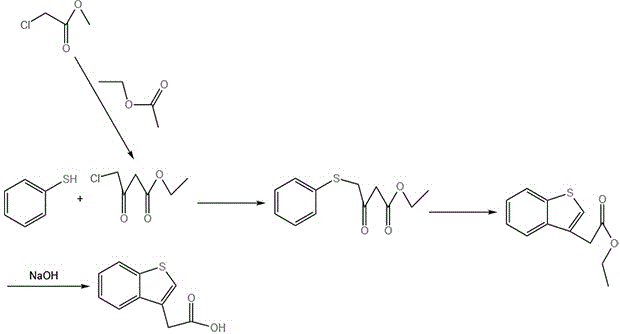
Historically, the synthesis of benzothiophene-3-acetic acid has relied on routes that are increasingly untenable for modern industrial standards due to safety and cost constraints. One prominent legacy method initiates from thiophenol, proceeding through a cyclization to form an ethyl ester intermediate before hydrolysis. As illustrated in the reaction scheme below, this pathway is not only lengthy, requiring four distinct steps, but it also necessitates the handling of thiophenol, a liquid renowned for its intensely unpleasant odor and toxicity, which complicates worker safety and waste management protocols significantly.
Furthermore, alternative strategies reported in literature, such as those utilizing palladium-catalyzed carbonylation, introduce severe economic and engineering barriers. These methods typically employ benzothiophene-3-methylene bromide reacting with carbon monoxide under pressure in the presence of palladium hydroxide. The reliance on noble metal catalysts drastically inflates raw material costs, while the requirement for high-pressure equipment to handle carbon monoxide gas imposes stringent safety regulations and capital expenditure requirements that are often prohibitive for small-to-medium batch production. Additionally, routes starting from benzothiophene-3-carbaldehyde involve expensive and less accessible starting materials, further limiting their commercial viability for large-scale cost reduction in pharmaceutical intermediates manufacturing.
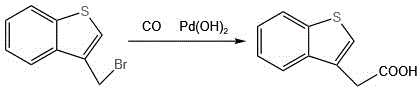
The Novel Approach
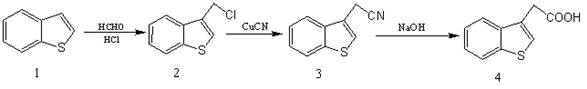
In stark contrast to these cumbersome legacy processes, the novel approach detailed in CN113896709B utilizes benzothiophene as the foundational feedstock, a commodity chemical that is odorless, stable, and economically priced. The strategy simplifies the synthetic architecture into three highly efficient operations: chloromethylation, cyano substitution, and alkaline hydrolysis. This route eliminates the need for specialized high-pressure reactors and removes the dependency on precious metal catalysts entirely. By operating at atmospheric pressure and utilizing common solvents like acetic acid and DMF, the process significantly lowers the barrier to entry for commercial production. The transition from hazardous thiols or expensive aldehydes to simple benzothiophene represents a paradigm shift in process chemistry, enabling manufacturers to achieve commercial scale-up of complex pharmaceutical intermediates with greater agility and reduced environmental footprint.
Mechanistic Insights into Electrophilic Chloromethylation and Nucleophilic Substitution
The core of this synthetic innovation lies in the precise control of electrophilic aromatic substitution during the initial chloromethylation step. In this stage, benzothiophene reacts with formaldehyde (generated in situ from paraformaldehyde) and hydrochloric acid within an acetic acid solvent matrix. The reaction mechanism involves the formation of a chloromethyl cation equivalent, which acts as a potent electrophile attacking the electron-rich 3-position of the benzothiophene ring. Maintaining the temperature strictly between 15-20°C is critical; this thermal control prevents poly-chloromethylation and polymerization side reactions, ensuring high regioselectivity for the mono-substituted product. The use of phosphoric acid as a co-catalyst further enhances the generation of the active electrophilic species, driving the reaction to completion without the need for harsh Lewis acids that could degrade the sensitive heterocyclic core.
Following the isolation of the chloromethyl intermediate, the process proceeds to a nucleophilic displacement reaction using cuprous cyanide in a polar aprotic solvent such as DMF. This step converts the reactive chloride leaving group into a nitrile functionality via an SN2-type mechanism. The choice of DMF is strategic, as it effectively solvates the cyanide anion while stabilizing the transition state, facilitating rapid conversion at moderate temperatures of 90-95°C. The subsequent alkaline hydrolysis utilizes sodium hydroxide to cleave the nitrile triple bond, forming the carboxylate salt which is then acidified to precipitate the final acid. This mechanistic sequence ensures that impurities are either volatile or remain in the aqueous phase during workup, resulting in a product with exceptional purity profiles suitable for downstream medicinal chemistry applications.
How to Synthesize Benzothiophene-3-Acetic Acid Efficiently
The operational simplicity of this three-step protocol makes it an ideal candidate for technology transfer and immediate implementation in pilot plants. The process begins with the careful addition of paraformaldehyde to an acid mixture, followed by the controlled introduction of the benzothiophene substrate to manage exotherms. Detailed standard operating procedures regarding stoichiometry, specifically the molar ratios of benzothiophene to paraformaldehyde and hydrochloric acid, are critical for maximizing yield. The subsequent cyanation and hydrolysis steps utilize standard unit operations such as filtration, extraction, and distillation, requiring no specialized equipment beyond standard glass-lined or stainless steel reactors. For a comprehensive breakdown of the exact reagent quantities, temperature ramps, and workup parameters, please refer to the standardized synthesis guide provided below.
- Perform chloromethylation of benzothiophene using hydrochloric acid and paraformaldehyde in acetic acid solvent at 15-20°C to obtain 3-chloromethylbenzothiophene.
- Conduct cyano substitution by reacting the chloromethyl intermediate with cuprous cyanide in DMF at 90-95°C to yield benzothiophene-3-acetonitrile.
- Execute alkaline hydrolysis of the nitrile using sodium hydroxide under reflux, followed by acidification to precipitate pure benzothiophene-3-acetic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers profound advantages that directly impact the bottom line and supply chain resilience. By shifting the raw material base from specialty thiols or aldehydes to commodity benzothiophene, procurement teams can leverage existing bulk supply chains that are less susceptible to market volatility. The elimination of noble metal catalysts removes a significant cost center associated with both the purchase of palladium salts and the downstream processing required to remove trace metal residues to meet regulatory limits. Furthermore, the avoidance of high-pressure carbon monoxide reactions reduces the need for specialized autoclaves, allowing production to occur in standard multipurpose facilities, thereby increasing asset utilization rates and reducing capital depreciation costs per kilogram of output.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the substitution of expensive reagents with low-cost commodities. The removal of palladium hydroxide eliminates the need for costly catalyst recovery systems or scavenging resins, which are typically required to meet strict heavy metal specifications in API production. Additionally, the solvents employed, such as acetic acid and DMF, are widely available industrial chemicals with stable pricing structures, insulating the production cost from the fluctuations often seen with specialty fine chemicals. The overall yield improvement across the three steps, combined with simplified purification workflows, results in a substantial reduction in the cost of goods sold (COGS) compared to legacy four-step routes.
- Enhanced Supply Chain Reliability: Supply continuity is significantly bolstered by the use of benzothiophene, a chemical produced at massive scales for various industries, ensuring that raw material shortages are highly unlikely. Unlike methods relying on custom-synthesized precursors like benzothiophene-3-carbaldehyde, which may have limited suppliers and long lead times, the starting material for this process is a shelf-stable solid that can be stockpiled easily. The robustness of the reaction conditions also means that production schedules are less prone to delays caused by equipment maintenance or safety interlocks associated with high-pressure or toxic gas handling, guaranteeing more predictable delivery timelines for downstream customers.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved, aligning with modern green chemistry principles and reducing the burden on waste treatment facilities. The absence of malodorous thiophenol eliminates a major source of community complaints and regulatory scrutiny regarding air emissions. Moreover, the aqueous waste streams generated during the hydrolysis and workup phases are simpler to treat compared to the complex organic waste from multi-step cyclizations. The mild reaction temperatures and atmospheric pressure operations lower energy consumption for heating and cooling, contributing to a lower carbon footprint per unit of production, which is increasingly important for meeting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on why this method represents a superior choice for industrial adoption. Understanding these nuances helps stakeholders make informed decisions regarding process validation and vendor qualification.
Q: Why is the new benzothiophene-based route preferred over the thiophenol method?
A: The traditional thiophenol route involves malodorous raw materials that pose significant safety and environmental handling challenges, whereas the new method utilizes odorless, commercially available benzothiophene.
Q: Does this synthesis require expensive noble metal catalysts?
A: No, unlike palladium-catalyzed carbonylation methods which require costly Pd(OH)2 and high-pressure CO equipment, this process relies on common inorganic reagents like cuprous cyanide and sodium hydroxide.
Q: What are the purity specifications achievable with this method?
A: The method employs mild reaction conditions and straightforward extraction workups, allowing for the production of high-purity white powder suitable for pharmaceutical applications without complex chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzothiophene-3-Acetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory discovery to commercial reality requires a partner with deep technical expertise and robust manufacturing infrastructure. Our team has extensively analyzed the pathway described in CN113896709B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring this efficient process to market. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to ensure that every batch of benzothiophene-3-acetic acid meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, minimizing risks during your drug development phases.
We invite you to collaborate with us to optimize your supply chain for this critical intermediate. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this novel synthesis route can improve your project economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can support your long-term strategic goals.
