Optimizing Melphalan Production: A Technical Breakthrough in Oncology API Manufacturing
Optimizing Melphalan Production: A Technical Breakthrough in Oncology API Manufacturing
The global demand for high-purity oncology therapeutics continues to surge, placing immense pressure on supply chains to deliver complex active pharmaceutical ingredients (APIs) with both economic efficiency and rigorous quality standards. In this context, the synthesis of Melphalan, a critical alkylating agent used in the treatment of multiple myeloma and ovarian cancer, represents a significant technical challenge due to its sensitive nitrogen mustard functionality. A pivotal advancement in this domain is detailed in patent CN100491339C, which outlines a refined synthetic methodology that fundamentally shifts the paradigm from traditional phthalic anhydride protection to a more robust tert-butoxycarbonyl (BOC) strategy. This innovation not only addresses the longstanding issues of toxicity and low yield associated with legacy processes but also establishes a new benchmark for the commercial scale-up of complex oncology intermediates. By leveraging mild reaction conditions and eliminating hazardous solvents, this process offers a compelling value proposition for stakeholders focused on sustainable and cost-effective pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Melphalan has been plagued by significant inefficiencies rooted in the protection chemistry of the amino acid backbone. Traditional routes, such as those described in US3032584, rely heavily on phthalic anhydride to protect the amino group of phenylalanine derivatives. This approach is inherently flawed as it necessitates a cumbersome two-step protection sequence that drastically compromises overall throughput, often resulting in yields as low as 65% for the protection stage alone. Furthermore, these legacy methods frequently employ benzene as a reaction solvent, a known human carcinogen that poses severe occupational health risks and creates substantial regulatory burdens for waste disposal. The reliance on mixed solvent systems like acetone and petroleum ether for recrystallization further exacerbates operational costs, as solvent recovery is difficult and energy-intensive. These factors collectively render conventional methods economically unviable for modern, high-volume pharmaceutical intermediates manufacturing where margin compression is a constant threat.
The Novel Approach
In stark contrast, the novel methodology introduced in CN100491339C revolutionizes the synthesis landscape by substituting the problematic phthalic anhydride moiety with di-tert-butyl dicarbonate ((BOC)2O). This strategic substitution allows for a direct, single-step protection of the amino group under mild ambient temperatures, achieving exceptional conversion rates with yields reaching up to 95%. The stability of the BOC protecting group is perfectly tuned for this specific pathway; it remains inert during the subsequent catalytic hydrogenation and hydroxyethylation steps yet is readily cleaved under acidic conditions in the final stage. This orthogonality simplifies the downstream processing significantly, eliminating the need for complex separation techniques required to remove phthalic acid byproducts. Moreover, the process replaces toxic benzene with safer, greener solvents like absolute ethanol and dichloromethane, aligning the manufacturing protocol with modern environmental, health, and safety (EHS) standards. This transition not only enhances operator safety but also facilitates cost reduction in API manufacturing by streamlining solvent recovery and waste treatment protocols.
Mechanistic Insights into BOC-Protection and Nitrogen Mustard Formation
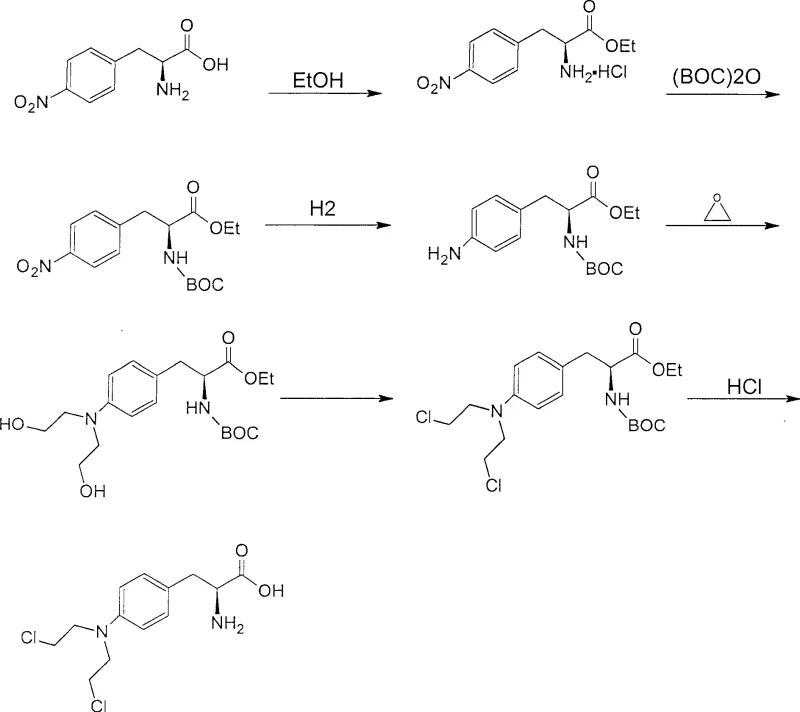
The core of this technical breakthrough lies in the precise manipulation of chemoselectivity throughout the six-step synthetic sequence. The process initiates with the esterification of L-4-nitrophenylalanine using thionyl chloride in absolute ethanol, effectively masking the carboxylic acid as an ethyl ester. Following this, the introduction of the BOC group creates a sterically bulky yet electronically stable carbamate. This protection is crucial because it prevents unwanted side reactions during the subsequent reduction of the nitro group to an amine using palladium on carbon (Pd/C) catalysts. The hydrogenation step proceeds with remarkable efficiency, reportedly achieving 100% yield, indicating that the BOC group does not interfere with the catalyst surface or undergo premature hydrogenolysis. Subsequently, the free amine undergoes hydroxyethylation with ethylene oxide. The BOC group ensures that alkylation occurs selectively at the nitrogen atom without affecting the ester functionality, leading to the formation of the bis-hydroxyethyl intermediate. This precise control over reactivity is essential for maintaining the optical purity of the L-isomer, which is critical for the biological activity of the final drug substance.
The final transformation involves the conversion of the hydroxyethyl groups into the cytotoxic nitrogen mustard motif via chlorination with phosphorus oxychloride, followed by global deprotection. The mechanistic elegance of this route is most evident in the final hydrolysis step, where hydrochloric acid serves a dual purpose: it cleaves the BOC carbamate to regenerate the free amine and simultaneously hydrolyzes the ethyl ester to reveal the carboxylic acid. This convergent deprotection strategy significantly reduces the number of unit operations compared to stepwise deprotection methods. The resulting Melphalan is isolated through a careful pH adjustment and activated carbon treatment, which effectively removes colored impurities and trace metal residues from the hydrogenation catalyst. This rigorous purification protocol ensures that the final product meets the stringent specifications required for injectable oncology therapies, demonstrating the robustness of the high-purity Melphalan synthesis route.
How to Synthesize Melphalan Efficiently
Implementing this optimized synthetic route requires strict adherence to the reaction parameters defined in the patent to ensure reproducibility and safety at scale. The process is designed to be operationally straightforward, utilizing standard reactor equipment found in most fine chemical facilities. Key control points include the temperature management during the exothermic esterification and the careful handling of ethylene oxide during the hydroxyethylation phase. The use of activated carbon for decolorization in the final step is a critical quality control measure that should not be overlooked, as it directly impacts the visual appearance and purity profile of the API. For process engineers and R&D teams looking to adopt this technology, the following guide outlines the standardized operational framework derived from the patent examples, providing a clear roadmap from raw materials to the final crystalline product.
- Esterification of L-4-nitrophenylalanine with absolute ethanol and thionyl chloride.
- Protection of the amino group using di-tert-butyl dicarbonate (BOC anhydride) to form the stable N-BOC intermediate.
- Catalytic hydrogenation to reduce the nitro group to an amino group using Pd/C.
- Hydroxyethylation reaction with ethylene oxide to introduce hydroxyethyl groups.
- Chlorination using phosphorus oxychloride to convert hydroxyl groups to chloroethyl groups.
- Final acid hydrolysis to remove protecting groups and obtain pure Melphalan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this BOC-based synthesis route translates into tangible strategic advantages beyond mere technical superiority. The elimination of benzene and the reduction in solvent complexity directly correlate to a significant decrease in hazardous waste disposal costs, which are often a hidden burden in API production budgets. Furthermore, the high yields observed in the protection and reduction steps mean that less raw material is required to produce the same amount of final product, effectively lowering the cost of goods sold (COGS). The robustness of the BOC protection also implies a more forgiving process window, reducing the risk of batch failures and ensuring a more consistent supply of critical intermediates. This reliability is paramount for maintaining uninterrupted production schedules in the highly regulated pharmaceutical sector, where delays can have cascading effects on drug availability.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis eliminates the need for expensive and toxic reagents associated with phthalic anhydride protection. By achieving higher yields in the early stages of the synthesis, the process minimizes the accumulation of waste and maximizes the utilization of expensive chiral starting materials like L-4-nitrophenylalanine. Additionally, the use of common solvents like ethanol and ethyl acetate simplifies the supply chain for raw materials, reducing procurement lead times and volatility in pricing. The ability to perform deprotection and ester hydrolysis in a single pot operation further reduces energy consumption and labor costs, contributing to substantial overall cost savings without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as di-tert-butyl dicarbonate and thionyl chloride ensures that the supply chain is resilient to market fluctuations. Unlike specialized reagents that may have limited suppliers, the inputs for this process are produced globally in large volumes, mitigating the risk of shortages. The mild reaction conditions also reduce the wear and tear on manufacturing equipment, leading to lower maintenance downtime and higher asset utilization rates. This operational stability allows manufacturers to offer more reliable delivery commitments to their clients, fostering stronger long-term partnerships and enhancing the overall security of the pharmaceutical supply network.
- Scalability and Environmental Compliance: From an environmental perspective, this process represents a significant step forward in green chemistry. The removal of benzene from the solvent list eliminates a major regulatory hurdle and reduces the environmental footprint of the manufacturing site. The high efficiency of the reaction steps means less waste is generated per kilogram of product, simplifying effluent treatment and lowering compliance costs. Furthermore, the process has been explicitly validated for industrial production, demonstrating that it can be scaled from laboratory benchtop to multi-ton commercial batches without loss of efficiency or purity. This scalability ensures that the technology can meet growing global demand for Melphalan while adhering to increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Melphalan synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the competitive positioning of the resulting API. The answers reflect a commitment to scientific accuracy and practical applicability in a real-world manufacturing environment.
Q: Why is the BOC protection method superior to phthalic anhydride for Melphalan synthesis?
A: The BOC protection method eliminates the need for toxic benzene solvents and cumbersome two-step protection processes associated with phthalic anhydride. It offers milder reaction conditions, significantly higher yields (up to 95% in the protection step), and easier deprotection, making it more suitable for large-scale industrial production.
Q: What is the overall yield of this optimized Melphalan synthesis route?
A: According to the patent data, the optimized process achieves an overall yield of approximately 47.4%. Individual key steps, such as the amino protection and catalytic reduction, demonstrate exceptional efficiency with yields reaching 95% and 100% respectively, minimizing raw material waste.
Q: Is this synthesis method scalable for commercial API manufacturing?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes readily available reagents like absolute ethanol and di-tert-butyl dicarbonate, avoids hazardous solvents like benzene, and features robust purification steps such as activated carbon decolorization, ensuring consistent quality and safety at commercial volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Melphalan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex oncology drugs relies on a partnership grounded in technical excellence and supply chain integrity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of Melphalan meets the highest international standards. Our commitment to continuous improvement drives us to adopt innovative synthetic routes like the BOC-protection strategy, allowing us to deliver superior value to our global partners.
We invite you to collaborate with us to explore how this optimized synthesis route can enhance your supply chain resilience and cost structure. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our expertise can support your mission to deliver life-saving therapies to patients worldwide.
