Scaling High-Purity 4'-Demethylpodophyllotoxin PEG Derivatives via Microwave Catalysis
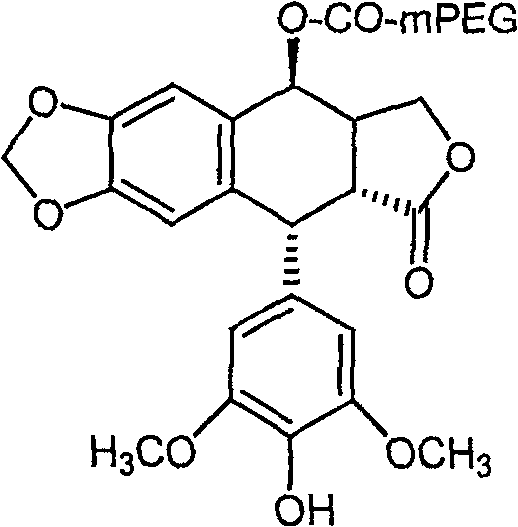
The pharmaceutical landscape for antitumor agents is constantly evolving, with a specific focus on improving the pharmacokinetic profiles of potent natural products like podophyllotoxin. Patent CN101314042B introduces a significant technological advancement in this domain by disclosing a novel water-soluble derivative of 4'-demethylpodophyllotoxin. This innovation addresses the longstanding challenge of poor aqueous solubility associated with many lignan-based anticancer compounds, which often limits their clinical efficacy and formulation options. By conjugating the active 4'-demethylpodophyllotoxin moiety with monomethoxy polyethylene glycol (mPEG) via an ester linkage, the invention creates a prodrug system that not only enhances solubility but also promises improved stability and controlled release mechanisms within the biological environment. This strategic modification represents a critical step forward for manufacturers seeking to develop next-generation chemotherapeutic intermediates with superior safety profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for modifying complex natural products like 4'-demethylpodophyllotoxin with high molecular weight polymers such as PEG are fraught with inefficiencies. Conventional esterification processes typically rely on prolonged heating under reflux conditions, often requiring hazardous organic solvents and extended reaction times that can span several hours to days. These thermal methods frequently suffer from low energy transfer efficiency, leading to uneven heating and potential degradation of the sensitive lignan scaffold. Furthermore, the steric hindrance presented by the bulky PEG chain often results in sluggish reaction kinetics, necessitating excessive amounts of coupling reagents and complicating the downstream purification process. The accumulation of byproducts, particularly urea derivatives from carbodiimide coupling agents, becomes a significant burden, requiring multiple recrystallization steps that erode overall yield and increase production costs substantially.
The Novel Approach
In stark contrast, the methodology outlined in the patent leverages microwave-assisted organic synthesis (MAOS) to overcome these kinetic and thermodynamic barriers. By utilizing direct dielectric heating, the process achieves rapid and uniform energy distribution throughout the reaction mixture, effectively activating the reactants without the need for excessive external thermal input. This approach allows for the efficient coupling of 4'-demethylpodophyllotoxin with carboxyl-functionalized mPEG in a remarkably short timeframe, typically completing the esterification within 25 to 35 minutes. The result is a streamlined workflow that not only accelerates production cycles but also enhances the selectivity of the reaction, minimizing side reactions and preserving the integrity of the chiral centers essential for biological activity. This shift from conventional thermal heating to microwave catalysis represents a paradigm shift in process chemistry for high-value pharmaceutical intermediates.
Mechanistic Insights into Microwave-Assisted Steglich Esterification
The core chemical transformation described involves a Steglich esterification mechanism, facilitated by the coupling agent dicyclohexylcarbodiimide (DCC) and the nucleophilic catalyst 4-dimethylaminopyridine (DMAP). In this pathway, DCC first reacts with the carboxylic acid group of the mPEG chain to form a highly reactive O-acylisourea intermediate. This activated species is then attacked by the phenolic hydroxyl group at the 4'-position of the demethylpodophyllotoxin, a step that is significantly accelerated by the presence of DMAP. DMAP acts as an acyl transfer agent, forming an even more reactive acylpyridinium ion that readily undergoes nucleophilic attack by the sterically hindered phenol. The application of microwave irradiation further enhances this mechanism by lowering the activation energy required for the formation of these transition states, thereby driving the equilibrium towards the desired ester product with high efficiency.
From an impurity control perspective, the management of the dicyclohexylurea (DCU) byproduct is critical for ensuring the purity of the final API intermediate. The patent details a robust purification strategy wherein the insoluble DCU is initially removed via filtration after the reaction mixture is cooled and treated with anhydrous methylene chloride. Subsequent washing steps involving dilute acetic acid in THF and hydrochloric acid solutions are employed to remove residual DMAP and any unreacted starting materials. This multi-stage purification protocol ensures that the final white solid product meets stringent quality standards, free from toxic coupling residues that could compromise patient safety. The ability to effectively separate these impurities while maintaining high recovery rates is a testament to the robustness of the proposed synthetic route.
How to Synthesize 4'-Demethylpodophyllotoxin-PEG Derivative Efficiently
The synthesis of this water-soluble prodrug is designed for operational simplicity and scalability, making it highly attractive for industrial adoption. The process begins with the precise stoichiometric mixing of the key reagents: 4'-demethylpodophyllotoxin, carboxyl monomethoxy polyethylene glycol (with a molecular weight range of 2000 to 20000), DCC, and DMAP. These components are homogenized in a suitable reaction vessel before being subjected to microwave irradiation at a power level of approximately 510W. The reaction progress is monitored by the cessation of globule formation, indicating the completion of the esterification. Following the reaction, a systematic workup involving solvent evaporation, precipitation with absolute ether, and rigorous washing ensures the isolation of the pure derivative. For a detailed, step-by-step breakdown of the standardized operating procedures and specific parameters, please refer to the technical guide below.
- Mix 4'-demethylpodophyllotoxin, carboxyl monomethoxy polyethylene glycol (mPEG-COOH), DCC, and DMAP in a reaction vessel.
- Subject the homogeneous mixture to microwave irradiation (approx. 510W) for 25-35 minutes to complete esterification.
- Purify the crude product via filtration, washing with acetic acid/THF and HCl, followed by recrystallization in isopropyl alcohol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microwave-catalyzed synthesis offers tangible benefits that extend beyond mere technical novelty. The drastic reduction in reaction time translates directly into increased throughput capacity, allowing manufacturing facilities to produce larger batches of high-value intermediates within the same operational window. This efficiency gain is crucial for meeting the demanding delivery schedules of global pharmaceutical clients who require reliable access to complex oncology building blocks. Furthermore, the simplified purification process reduces the consumption of solvents and consumables, contributing to a leaner and more cost-effective production model that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The elimination of prolonged heating periods and the reduction in solvent usage significantly lower the operational expenditures associated with energy and waste disposal. By achieving high conversion rates in a single step, the process minimizes the loss of expensive starting materials like 4'-demethylpodophyllotoxin, thereby optimizing the overall material cost per kilogram of finished product. Additionally, the reduced need for extensive chromatographic purification lowers the dependency on costly silica gel and eluents, further driving down the total cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of the microwave method ensures consistent batch-to-batch quality, reducing the risk of production failures that can disrupt supply chains. The use of commercially available reagents such as mPEG-COOH and standard coupling agents means that raw material sourcing is straightforward and less susceptible to geopolitical or market volatility. This reliability allows supply chain planners to maintain lower safety stock levels while still guaranteeing on-time delivery to downstream customers.
- Scalability and Environmental Compliance: The process is inherently scalable, as microwave technology has advanced to support continuous flow and large-batch processing suitable for commercial production. The reduced generation of hazardous waste and the efficient use of resources make this method more compliant with increasingly strict environmental regulations. This sustainability advantage not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized pharmaceutical intermediate. Understanding these details is essential for R&D teams evaluating the feasibility of incorporating this derivative into their drug development pipelines. The answers provided are derived directly from the experimental data and technical specifications outlined in the patent documentation.
Q: What is the primary advantage of using microwave catalysis for this esterification?
A: Microwave catalysis significantly accelerates the reaction rate, reducing processing time from hours to approximately 30 minutes while maintaining high yields (over 81%) and simplifying the workup procedure compared to conventional heating methods.
Q: Why is PEGylation critical for 4'-demethylpodophyllotoxin derivatives?
A: Native podophyllotoxins often suffer from poor water solubility and high toxicity. PEGylation creates a water-soluble prodrug that improves bioavailability, reduces toxic side effects, and allows for controlled release via enzymatic hydrolysis in the body.
Q: What represent the key impurities in this synthesis and how are they removed?
A: The primary byproduct is dicyclohexylurea (DCU) formed from the coupling agent DCC. It is effectively removed through filtration and subsequent washing steps with dilute acid and organic solvents, ensuring high purity of the final white solid product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4'-Demethylpodophyllotoxin Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We are committed to maintaining stringent purity specifications and operate rigorous QC labs to verify that every batch of 4'-demethylpodophyllotoxin derivative meets the highest international standards. Our expertise in microwave-assisted synthesis allows us to offer this complex intermediate with superior consistency and competitive lead times.
We invite you to collaborate with us to optimize your supply chain for antitumor drug development. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and manufacturing goals, ensuring a seamless transition from laboratory scale to commercial success.
